Q: The volume of a sample of hydrogen gas was decreased from 13.82 L to 7.11 L at constant temperature.…
A: Given,initial volume of hydrogen gas = 13.82 Lfinal volume of hydrogen gas = 7.11 LFinal pressure…
Q: Consider the following reaction: 2Mg(s) + O2(g) →2MgO(s), AH=-1204 kJ endothermic Submit Previous…
A:
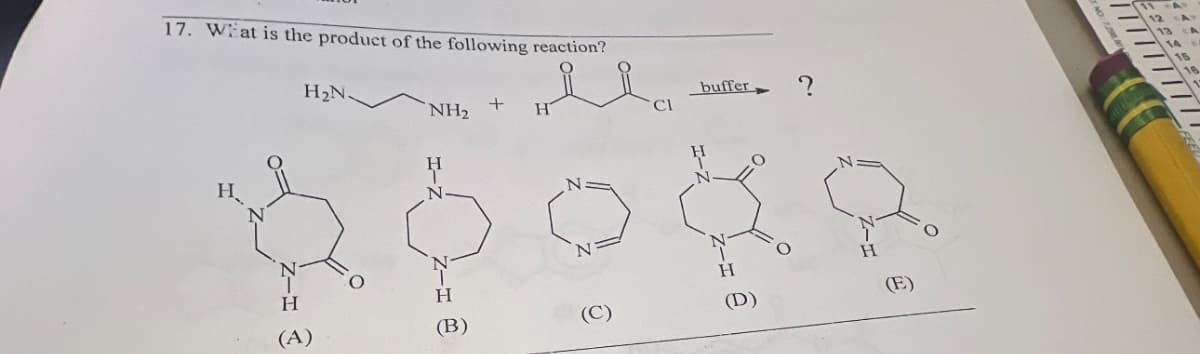
Q: Draw the major product of this reaction. Ignore inorganic byproducts and the carboxylic acid side…
A: Given reaction:We have to draw the major product of this reaction.
Q: Calculate the solubility at 25 °C of CuBr in pure water and in a 0.0140M CoBr, solution. You'll find…
A: Given that,The concentration of solution =Ksp value of Molar mass of Then,
Q: 9.46 Draw the substitution product formed (including stereochemistry) when (R)-hexan-2-ol is treated…
A: Here is your answer.......Explanation:If you have any doubt please comment below I will…
Q: For the reaction 2A(g) <===> B(g)+2C(g), a reaction vessel initially contains only A at a…
A: Chemical equation is 2A(g)↔B(g)+2C(g)Equilibrium constant Kp for above chemical equation is…
Q: This is a synthesis problem. shown below is the synthesis of the target compound on the right from…
A:
Q: right? is the relationship between the parent structure on the left and the structure on the COOH HO…
A:
Q: Use the Henderson-Hasselbalch equation to calculate the pH of each solution: a solution that is 0.28…
A: The objective of this question is to calculate the pH of a solution using the Henderson-Hasselbalch…
Q: Curved arrows are used to illustrate the flow of electrons. Using the provided starting structure,…
A: In this reaction, we have to reduce carbonyl compound (i.e ketone) by grignard reagent.
Q: One cup of fresh orange juice contains 139 mg of ascorbic acid (vitamin C, C6H8O6). Part A Given…
A: Given:Mass of ascorbic acid in one cup of fresh orange juice = 139 mg = 0.139 g Volume of one cup =…
Q: Identify the Major and ALL Minor product(s) that are expected for each of the following reactions.…
A: Given reaction fallows Elimination and substitution reaction.
Q: For the given SN2 reaction, draw the organic and inorganic products of the reaction, and identify…
A:
Q: 1 a Predict the direction in which each of the following equilibria will shift if the pressure on…
A: According to Le Chatelier's principle if some factors (like temperature, pressure, molar…
Q: Consider the following reaction: 2HI(g) = H2 (g) + I2 (g) If 2.99 moles of HI(g) , 0.359 moles of…
A: The objective of the question is to calculate the equilibrium constant, Kc, for the given reaction…
Q: 9
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: When the following equation is balanced properly under acidic conditions, what are the coefficients…
A: Overall Reaction: 8H2O(l)+7Fe(aq)2++2Cr(aq)3+→2CrO4(aq)2++7Fe(s)+16H(aq)+Cr3+ : 2Fe2+ :…
Q: Consider the product of the following SN 2 reactions. Part 1 of 2 Draw the major organic product…
A: SN2 reaction is bimolecular nucleophilic substitution reaction. The rate of reaction is depends upon…
Q: Draw the major organic product of the reaction below. H3C H KOH, H₂O CH3
A:
Q: Draw an alkyl bromide that would produce the following alkene through an E1 elimination reaction.…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: 1) Base, Alcohol 2) H3O+
A: The question is based on organic reactions.We need to identify the product and explain its…
Q: Predict the major product for the following reaction, include Stereochemistry of appropriat a. NaBH4…
A: The objective of the question is to predict the major product of a series of reactions involving…
Q: For each of the following strong base solutions, determine [OH−],[H3O+][OH−],[H3O+], pHpH, and…
A: The objective of the question is to determine the concentration of hydroxide ions [OH-], hydronium…
Q: Draw the x anomer product of this reaction. Ignore inorganic byproducts. H H HO HO HO H H OH Н -HI…
A: We have to predict the product.
