Q: O H3C H H CH3 + H3N NH3 `N and H CH3 H H H3C H O=U Z-I
A: The determination of configuration at every stereogenic centre in the given mole- ule is quite impor...
Q: H* (d) OH H* OH
A:
Q: Calculate the % composition of CuSO4.5H20 (Atomic masses:Cu = 63.55 amu; S = 32.06 amu; O = 16.00 am...
A:
Q: 1 Based on your observation, how can you identify the intermolecular forces of liquid on a given sub...
A: 1. Longer it takes to evaporate from surface of coin higher is the strength of intermolecular forces...
Q: As part of an experiment to determir measures the volume of a solid sample. Her four trials yield vo...
A:
Q: • Now pretend we have some amount of another ideal gas in a chamber with a sealed top which moves up...
A: Ideal gas equation PV = nRT. P:- pressure . V :- volume . n :- moles of gas R:- gas constant T:-...
Q: Determine the number and label the chiral centers in the molecule
A: Chiral carbon is asymmetric centre which is having four different groups surround the carbon atom.
Q: A grease fire started on the stove, it was then covered completely with a fire blanket so that the f...
A: We need to determine limiting reactant in combustion reaction.
Q: 9) drug shown below: Select the correct number of chirality centers in heroin, the illicit A) 4 B) 5...
A:
Q: How many liters are present in 179.0 g of Krypton (Kr)? V- M. a. 1,536 Liters b. 0.9313 Liters С. 47...
A:
Q: What mass of cobalt III hydroxide, Co(OH)3 is needed to make 5.2 liters of a 0.21 M cobalt III hydro...
A:
Q: Calculate the pH for a 0.1 M solution of C6H5COOH (K, = 6.46 x 105) and the pH for a 0.2 M %3D solut...
A:
Q: Question 4 (4 points) An aqueous solution of KF has a freezing point of -2.48 °C. Assuming total dis...
A:
Q: From the given starting material provide a reasonable synthetic route to the following plecule. O,N.
A: Here we have to prepare the given compound from the starting material 2-methylbuta-1,3-diene in mult...
Q: 11. The heat of solution ( Δ Hsoln) is the algebraic sum of the heat of mixing (Δ Hmix), the heat of...
A:
Q: 21. What is the order of a reaction where each successive half life gets longer and is dependent upo...
A: The half-life of second-order of a reaction: t1/2 = 1K[A]∘ In the second-order reaction, the ha...
Q: Provide IUPAC name for the following compounds.
A:
Q: I I Least reactive Most reactive
A: SN1 reaction is substitution nucleophilic unimolecular reaction. Stability of reaction depend upon ...
Q: e formulas for
A:
Q: 2NaOH + H2SO4 ® Na2SO4 + 2H2O What is the mole ratio of sodium hydroxide to water?
A:
Q: 1. For the reaction 2NO(g) + 02(g) 2NO,(g) rate = k[NO] [02] %3D Is the following proposed mechanism...
A: Here we have to verify whether the proposed mechanism of reaction is plausible for the given reactio...
Q: A mixture of 0.4410 M CÓ and 0.3650 M Cl, is enclosed in a vessel and heated to 1000 K. CO(g) + Cl, ...
A: CO (g) + Cl2 (g) ⇔ COCl2 (g)at T =0 0.4410 M ...
Q: CH3 H3C H- and H3C CH3 H H
A: Enantiomers are the stereo isomers having non superimposable mirror image relationship and the diast...
Q: Question 9 of 9 How much water (in dL) would you need to add to 140 dL of a 9.4 g/dL solution of Mg(...
A: This is an example of dilution law. Here We are required to find the volume of water added to dilute...
Q: You are treating the TLC plate with 5% of sulfuric acid in ethanol. Briefly describe how this works.
A: Given that : We have to describe how treating the TLC plate with 5% of sulfuric acid in ethanol work...
Q: What is the correct procedure in diluting a concentrated acid solution? A add the acid to the water ...
