Q: NBS CCI4
A:
Q: A secondary step in the process to produce ultra-pure silicon is to combine silicon tetrachloride wi...
A:
Q: 11. The net reaction for formation of HI from H2 and I2 is H, +I, 2 2HI The following mechanism was ...
A:
Q: Identify the peaks and structer.
A: 13C-NMR shows an non-equivalets of the carbons in the spectrum. The number of signals in the spctrum...
Q: Draw the structure of the following compounds. Use line-angle structural formula. 1-bromo-4-ethyl-5-...
A:
Q: Identify the molecular orbital indicated by the arrow. Answer in this format: sigma sp3-sp3 In case ...
A:
Q: Describe how you would prepare 100 m L of a solution that is 25 % acetic acid by volume .
A: Given Volume % = 25% Volume of a solution = 100 mL Method of Preparation of 25% acetic acid by...
Q: E2B.3(a) The constant-pressure heat capacity of a sample of a perfect gas was found to vary with tem...
A:
Q: 2. Consider the following equilibrium process: co,(g) + H,(g)=C() + H,0(g) At equilibrium, [Co] = 0....
A:
Q: 3. 4. 500 mL of 0.75 N of K2Cr207 from a pure solid reagent if it follows the following half reactio...
A: In this type of question first thing should be balance the half reaction. Cr2O72- ...........> Cr...
Q: A children's liquid medicine contains 100 mg of the active ingredient in 5 mL. If a child should rec...
A: Given, A children's liquid medicine contains 100 mg of the active ingredient in 5 mL. Therefore, t...
Q: How many grams of Mg are there in a sample of Mg that contains 6.21x1023 atoms? grams
A: The amount of Mg present can be calculated as
Q: Please explain thoroughly. Among H2O, CH3F, Cl2, and Xe… Which has the strongest London dispersion f...
A: There are different types of forces act between the molecules:- Hydrogen bonding Dipole-dipole attr...
Q: ОН H2N, CH2CH3 CH2NH2 HgCO,,, HOOC,,, СООН CHO HOH2C НО A H2NH2C OH НООС. E `NH2 D H3CSH2C
A:
Q: A mixture of calcium oxide, Cao, and calcium carbonate, CacO 3, that had a mass of 3.454 g was heate...
A:
Q: 1. Which match is correct for thermal [2+2] cycloaddition reaction (a) suprafacial /suprafacial →All...
A: [2+2] cycloaddition
Q: Calculate the concentration equilibrium constant, K’sp for La(IO3)3 in a saturated solution of La(IO...
A: For a sparingly soluble salt, there is a dynamic equilibrium between the dissociated ions and undiss...
Q: . Based from the molecular structure below, which shows the weakest Dispersion Forces? II H HH H C. ...
A: Note : As the size of the molecule (molar mass) increase or the longer the carbon ttje magnitude of...
Q: Draw the expanded structural formula of an organic compound with sixteen carbons using the following...
A: Solution :- Here C1 and C2 has bond order 3 means it has triple bond and C4 form single bond with C...
Q: If 20.0 g of each reactant were used in performing the following reaction, which would be the limiti...
A:
Q: A certain reaction is second order in N, and first order in H,. Use this information to complete the...
A:
Q: How many mL of 15.8 M nitric acid should be diluted to 0.250 L to make 3.00 M HNO3?
A: Given Initial Molarity of HNO3 ( M1 ) = 15.8 M Final Volume ( V2 ) = 0.250 Liter ...
Q: The pectral data below i8 for a Compaund once OSed as a riot Control agunt, that is no longer due to...
A:
Q: 5. Explain the trend in binding constants (in CH,Cl2) for the complexation of phenol derivatives by ...
A: Solution -
Q: A compound is found to contain 37.32 % phosphorus, 16.88 % nitrogen, and 45.79 % fluorine by mass. W...
A: Empirical formula of a compound can be defined as the simplest whole number ratio of atoms present ...
Q: %. The percent by mass of bromine in PNBR2 is (Enter your answer to four significant figures.)
A:
Q: 1. How many MOLES of dinitrogen monoxide are present in 1.90 grams of this compound ? moles. 2. How ...
A: Mole concept: The number of molecules or atoms present in the one mole of the substance is equal to ...
Q: A student is about to dissolve 1.1 g of compound Z in solvent Y. Is 100 mL of solvent Y already enou...