Q: 9.60 Draw the products of each reaction. a. CH3CH2OH H₂SO4 b. ن C. d. [1] CH3CH₂O- Na+ Ο [2] H₂O HBr…
A: The required answer is given below-Explanation:Step 1:Step 2: Step 3: Step 4:
Q: The pH of a solution is 3.42. What is the OH⁻ concentration in the solution?
A: The objective of this question is to find the concentration of hydroxide ions (OH⁻) in a solution…
Q: HO для aqueous H₂SO + NaCN •You do not have to consider stereochemistry.
A: Given reactionWe have to find the product.Note:Do not have to consider stereochemistry
Q: Question 6. Predict the product of the following reaction and draw the mechanism. Includes all…
A: In electrophilic aromatic substitution, the hydrogen of the benzene is replaced by an electrophile.
Q: How would you use a Grignard reaction of an aldehyde or ketone to synthesize 3-pentanol? Draw the…
A:
Q: 3. Draw two different mechanisms to account for the following transformation under acidic conditions…
A: In the given reaction, there is an aldehyde, a ketone, and a secondary amine group which react to…
Q: What is the photon energy of light with a wavelength of 399 nm? Note that h = 6.626 × 10-34 J's and…
A: Given:Wavelength of light = 399 nm = 399 x 10-9 m = 3.99 x 10-7 mPlanck's constant (h) = 6.626 x…
Q: Draw structural formulas for the a,ẞ-unsaturated aldehyde or ketone and the lithium diorganocuprate…
A: The product formed is given as,
Q: Which of the following diseases is associated with: symptoms of skin sensitivity, malaise,…
A: The objective of the question is to identify the disease associated with the given symptoms: skin…
Q: 10.47 What alkene can be used to prepare each alkyl halide or dihalide as the exclusive or major…
A: As images cannot be uploaded to this section, see below for the answers:Explanation:The answers are…
Q: a. Both the reactant and product show very similar IR spectra. For the reactant, indicate which IR…
A: The objective of the question is to interpret the IR spectrum of the acetanilide and 4-bromo…
Q: The titration of 20.0-mL sample of 0.105 M Succinic Acid (Ka= 6.16 X10^-5) with 0.125 M NaOH. The…
A: Here's my answer step by step,read carefully to follow. The questions are bit tricky and I hope my…
Q: help?
A: a). hljbb). hlnaExplanation:Step 1: (a) The first step, reduction of aldehyde into alcohol then…
Q: Q, Rotate your molecule so that you can see what the rat is looking at below. Draw the Newman…
A: The dot represents the front carbon and circle represents the back carbon.
Q: In the provided boxes, provide synthetic sequences (including reagents used and the compound…
A: The objective of this question is to convert a given compound into a specified target compound…
Q: Problem 8 of 14 Submit Curved arrows are used to illustrate the flow of electrons. Using the…
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: Propenoic acid, C3H4O2, is a reactive organic liquid that is used in the manufacturing of plastics,…
A: Mass of propenoic acid combusted = 0.338 gMass of water = 0.237 gMass of carbon dioxide = 0.517 gWe…
Q: Question 12. Provide a synthetic pathway, i. e. reaction conditions and expected intermediate…
A: The question is based on the concept of organic synthesis.We need to synthesize the product using…
Q: Question 23 Identify the Major and ALL Minor product(s) that are expected for each of the following…
A:
Q: Consider the following compound: Part 1 of 2 Given that syn addition of H₂ occurs from both sides of…
A: Alkenes are hydrogenated by the reaction with hydrogen in the presence of finely divided nickel,…
Q: OLİ OEt, THF, -78 °C 2) NH4Cl, H₂O OEt 1)
A: Final answer is given in explanation please see from there.Explanation:Approach to solving the…
Q: For each of the following strong base solutions, determine [OH−],[H3O+],pH, and pOH. Determine…
A: The objective of the question is to determine the concentration of hydroxide ions [OH−], hydronium…
Q: Draw the major product of this reaction. Ignore inorganic byproducts. Sn/HCI NO2
A: Given reaction:We have to find the major product
Q: HO ??? LOH
A: The given conversion is from propanol to butanoic acid . Propanol ----------> Butanoic acid .
