
Organic Chemistry - Standalone book
10th Edition
ISBN: 9780073511214
Author: Francis A Carey Dr., Robert M. Giuliano
Publisher: McGraw-Hill Education
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12.18, Problem 20P
Give an explanation for each of the following observations:
Compound A has six
electrons but is not
Compound B has six
Compound C has
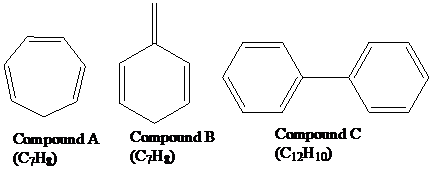
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
Cyclopropenones are described as having aromatic character. How would you account for this, given that the ring contains three ℼ-electron
Cyclopropenones are described as having aromatic character. How would you account for this, given that the ring contains three π-electrons?
Compounds A and B are isomers having the molecular formula C4H8O3. Identify A and B on the basis of their 1H NMR spectra.Compound A: δ 1.3 (3H, triplet); 3.6 (2H, quartet); 4.1 (2H, singlet); 11.1 (1H, broad singlet)Compound B: δ 2.6 (2H, triplet); 3.4 (3H, singlet); 3.7 (2H triplet); 11.3 (1H, broad singlet)
Chapter 12 Solutions
Organic Chemistry - Standalone book
Ch. 12.2 - Write structural formulas for toluene (C6H5CH3)...Ch. 12.3 - Prob. 2PCh. 12.5 - Prob. 3PCh. 12.5 - Prob. 4PCh. 12.6 - Prob. 5PCh. 12.6 - Chrysene is an aromatic hydrocarbon found in coal...Ch. 12.8 - Prob. 7PCh. 12.9 - As measured by their first-order rate constants,...Ch. 12.9 - Give the structure of the principal organic...Ch. 12.9 - Prob. 10P
Ch. 12.10 - Prob. 11PCh. 12.11 - Prob. 12PCh. 12.12 - Prob. 13PCh. 12.13 - Prob. 14PCh. 12.13 - Prob. 15PCh. 12.15 - The regioselectivity of Birch reduction of...Ch. 12.16 - Prob. 17PCh. 12.17 - Both cyclooctatetraene and styrene have the...Ch. 12.17 - Prob. 19PCh. 12.18 - Give an explanation for each of the following...Ch. 12.19 - Prob. 21PCh. 12.19 - What does a comparison of the heats of combustion...Ch. 12.20 - Prob. 23PCh. 12.20 - Prob. 24PCh. 12.20 - Prob. 25PCh. 12.20 - Prob. 26PCh. 12.20 - Prob. 27PCh. 12.20 - Prob. 28PCh. 12.21 - Prob. 29PCh. 12.21 - Prob. 30PCh. 12.22 - Prob. 31PCh. 12.22 - Prob. 32PCh. 12 - Write structural formulas and give the IUPAC names...Ch. 12 - Prob. 34PCh. 12 - Prob. 35PCh. 12 - Prob. 36PCh. 12 - Prob. 37PCh. 12 - Acridine is a heterocyclic aromatic compound...Ch. 12 - Prob. 39PCh. 12 - Prob. 40PCh. 12 - Prob. 41PCh. 12 - Evaluate each of the following processes applied...Ch. 12 - Prob. 43PCh. 12 - Prob. 44PCh. 12 - Prob. 45PCh. 12 - Prob. 46PCh. 12 - Anthracene undergoes a DielsAlder reaction with...Ch. 12 - Prob. 48PCh. 12 - Prob. 49PCh. 12 - The relative rates of reaction of ethane, toluene,...Ch. 12 - Both 1,2-dihydronaphthalene and...Ch. 12 - Prob. 52PCh. 12 - Prob. 53PCh. 12 - Prob. 54PCh. 12 - Prob. 55PCh. 12 - Prob. 56PCh. 12 - Each of the following reactions has been described...Ch. 12 - Prob. 58PCh. 12 - A compound was obtained from a natural product and...Ch. 12 - Prob. 60PCh. 12 - Suggest reagents suitable for carrying out each of...Ch. 12 - Prob. 62PCh. 12 - Prob. 63DSPCh. 12 - Prob. 64DSPCh. 12 - Prob. 65DSPCh. 