Q: The pH of swimming pools is made slightly basic by spreading solid Na2CO3 across the surface.…
A: Answer:- This question is answered by using the simple concept of calculation of ph of a slat…
Q: How the aqueous chemistry of amphoteric hydroxides involves precipitation, complex-ion formation,…
A: The aqueous solution is the one in which the solvent is water. Different chemical reactions can be…
Q: During a titration, the pH of an analyte solution containing HA(aq) is 5.56 and the ratio of (A…
A: Given pH of solution is 5.56 ratio of [A-]/[HA] is .842 we have to determine Ka
Q: If the partial pressure of the carbon dioxide in the gas in contact with leachate within a landfill…
A: Solutions- partial pressure of the carbon dioxide in the gas in contact with leachate within a…
Q: d. During the fermentation of wine, a buffer system consisting of tartaric acid and potassium…
A: Given that, during the fermentation of wine, a buffer system consisting of tartaric acid and…
Q: Write the whole acid base equation for the reaction of KOH(aq) with HC4H7O2(aq) b) write the net…
A: (a) Acid base equation KOH(aq) + HC4H7O2(aq) → KC4H7O2(aq) + H2O (l) (b) Net ionic…
Q: The pH of an aqueous solution of 0.223 M sodium hypochlorite, NaCiO (aq), is This solution is (
A:
Q: The pH of an aqueous solution of 0.124 M ammonium nitrate, NH,NO3 (aq), is This solution is
A: NH4+ ion released H+ ion in water so concentration of H+ ion is [H+] = √(Kw × C / Kb ) Where, Kw =…
Q: Carbon dioxide dissolves in water to form carbonic acid, which is primarily dissolved CO,. Dissolved…
A: Given partial pressure = 6.8x10-4 bar. Atmospheric pressure = 0.9869 bar. Determine the mole…
Q: The pH of an aqueous solution of 0.134 M sodium fluoride, NaF (aq), is This solution is
A: Answer:- This question is answered by using the simple concept of calculation of pH of aqueous…
Q: Calculate the pH, pOH and [OH ] of a 0.0420 mol/L a strong acid, HNO, (aq) solution.
A:
Q: Sodium hydroxide is a strong electrolyte and hydrates in water according to the following equation:…
A: Ph Based question first find pOH value then pH
Q: A buffer solution with a pH of 4,9, consists of 0,25 mol∙dm–3 acetic acid (HC2H3O2; Ka= 1,8 x 10–5)…
A:
Q: Determine the pH of a 0.18 M solution of pyridinium nitrate (C5H5NHNO3) at 25 ° C. [Pyridinium…
A: Given,Molarity of pyridinium nitrate (C5H5NHNO3) = 0.18 MKb = 1.7 × 10−9The value of Ka can be…
Q: Estimate the pH of 0.010 M NaCH3CO2(aq).
A:
Q: Calculate the pH at the stoich iometric po int of the titration of 25.00 cm3 of 0.150 M lactic acid…
A:
Q: The pH of an aqueous solution of 0.021 M hydrosulfuric acid, H2S (aq), is .
A: H2S is a weak diprotic acid. Since the Ka2 of H2S is << Ka1 of H2S Hence we can assume only…
Q: The pH is measured to be 2.58 for a solution of 0.035 M cyanic acid, HOCN (aq). Write the hydrolysis…
A: HOCN is a weak acid. Weak acid are those acid which does not dissociates completely in equilibrium.…
Q: What is the molar solubility of silver chloride in 0.65 M NH3(aq)? Solubility
A: “Since you have asked multiple question, we will solve the first question for you. If youwant any…
Q: For the reaction - H2 (g) + I2 (s) = HI (g) at 25°C and 1 atm, the equilibrium mixture contained 39%…
A: Given the reaction is: 12H2(g) +12I2 (s) = HI (g) For this, equilibrium constant expression is: K =…
Q: The Ka value for acetic acid, CH3COOH(aq), is 1.8×10−5. Calculate the pH of a 2.80 M acetic acid…
A:
Q: Estimate the pH of a solution of 0.50 M HCI(aq), assuming ideal behaviour. The mean activity…
A: Deviation from ideal behavior can be considered by the electrostatic force of attraction between…
Q: i Calculate the pH, pOH, and fraction of solute protonated or deprotonated in the following aqueous…
A: Hello. Since the question contains more than three sub-parts, the first three sub-parts shall be…
Q: Sketch the pH curve of a solution containing 0.10 M NaCH3CO2(aq) and a variable amount of acetic…
A: Acetic acid is a weak acid and the given salt NaC H3CO2 (aq) is salt of the weak acid that is of…
Q: A buffer solution of volume 100 cm3 consists of 0.10 M CH3COOH(aq) and 0.10 MNaCH3CO2(aq). (a) What…
A:
Q: Calculate the pH of 0.042 M HNO2 solution (Ka = 4.5 x 10 "). HNO, (aq) H' (aq) +NO; (aq)
A:
Q: 10p in recent years, technology has developed considerably in analytical devices as in many areas…
A:
Q: What will happen to the pH and alkalinity of the water as alum is added? Justify your answer.