Q: Draw a structural formula for the major organic product of the following reaction:
A: When an alkene reacts with Br2 in the presence of CH2Cl2, a dibromo alkane will be formed.This…
Q: Select the correct product for the oxidation reaction below. OH HO Na Cr₂O OH H₂SO
A: The oxidation of alcohol depends on the number of hydrogen atoms attached to the carbon atom that…
Step by step
Solved in 3 steps with 2 images

- 16. If all the N in 10.0 mmols urea, CO(NH2)2, is converted to NH4HSO4, and if with excess NaOH the NH3 is evolved and caught in 50.0 mL of HCl (1.00 mL ≈ 0.03000 g CaCO3), what volume of NaOH (1.00 mL ≈ 0.3465 g H2C2O4.2H2O) would be required for complete titration? 17. The percentage of protein in meat products is determined by multiplying the %N as determined by the kjeldahl method by the arbitrary factor 6.25. A sample of processed meat scrap weighing 2.000 g is digested with concd H2SO4 and Hg (catalyst) until the N present has been converted to NH4HSO4. This is treated with excess NaOH, and the liberated NH3 is caught in 50.0 mL pipetful H2SO4 (1.000 mL ≈ 0.01860 g Na2O). The excess acid requires 28.80 mL NaOH (1.000 mL ≈ 0.1266 g KHP). Calculate % protein in the meat scrap. 18. A 2.00 g sample of steel is burned in O2, and the evolved CO2 after passing through appropriate purifying trains is caught in 100 mL Ba(OH)2 solution. The supernatant liquid requires 75.0 mL HCl (1.00 mL…What is the pH of a buffer that is 0.12 M in lactic acid [CH3CH(OH)COOH, or HC3H5O3] and 0.10 M in sodium lactate [CH3CH1OH2COONa or NaC3H5O3]?For lactic acid, Ka = 1.4 * 10-4.What is the pH of the ethanoic acid solution CH3COOH 0.15 M with Ka = 1.8 × 10-⁵? b) What will be the pH when in 82 liters of 0.15 M solution of this acid we add 82 grams of sodium ethanoate CH3COONa?
- What volume of 0.317 M KOH solution will be required to titrate 25.00 ml of 0.285 MHNO3?What is the pH of the solution formed when 1.13 g of diethylammonium chloride ((CH3CH2)2NH2)+Cl- is added to 100.0 mL of 0.754 M diethylamine ((CH3CH2)2NH)?When H2SO4(aq) was reacted with NaOH(aq) in the titration, why was a precipitate not formed?
- The Tris buffer system is commonly used in biochemistry as its pKa of 8.1 allows it to buffer close to physiological pH. (CH2OH)3CNH2 + H+ Û (CH2OH)3CNH3+ What concentrations of Tris and Tris H+ are obtained in a 200 mL solution at pH 8.20 containing 6.1g of Tris? The molecular mass of Tris is 121.1 gmol-1 Please do this step by step thank you!8. The addition of acetic acid and ________ to water produces a buffer solution. A. HC2H3O2 B. KI C. KC2H3O2 D. NaNO3 E. NaBrWhat is the concentration of free Ag+(aq) in a solution that is initially 0.01254 M in AgNO3 and 1.04842 M NH3? These two compounds react together to form [Ag(NH3)2]+. Kf([Ag(NH3)2]+) =1.600e7 1.023 7.484e-10 0.01254 6.250e-8
- What is the pH of a 0.1848 M H2NCH2COONa? Ka for H2NCH2COOH is 4.50e-3The desired pH is 6.4 in creating a buffer system/buffer solution of about 50-60mL. Therefore, a student picks Silicic Acid Si(OH)4, as 1mL of 1M of the acid has a pH of 6.4, which also has a pKa of 9.82. What is the ratio HA/A- and ratio A-/HA-? The buffering capacity will be checked by the addition of 1mL of 1M HCl or 1mL of 1M NaOH and should not change by more than 0.25pH units.Tris(hydroxymethyl)aminomethane [(HOCH2)3CNH2—Tris, or THAM] is a weak base frequently used to prepare buffers in biochemistry. Its Kb is 1.2 × 10−6 and pKb is 5.92. The corresponding pKa is 8.08, which is near the pH of the physiological buffers, and so it exhibits good buffering capacity at physiological pH. What weight of THAM must be taken with 100 mL of 0.50 M HCl to prepare 1 L of a pH 7.40 buffer?