12 - Prob. 66DSP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Select the most accurate statement about the following compound. The compound is aromatic because there are 4n+2 pi electrons. O The compound is aromatic because it is completely conjugated throughout the entire ring. O The compound is non-aromatic (not aromatic) because it is not planar. O The compound is anti-aromatic because there are 4n pi electrons.arrow_forwardBased on the structures given below, which of the following statements is entirely true? Quinoline NH Isoquinoline Indole Isoindole All four compounds are non-aromatic, with 12 pi-electrons each and are equal in basicity. Only quinoline and Isoquinoline having 10 pi-electrons each are aromatic while indole and isoindole are antiaromatic having 8 pi-electrons each The nitrogen atoms of quinoline and isoquinoline are sp2 hybridized while the nitrogen atoms of indole and isoindole are sp³ hybridized. All four compounds are aromatic, with 10 pi-electrons each and are equal in basicity. All four compounds are aromatic, with 10 pi-electrons each, and with quinoline and isoquinoline being stronger bases than indole and isoindarrow_forwardBased on the structures given below, which of the following statements is entirely true? N Quinoline NH Isoquinoline Indole Isoindole All four compounds are non-aromatic, with 12 pi-electrons each and are equal in basicity. All four compounds are aromatic, with 10 pi-electrons each and are equal in basicity. Only quinoline and isoquinoline having 10 pi-electrons each are aromatic while indole and isoindole are antiaromatic having 8 pi-electrons each. All four compounds are aromatic, with 10 pi-electrons each, and with quinoline and isoquinoline being stronger bases than indole and isoindole. The nitrogen atoms of quinoline and isoquinoline are sp2 hybridized while the nitrogen atoms of indole and isoindole are sp³ hybridized Previousarrow_forward
- Compound A of molecular formula C3H6O shows a noteworthy infrared absorption at 1716 cm-1. Its 1H-NMR spectrum shows one singlet – δ 2.2 (6H) ppm. Its 13C-NMR spectrum has two signals – δ 30, 207 ppm. Suggest a structure for this compound.arrow_forwardCompound A has molecular formula C5H10O. It shows three signals in the 1H-NMR spectrum - a doublet of integral 6 at 1.1 ppm, a singlet of integral 3 at 2.14 ppm, and a quintet of integral 1 at 2.58 ppm. Suggest a structure for A and explain your reasoning.arrow_forwardCompound P has molecular formula C5H9ClO2. Deduce the structure of P from its 1H and 13C NMR spectra.arrow_forward
- Is the following compound aromatic? Explain the reasons for your answer?arrow_forwardOne of the four criteria of aromaticity by Hückel is planarity of the rings containing 4n+2 electrons. Based on the Hückel rules, can a compound be aromatic but not planar? Please explain your answer.arrow_forwardCompound A, C8H10O, has the IR and 1H NMR spectra shown. Propose a structure consistent with the observed spectra, and label each peak in the NMR spectrum. Note that the absorption at 5.5 î disappears when D2O is added.arrow_forward
- Some of the following compounds show aromatic properties, and others do not predict which ones are likely to be aromatic and explain why they are aromatic or not.arrow_forwardA chemist isolated an aromatic compound with molecular formula C6H4Br2. He treated this compound with nitric acid and sulfuric acid and isolated threedifferent isomers, in different amounts, with molecular formula C6H3Br2NO2. What was the structure of the original compound?arrow_forwardCompound X (molecular formula C10H12O) was treated with NH2NH2,−OH to yield compound Y (molecular formula C10H14). Based on the 1H NMR spectra of X and Y given below, what are the structures of X and Y?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
NMR Spectroscopy; Author: Professor Dave Explains;https://www.youtube.com/watch?v=SBir5wUS3Bo;License: Standard YouTube License, CC-BY