A: We have to tell the procedure of diluting a concentrated acid solution. The given options are Add t...
Q: Calculate the mass of Silver (Ag) in Silver Chloride (AgCl) when 8.26 g Magnesium Chloride (MgCl2) r...
A:
Q: grams
A:
Q: Draw the structure(s) of the product(s) you would expect when tetrahydrofuran is cleaved with HI.
A: The presence of lone pair on the oxygen atom in the THF molecule is the key factor to progress the m...
Q: If the osmotic pressure of a 8.25×10-2-M aqueous solution of CaCl2 was found to be 5.41 atm at 20°C,...
A:
Q: a ease and wavelength for all transitions indicated using the following equation Also correction the...
A: Given values as,
Q: What mass of HCI, in grams, is required to react with 0.860 g of Al(OH)3? Al(OH)3(s) + 3HCI(aq) → AI...
A: The balanced equation is, AlOH3s+3HClaq→AlCl3aq+3H2Ol Mass of Al(OH)3=0.860 g Moles of Al(OH)3 is, m...
Q: A 0.312-kg sample of methylene chloride has a density of 1.326 g/cm°. Calculate its volume in cubic
A: • The values provided in the question are:- i) Mass of methylene chloride, m = 0.312 kg = 0.312 ...
Q: Hello, I need help taking all the information and turning it into a balanced molecular equation. Tha...
A:
Q: A car bumper is plated with chromium using chromium (III) ions in solution. If a current of 54A flow...
A:
Q: CH2OH CH,OH HO and но - OH он OH но OH он
A: If configuration of substituents change only at one position all other have same configuration then ...
Q: Which of the following compounds contain primary (1°) radical carbons? II III IV A) Only I B) Only I...
A:
Q: Data for the decomposition of nitrosyl bromide: 2NOBr@)- → 2NO@) + Br2) is given below: Time (s) 0 [...
A: A question based on kinetics of reaction in solution that is to be accomplished.
Q: Cyanogen has the formula C2N2. Propose a bonding scheme that gives each atom the correct number of c...
A: The determination of the hybridization of central carbon atom in the given molecule is quite necessa...
Q: Which of the following statements concerning the limiting reactant is/are correct? The mass of the l...
A:
Q: . What is the normal boiling point of a 3.75 m aqueous glucose solution? Kb = 0.51 oC/m Group of ans...
A:
Q: 4 van der Waals interactions • List and briefly explain three contributions to total van der Waals i...
A: Answer would be
Q: The density of mercury is 13.35 g/cm3. Experimental results gave the following data: 10 4.5 g/cm3 16...
A: Given, True value of density = 13.35 g/cm3 Experimental results are : 14.5 g/cm3, 16.65 g/cm3, 15...
Q: Provide the correct IUPAC name for S2Cl8
A:
Q: Submit Barium hydride reacts with water to produce barium hydroxide and hydrogen gas. BaH2 (s) + 2 H...
A: Barium hydride reacts with water to form barium hydroxide and water. The equation for the balanced c...
Q: - molar heat of combustioN
A:
Q: Create a graphic organizer comparing ionic, non-polar covalent, network covalent, polar covalent, an...
A: Answer is below
Q: The complete combustion of methane, CH4, can be represented by the equation: CH4 (g) + 2 O2 (g) --->...
A: At STP molar volume of an ideal gas is 22.4 L.
Q: Explain why this reaction wouldnt work and what product would actually form? What additional rea...
A: The direct conversion of a acid to AN amide is troublesome as a result of amines are basic and have ...
Q: description of typical properties and how you could recognize the bonding in a substance from those ...
A: The force of attraction which holds two atoms, ions together is known as chemical bonds. Atoms combi...