A: Solubility of Z = 3.44 g/L So, 1 L Y : 3.44 g Z
Q: Under certain conditions the rate of this reaction is zero order in hydrogen iodide with a rate cons...
A:
Q: ОН A `NH2 NH2 Which labelled carbon (A or B) is chiral? (A) B B) A
A:
Q: For the reaction 3A(g) + 3B(g) C(g) K. = 76.4 at a temperature of 113 °C . Calculate the value of Kp...
A: Equilibrium constant is defined as products concentrations divided by reactant concentrations to the...
Q: What is the effect of the ratio of conjugate base to acid on buffering capacity?
A: In essence buffer solutions resist changes in pH. A buffer contains appreciable amounts of a weak ac...
Q: Read carefully the given partial names in IUPAC alkane nomenclature and check the columns where they...
A: answer :
Q: This method is to be used when measuring the volume of a solution containing sufficient reagent to r...
A: In volumetric method, to determine the concentration of analyte it is reacted with a suitable reage...
Q: Draw the product that could be formed when 2,3- dimethylbuta-1,3-diene reacts with (E)-2-butenedinit...
A:
Q: t a certain temperature this reaction follows second-order kinetics with a rate constant of 1.47M CI...
A: Given Reaction followed second order kinetics
Q: 3 complete total synthesis of Delta9 THC (all the steps of the mechanism)
A: Total synthesis of Delta9 THC: Total synthesis: Total synthesis is the complete chemical synthesis...
Q: Determine the point group of the following molecule: Fe
A: The given compound is Ferrocene. It is a staggered compound.
Q: What is sampling in analytical chemistry?
A: Analytical chemistry is a branch of chemistry in which we analyze the sample by different techniques...
Q: 4.Write the products of the reactions below showing stereochemistry and regiochemistry (you may writ...
A:
Q: 1. Which of the following isn't an example of an oxidation-reduction reaction? (a) CasP2(s) + 6 H2O(...
A:
Q: The chemicals below are incompatible with each other. A companyhas to store thechemicals in separate...
A: Serial number Chemical Incompatible with 1 Ammonia Silver, Mercury, Chlorine, Calcium hypochlori...
Q: An unknown substance was subjected to boiling point determination. Temperature is recorded at 75℃ wh...
A: In the method of finding boiling point of substance using capillary tube, when the bubbles escape ri...
Q: Write the ionization constant expression for a 50 mM aniline solution (pKb=9.40 @ 25 C).
A:
Q: 1. In a combustion process of a coal-fired power plant, 20% excess of the theoretical amount of Oxyg...
A: Answer is below
Q: Convert 0.05 ppmv of SO2 in the air to μgSO2m3airμgSO2m3air. Assume atmospheric pressure and a tempe...
A: We have to convert, 0.05 ppmv of SO2 in the air to μgSO2 /m3 air
Q: which aqueous solution will have the lowest freezing point
A: Ionic aqueous electrolyte solutions have the lowest freezing point. The freezing point is a colligat...
Q: How does the following affect the acidity or basicity of compounds? Explain clearly and thoroughly. ...
A: Answer is :
Q: The 3D image below is that of an allylic carbocation intermediate formed by the protonation of a con...
A: The addition of HBr to conjugated dienes can lead to 1,2 (Kinetic product) or 1,4 addition products ...
Q: Complete this table for the reaction T(k) [N2] [H2] [NH3] [Kc] 500 0.115 0.105 0.439 575 0.110 ...
A: KC can be calculated by writing the balanced equation.