A: Alkalinity is a measure of ability to neutralize acid. Alkalinity levels are expressed as…
Q: Determine the pH of the dissociation of 0.1 M HCN (aq). For which the dissociation constant is equal…
A: Given data contains, Molarity of HCN is 0.1M. Dissociation constant is 4.9×10-5. Weight of the…
Q: what is acid-base equilibrium or electrochemistry? what are its practical applications?
A:
Q: The pH of a 0.016-M aqueous solution of p-toluidine (CH3 CgH,NH2 ) is 8.60. Calculate Kp. K,
A:
Q: The pH of an aqueous solution of 0.175 M ammonium iodide, NH¼I (aq), is This solution is
A: Given : Concentration of NH4I = 0.175 M Since NH4I is made up of weak base NH3 and strong acid HI.…
Q: The molar concentration of H3O+ ions in the following solutions was measured at 25 °C. Calculate the…
A: pH is used to measure acidity or basicity of an aqueous solution. pH of any solution can be…
Q: Calculate the pH of a 0.100 M HC2H3O2(aq) solution.
A: The acid in the given problem is depicted by the formula C2H3O2H is acetic acid . It can be written…
Q: When acidulated water (dil.H2SO2solution) is electrolysed, will the pH of the solution be affected?…
A: Anode reaction is given
Q: The pH of an aqueous solution of 0.223 M sodium hypochlorite, NaC1O (aq), is This solution is…
A: pH of salt produced from reaction of weak acid and strong base would be basic and pH of salt…
Q: The pH of an aqueous solution of 0.0103 M hydrosulfuric acid, H2S (aq), is
A: Given, Molarity of H2S = 0.0103 M First acid dissociation constant of H2S = Ka1 = 1.0 x 10-7…
Q: What is the pH of 3.5 × 10−4 M HNO3 (aq)?
A: An acidic substance is the substance that can give H+ ions. They are corrosive and sour in nature.…
Q: What is the pH at 25 degrees Celsius of a saturated solution of magnesium hydroxide (the active…
A: Since the solubility product (i.e Ksp ) of magnesium hydroxide = 4 X 10-12 And the dissolution…
Q: 98 grams of sodium acetate (Mol. wt =82 g/mol) were placed in 764 mL of water such that the final…
A:
Q: Nitrous acid, HNO2 (aq) has a Ka value of 4.0 x 10-4. What is the pH and POH of a 0.0500 M solution…
A: The question is based on the concept of equilibrium. we have to determine PH and pOH of weak acid…
Q: The pH of an aqueous solution of 0.136 M sodium nitrite, NaNO2 (aq), is This solution is
A: NaNO2 is a salt of a weak acid (HNO2) and a strong base NaOH pH = 7 + (pKa + log C)/2
Q: If the equilibrium constant for A + B C is 0.175, then the equilibrium constant for 2C 2A + 2B is
A: Given data,K=0.175,for the reaction :A+B→C
Q: Assuming the base completely dissociates in water, what is the pH of a 5.20 g/L solution of Ba(OH)2…
A: pH is basically the negative logarithm of concentration of H+ and it is given by the formula:…
Q: A brand of window-cleaning fluid is composed of ammonia solution, water, a colouring agent, and a…
A:
Q: Calculate the pH, pOH, and fraction of solute protonated or deprotonated in the following aqueous…
A: Hello. Since your question has multiple sub-parts, we will solve the first three sub-parts for you.…
Q: Sodium acetate, NaCH3CO2, of mass 7.4 g is used to prepare 250 cm3 of aqueous solution. What is the…
A:
Q: The pH of body fluids can be assessed by the measurement of plasma pH, total CO2, and: РОН b. pkw с.…
A:
Q: The pH of an aqueous solution of acetic acid (CH,COOH) is 2.5229. What is the initial molar…
A:
Q: Would you expect an aqueous solution of manganese (VII) oxide to have a ph greater or less than 7.0?…
A: Oxidation state is the number of electrons that are either gain or lose by an atom in a chemical…
Calculate the pH of (a) 0.10 M NH4CI(aq), (b) 0.25 M NaCH3CO2(aq), (c) 0.200 M CH3COOH(aq).