What is the net ionic equation for the following reaction? NazCO3(aq) + FeCI2(aq) --> FeCO3(s) + 2NaCl(aq)
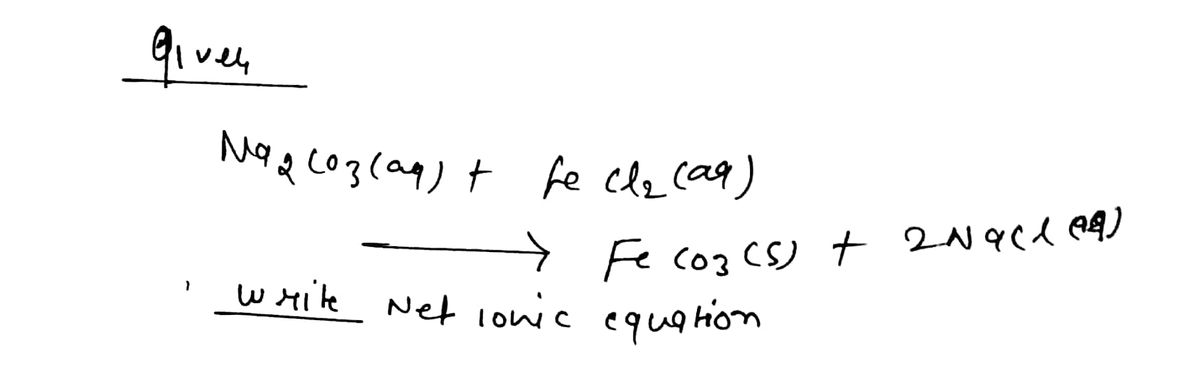
Step by step
Solved in 2 steps with 2 images

- A phosphate buffer contains KH2PO4 and K2HPO4. What is the net ionic equation for the buffering reaction that occurs when some HCl is added to the buffer solution? (A) H2PO4–(aq) + Cl–(aq) --> HPO42–(aq) + HCl(aq); (B) H2PO4–(aq) + H3O+(aq) --> H3PO4(aq) + H2O(aq); (C) HPO42–(aq) + Cl–(aq) --> PO43–(aq) + HCl(aq); (D) HPO42–(aq) + H3O+(aq) --> H2PO4–(aq) + H2O(aq);Limestone consists mainly of the mineral calcite, CaCO3. The carbonate content of 0.5143 g of powdered limestone was measured by suspending the powder in water, adding 10.00 mL of 1.396 M HCl, and heating to dissolve the solid and expel the CO2: CaCO3(s) + 2H+ ⟶⟶ Ca2+ + CO2↑↑ + H2O The excess acid requied 39.96 mL of 0.1004 M NaOH for complete titration: OH- + H+ ⟶⟶ H2O Find the weight percent of calcite in the limestone.You prepare a standard NaOH solution for the laboratory, using potassium hydrogen phthalate (KHC₈H₄O₄, abbreviated KHP) as the primary standard. KHP (molar mass = 204.22 g/mol) has one acidic hydrogen. It took 31.55 mL of the NaOH solution to titrate (react exactly with) 0.750 g KHP. You then use the standard base solution to determine the amount of ascorbic acid in a 500.0 mg tablet of vitamin C. Ascorbic acid (HC₆H₇O₆, molar mass 176.12 g/mol) also has one acidic hydrogen, and is usually mixed with some filler in preparing the tablets. It requires 17.54 mL of the NaOH to titrate (react exactly with) the ascorbic acid found in one 500.0 mg tablet. What is the percent vitamin C by mass in the tablet?