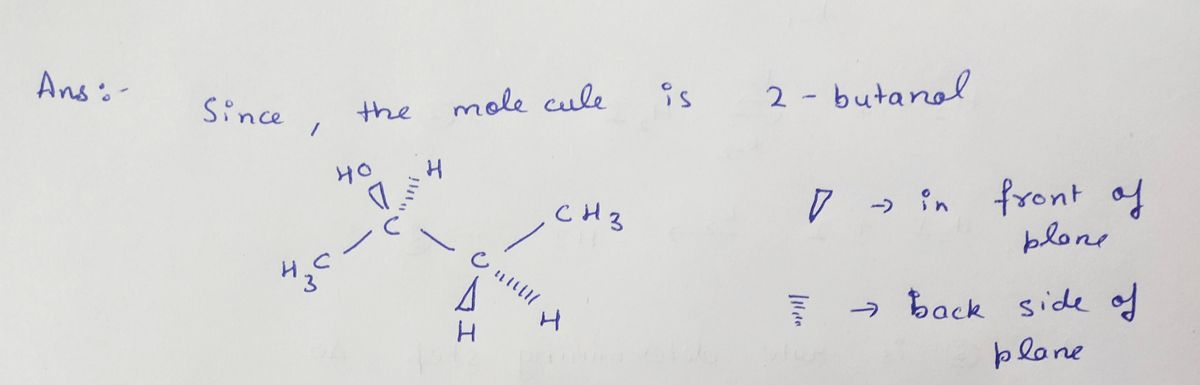
Step by step
Solved in 2 steps with 2 images

- Naming Alkenes 7-37 Name the following alkenes: (a) CH3 (b) CH3 CH2CH3 (c) CH2CH3 CH3CHCH2CH,CH CH3 H2C=CCH2CH3 CHCH2CH3 C=C H C=C H3C H3C H. H (f) H2C=C=CHCH3 (е) CH3 C=C (d) H H3C C=C CH3 C=C CH3 H3C H. H2C=CHCHCH CH3CH2CH2 CH37-37 Name the following alkenes: (a) CH3 (b) CH3 CH2CH3 (c) CH2CH3 CH3CHCH2CH2CH CH3 H2C=CCH2CH3 H CHCH2CH3 c=C c=C H3C H H3C (d) H CH3 (e) H H. (f) H2C=C=CHCH3 c=c H2C=CHCHCH H3C H3C c=C CH3CH2CH2 C= H CH3 CH3 CH3Identify the correct structure below that represents the following molecule: (3E,5 E)-6-chloro-3,5-octadien-1-ol CI HO CI OH CI OH CI OH CI OH
- 3) What is the approximate pKa of the hydrogen indicated by the arrow below: A) -7 B) 0 C) 5 D) 10 E) 15 ο ο ο ο ο O(A) O (B) O (C) O (D) O (E) Question 4 OH 4) Which of the following represents a correct Newman Projection sighting down the C molecule shown in the Box below: eye4-44 Draw the important resonance forms of the following free radicals. (a) CH₂=CH-CH₂ (b) (d) (e) -CH₂ (c) (f) CH₂-C-0.1. Identify the functional group in the following molecules. Circle them on the structures and mark which functional groups they have. (a) Ⓡ +H₂N-CH- HO CO CH₂ C=0 NH₂ C-H H-C-OH CH₂OH OH H OH Vitamin C -C-CH₂OH H₂CO (b) H₂CO CH3 (f) _NH CH HO-C-H HN COOH C=O CH₂ H-C-OH CH3 H-C-OH H-C-OH CH₂OH Nicotinic acid (niacin) OH Coenzyme Q (c) (d) CH,-O- 1-(CH₂) HỌ–CH CH3 (CH₂-CH=C-CH₂) 10H +H₂N-CH- O CH,—O- -OH OH مل CH₂ SH -(CH₂)14-CH3
- 5-56 Identify the indicated hydrogens in the following molecules as pro-R or pro-S: (a) (b) (c) H H H H CO2H CH3S. CO, .CO2 HO2C HS но н H H H3N H H3N H Malic acid Methionine CysteineBr LiCu(CH3)2 6 (1) LiAlH4 2 3 Ahol follow (2) H₂O 0 Aci HBr (xs) H (2) H₂O+ -Li3-39 The following names are all incorrect or incomplete, but they represent real structures. Draw each structure and name it correctly. (a) 2-ethylpentane (d) 2-dimethylbutane (b) 3-isopropylhexane (e) 2-cyclohexylbutane (c) 5-chloro-4-methylhexane (f) 2,3-diethylcyclopentane
- 3) Add curved arrows to the following polar reactions to indicate the flow of electrons in each: (al + H-N-H H--N-H (b) CH-ö H-C-Br: CH-ö-CH3 (e) H3CThe correct configurations of the carbons indicated by 1, 2 and 3 below are: CH3 2 Br" ''OH 1 3 (a) 1=R, 2=R, 3=S (b) 1=S, 2=R, 3=R (c) 1=R, 2=S, 3=S (d) 1=S, 2=S, 3=R (e) None of the above2-38 Draw as many resonance structures as you can for the following species: (a) :0: (b) (c) :NH2 + .. H3C-C-CH2- H2N-C=NH2 H (d) H3C-S-CH2 (e) H2C=CH-CH=CH-CH-CH3