(a)
0.10 M NH4Cl(aq)
The reaction is as follows,
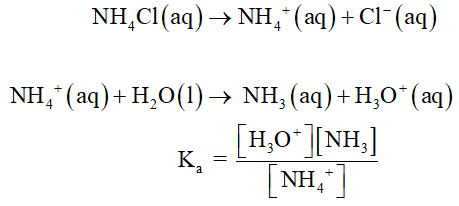
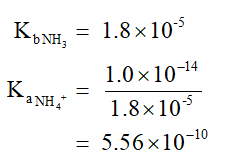
The pH value is calculated as shown below,
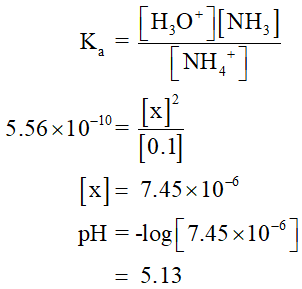
Step by step
Solved in 4 steps with 8 images

- What is the concentration of hydronium ions in (a) a solution that is 0.12 M HBRO(aq) and 0.160 M NaBrO(aq); (b) a solution that is 0.250 M CH 3 NH 2 ( aq ) and 0.150 M CH 3 NH 3 CI ( aq ); (c) a solution that is 0.160 M HBRO(aq) and 0.320 M NaBrO(aq); (d) a solution that is 0.250 M CH 3 NH 2 ( aq) and 0.250 M NaBr(aq)?Calculate the pH of (a) 0.050 M HCI(aq) and (b) 0.100 M KOH(aq).Calculate the molar concentration of H3O+ ions and the pH of the following solutions: (a) 25.0 cm3 of 0.144 M HCI(aq) was added to 25.0 cm3 of 0.125 M NaOH(aq), (b) 25.0 cm3 of 0.15 M HCI(aq) was added to 35.0 cm3 of 0.15 M KOH(aq), (c) 21.2 cm3 of 0.22 M HNO3(aq) was added to 10.0 cm3 of 0.30M NaOH(aq).
- (b) Calculate the pH of 0.1 mol dm-3 potassium hydroxide (KOH(aq)) pH =Calculate the pH, pOH, and fraction of solute protonated or deprotonated in the following aqueous solutions: (a) 0.120 M CH;CH(OH)COOH(aq) (lactic acid), (b) 1.4× 10“M CH;CH(OH) COOH(aq), (c) 0.15 M NH,CI(aq), (d) 0.15 m NaCH;CO,(aq), and (e) 0.112 m (CH;),N(aq) (trimethylamine).Calculate the molar solubility of Mg(OH)2 in 1.00 M NH4Cl (aq).
- Estimate the pH of 0.010 M NaCH3CO2(aq).Consider the titration of a 50.00 mL sample of 0.500 M hydrazoic acid, HN3, with 0.500 M NaOH (aq., 25 oC). The Ka HN3 = 4.50 x 10–4. What is the pH of the solution after 15.00 mL of NaOH has been added?Calculate the pH of a buffer containing 0.1 M HC2H3O2 (aq) mixed with 0.1 M NaC2H3O2 (aq)?
- Calculate the ph of a solution prepared by dissolving 1.00g of sodium acetate,CH^3COONa, in 50.0ml of 0.15 M acetate acid, CH^3COOH(aq)A buffer solution of volume 100 cm3 consists of 0.10 M CH3COOH(aq) and 0.10 MNaCH3CO2(aq). (a) What is its pH? (b) What is the pH after the addition of 3.3 mmol NaOH to the buffer solution? (c) What is the pH after the addition of 6.0 mmol HNO3 to the initial buffer solution?Estimate the pH and percentage of dissociation of 0.010 M CH3CH(OH)COOH(aq) (lactic acid). pKa of lactic acid is 3.86.