- 43. A buffer is prepared using acetic acid, CH3COOH, (a weak acid, pKa = 4.75) and sodium acetate, CH3COONa (which provides acetate ions, the conjugate base), according to the following proportions: Volume of CH3COOH(aq): 125.0 mL Concentration of CH3COOH(aq): 1.263 M Volume of CH3COONa(aq): 106.0 mL Concentration of CH3COONa(aq): 1.279 M Determine the volume of 6.00 M HCl required to change the pH of this buffer by one pH unit. 18.15 mL 36.30 mL 21.87 mL 19.30 mL 43.73 mL42. A buffer is prepared using acetic acid, CH3COOH, (a weak acid, pKa = 4.75) and sodium acetate, CH3COONa (which provides acetate ions, the conjugate base), according to the following proportions: Volume of CH3COOH(aq): 139.0 mL Concentration of CH3COOH(aq): 1.103 M Volume of CH3COONa(aq): 127.0 mL Concentration of CH3COONa(aq): 1.259 M Calculate the pH of this buffer after the addition of 20.00 mL of 3.00 M sodium hydroxide, NaOH, a strong base. 4.38 5.90 4.69 4.81 5.12What is the equilibrium expression for the following reaction? Zn(s) + 2NAOH(aq) + 2H2O(e) = NazZn(OH)4(aq) + H2(g)
- A 0.9600 M NaOH solution was diluted with a factor of 4. Then the diluted solution of NaOH was titrated against an unknown acid. The volumes of NaOH(aq) titrated are shown in the table below. Volumes of NaOH(aq): Rough titration First Second Third Third titration titration titration titration Final burette reading: 19.00 34.05 20.86 36.01 23.57 (mL) Initial burette reading: (mL) 19.00 19.07 5.76 21.02 8.54 14 -98 is.ld 14.99 15.0g The molarity of the unknown acid is 0.08108 M and the volume of the unknown acid used in each titration is 14.80 mL. (1) Was the unknown acid monoprotic, diprotic or triprotic? Show your calculations and reasoning.7.7 g of citric acid (MM = 192.1 g/mol) can be titrated with NaOH according to the following balanced chemical equation. H3C6H5O7 (aq) + NaOH(aq) → H2O(l) + Na H2C6H5O7(aq) what would the curve for the titration of the neutralization of the three hydrogens of this acid with NaOH look like?Which of the following is the correct net ionic equation for this reaction? H3PO4(aq) + 3NaOH(aq) --> Na3PO4(aq) + 3H2O(l) (A) H+(aq) + OH–(aq) --> H2O(l) (B) 3Na+(aq) + PO43–(aq) --> Na3PO4(aq) (C) PO43–(aq) + 3NaOH(aq) --> Na3PO4(aq) + 3OH–(aq); (D) H3PO4(aq) + 3OH–(aq) --> 3H2O(l) + PO43–(aq)
- 24. A buffer is prepared using acetic acid, CH3COOH, (a weak acid) and sodium acetate, CH3COONa (which provides acetate ions, the conjugate base), according to the following proportions: Volume of CH3COOH(aq): 135.0 mL Concentration of CH3COOH(aq): 1.014 M Volume of CH3COONa(aq): 123.0 mL Concentration of CH3COONa(aq): 1.204 M What is the concentration of the conjugate base, CH3COO-, component in the final buffer solution? Give your answer in M, with correct significant digits.Given the equation: Ag+(aq)+2NH3(aq)⟶[Ag(NH3)2]+(aq) ?f=2.00×10^7 determine the concentration of NH3(aq) that is required to dissolve 295 mg of AgCl(s) in 100.0 mL of solution. The ?sp of AgCl is 1.77×10^−10. I've worked this through several times, but I am just not coming up with the right answer! Thanks :)Consider the following reactions: Reaction 1: HCIO, + KOH → KC1O, + H;O Reaction 2: HC;H;O2 + KOH → KC;H;O2+ H;O In both reactions, 15.00 mL of 0.8116 M acid is titrated with 0.4217 M KOH. A volume of 28.87 mL of KOH is required to reach the equivalence point in both reactions. a) What is the pH of the HC1O, solution before the titration has started? (No KOH has been added.) b) What is the initial pH of HC;H;O2 solution before the titration has started? K= 6.46 x 10-5 (Ok to use x is small.) c) What is the pH of Reaction 1 at the equivalence point? d) What is the pH of Reaction 2 at the equivalence point? e) What is the pH of Reaction 1 after 32.00 mL of KOH has been added? f) What is the pH of Reaction 2 after 18.00 mL of KOH has been added?

