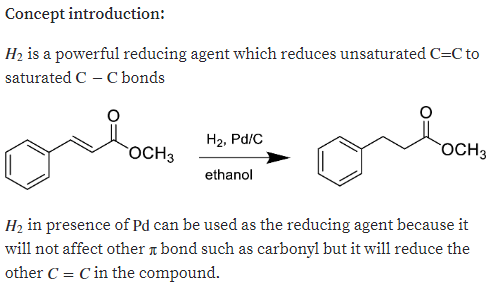
Q: Show the product expected when the following unsaturated 8-ketoester is treated with each reagent.…
A: NaBH4 is known as Sodium borohydride. It is a selective reducing agent. It is used for the reduction…
Q: 28. What reagent(s) will accomplish this conversion? (Et = ethyl) * EtMgBr followed by an acid wash…
A: 29.) First we would convert the Fischer projection to Haworth projection. Both the alcohol OH…
Q: The following molecule undergoes an intramolecular reaction in the presence of pyrrolidinium…
A:
Q: i. butanoic anhydride + sodium hydroxide j. butanoyl bromide + potassium ethoxide chs-CHz- CHz- ċ-Br…
A: When an acid or its derivative reacts with alcohol or its derivative, there is a formation of ester…
Q: 2. What reagents will accomplish the following multistep synthesis? A) HBr, CH;0OCH3 B) HBr C)…
A:
Q: OH H30* он
A: To show the mechanism of the following reaction
Q: Show the product expected when the following unsaturated 8-ketoester is treated with each reagent.…
A: The product of the given reaction can be shown as
Q: Propose a mechanism for the following reaction:
A: In the first step Π-bond abstracts the H+ ion from HCl to form a resonance stabilized C+…
Q: H. H+ + H20 HO.
A: In the presence of acid, nuceophilic attack of OH on carbonyl carbon takes place.
Q: Show the product from each step in the sequence of reactions from ethyl acetoacetic ester. il Br…
A: CH3CH2OK is a strong base which removes a proton from Alpha carbon of ethyl acetoacetic ester to…
Q: *Pu OK it 90 °C
A: An aldol condensation is a condensation reaction in which an enolate ion reacts with a carbonyl…
Q: UESTION 1 tify the reagents that you would use to perform the following reaction Br Br OH a. -b. OH…
A:
Q: Propose a mechanism (step by step) for the following transformation. Explain each step. HO. H,SO,…
A: Alkenes contain loosely bonded pi-electrons. In the presence of an acid, the alkenes are protonated…
Q: Question attached
A: The ylide is basically a neutral dipolar molecule in which the molecule contains a partially…
Q: 11. Propose a mechanism for the following reaction: OH H₂SO4 Heat
A: When an alkene reacts with an acid, it leads to the formation of a carbocation.
Q: 1. 2 CH,Li, ether 2. H,O, HCI
A:
Q: Show how m-toluidine can be converted tom-toluidineCH3 NH2the following compounds, using any…
A:
Q: Show how to convert each starting material into benzylamine in good yield
A: Given starting material,
Q: Upon treatment with Tollen's reagent, you would expect one of these compounds to be unreactive. OH…
A:
Q: What is the best choice of reagent(s) to perform the following transformation? ОН O H20, H2SO4 O…
A:
Q: Tat is the expe EtO O product for the following transformation? t-BuOK OEt A t-BuOH Br 1. NaOH, A 2.…
A: Note : DEM on reaction with t-BuOK produces compound A. Ester hydrolysis of compound A is done by…
Q: Q2-) Draw a detailed mechanism for the following Fischer esterification reaction.(13 points) A few…
A: When acetic acid and ethanol react in the presence of concentrated H2SO4 they give rise to a sweet…
Q: Show how m-toluidine can be converted tom-toluidineCH3 NH2the following compounds, using any…
A: a.
Q: 3. Draw a mechanism for the following: .心8.人發火 OH H3O Dilute HSO (b) OH (c) 4. If an alcohol is used…
A: Hello. Since multiple questions have been posted, the first question shall only be solved in this…
Q: `OH ZI
A:
Q: 7. Propose a mechanism for the following transformation. H* `NH2
A: In the mechanism of the above reaction ; 1st step involves the protonation 2nd step : attack on…
Q: Propose a mechanism for the following reaction: H. NaOH H20
A: Michael addition,followed by ring opening and then ring closing will give the product
Q: 1. Show the method to carry out each transformation: two-step method using a Grignard reagent. CH3…
A:
Q: la. Show the reaction needed to prepare propyl phenyl ketone from CH,CH,CN and a Grignard reagent.…
A: Grignard reaction: It is an organometallic chemical reaction in which alkyl or aryl magnesium…
Q: Propose a mechanism for the following transformation: to OH (H,SO4] MEOH Meo
A:
Q: Propose a mechanism for the following reaction:
A:
Q: Reaction of benzylmagnesium chloride with formaldehyde yields alcohols N and P after protonation.…
A: The Benzyl magnesium chloride undergoes electrophillic addition reaction with formaldehyde to form…
Q: 1. Identify the reagents necessary to make following transformation (show step by step reaction).…
A:
Q: What is the best choice of reagent(s) to perform the following transformation? O 03; followed by…
A: To solve this problem we have give the reagents which gives the desired product .
Q: 7. Propose a mechanism for the following transformation. EtO,C. CO,Et + NH3 OEt H.
A:
Q: (1] CH,CH,MgBr [2] H20
A:
Q: N2 PHCOOA9 heat Me
A: Given a reaction with reagent and reaction condition and we have to find out the product of the…
Q: Show the product expected when the following unsaturated 8-ketoester is treated with each reagent.…
A: The product expected when the given unsaturated δ-ketoester is treated with the given reagent has to…
Q: Draw the product of the following reaction: 1. NaCN 2. H₂O*. A 'Br Draw the most stable enol…
A:
Q: CI HCI + H2O
A: In this question we will write a mechanism see below. 1- In the first step carbocation intermediate…
Q: Why is a 6-membered acetal formed instead of a 5-membered 2,3- or 3,4- O-benzylidene acoetal? SROW…
A: The following mechanism shows the reason to support the formation of product in this reaction:
Q: 8. Provide a series of steps to accomplish the following transformation. CO₂Me Ph d
A:
Q: Propose a mechanism for the following reaction:
A:
Q: Show the product of treating y-butyrolactone with reagent.
A: The product of gamma-butyrolactone with the given reagent should be shown.
Q: 3.4 Propose a stepwise mechanism for the following reaction: CH,OH OCH,CH3 CH2OH OH CH2OH H30 H H H…
A: Detail mechanistic pathway is given below
Q: 10) Which of the following reagents with not allow conversion of acetone into bromoacetone? Answer…
A: The Answer to the following question is-
Q: What is the best choice of reagent(s) to perform the following transformation? ÓH a. H20, H2SO4 b.…
A:


Step by step
Solved in 2 steps with 2 images

- Draw the major product(s) when the following molecule is reacted with excess NaOEt in EtOH, followed by aqueous acidic work-up. CH;CH,OĊCH(CH,)¿COCH,CH3 CHaShow the product expected when the following unsaturated 8-ketoester is treated with each reagent. OEtHow would you prepare the following compound using a Robinson annulation reaction between a β-diketone and an α, β-unsaturated ketone? Draw the structures of both reactants and the intermediate Michael addition product.
- Draw the structures of the initially formed enol tautomers in the reactions of propyne and dicyclohexylethyne with dicyclohexylborane followed by NaOHNaOH and H2O2H2O2Show how the following compounds can be prepared from cyclohexanoneWhich reagent(s) would accomplish the synthesis showWn below? NH2 NH2 но. H3CO. A) NaBH4 В) РСС C) LAH D) Na2Cr207 / H2SO4 E) NAOH www w
- Draw the products formed when p-methylaniline (p-CH3C6H4NH2) istreated with following reagent. HClWhat product result from nitration of propylbenzene ?Step 2: A hemiacetal formation has a similar mechanism to the hydrate reaction, except 1 equivalent of alcohol is added instead of water. 1² R₂ R₁ LOR3 R₁ R₂ The hemiacetal reaction is also reversible and can also be catalyzed by either acid or base. Hemiacetals are not usually isolated, except in the formation of 5- and 6-membered rings, as often seen in carbohydrate chemistry. Identify the hemiacetal formed from the intramolecular cyclization of the molecule shown. OH H OH R3OH OH OH OH CH₂OH NaOH ?

