
Concept explainers
Practice Problem G.1
When farnesol is treated with sulfuric acid, it is converted to bisabolene. Outline a possible mechanism for this reaction.
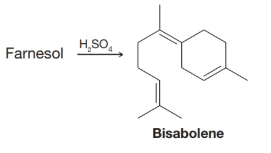
Interpretation:
The mechanism for the formation of Bisabolene from Farnesol in presence of 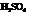 is to be outlined.
is to be outlined.
Concept introduction:
Farnesol is a compound that is formed from isoprene compounds. In general, when geranyl pyrophosphate reacts with isopenenyl pyrophosphate, farnesyl pyrophosphate (intermediate for the synthesis of sesquiterpenes) is formed. This, on oxidation can give farnesol.
Concentrated sulfuric acid acts as a strong dehydrating agent and removes the water molecule from alcohols to form unsaturated hydrocarbon.
Answer to Problem 1PP
Solution:
The mechanism for the synthesis of Bisabolene is explained as follows:

Explanation of Solution
During the first step, Farnesol (unsaturated alcohol) reacts with acid (HA = 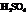 ) to form a hydrated product. Then, the hydrated product formed loses one water molecule to form a carbocation. Consequently, this carbocation rearranges to form a more stable carbocation. At last, the base
) to form a hydrated product. Then, the hydrated product formed loses one water molecule to form a carbocation. Consequently, this carbocation rearranges to form a more stable carbocation. At last, the base 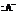 (
( 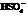 ) abstracts the hydrogen ion from the molecule, forming Bisabolene.
) abstracts the hydrogen ion from the molecule, forming Bisabolene.
The whole mechanism can be depicted as:

Hence, Bisabolene has been formed from Farnesol in the presence of concentrated sulfuric acid.
The Bisabolene has been synthesized from Farnesol in the presence of concentrated sulfuric acid. The mechanism for the synthesis of Bisabolene is outlined as:

Want to see more full solutions like this?
Chapter G Solutions
Organic Chemistry
Additional Science Textbook Solutions
Essential Organic Chemistry (3rd Edition)
CHEMISTRY-TEXT
Chemistry: Structure and Properties (2nd Edition)
The Organic Chem Lab Survival Manual: A Student's Guide to Techniques
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
Chemistry: A Molecular Approach
- don't provide answer in image draw a mechanism for this reactionarrow_forwardPROBLEM Predict the product(s) and propose a mechanism for each of the following reactions: 19-31 (a) (b) + HO H+ catalyst + OH HO OH H+ catalystarrow_forwardOChem Question: Starting with propyne and using any other required reagents, show how you would synthesize (2R, 3R)- and (2S, 3S)-2,3-dibromobutane (as a racemic form)arrow_forward
- Problum 13.22 Linalool and lavandulol are two of the major compo- nents of lavender oil. (a) What organolithium reagent and carbonyl compound can be used to make each alcohol? (b) How might lavandu- lol be formed by reduction of a carbonyl compound? (c) Why can't linalool be prepared by a similar pathway? OH linalool (three methods) Page 622 OH yo lavandulolarrow_forwardPROBLEM F.9 Draw the molecules that correspond to the following IUPAC names. (a) Pentyl pentanoate; (b) propyl butanoate; (c) ethyl methanoate; (d) ethyl (Z)-3-methylpent-2-enoate; (e) (R)-3-chlorobutyl (S)-2-hydroxypropanoatearrow_forwardProvide mechanism:arrow_forward
- Mechanism preparation of Aniline from nitrobenzenearrow_forwardDraw a curved arrow mechanism for the following acid-catalyzed isomerization reaction. Draw three intermediate structures and provide curved arrows. Use the spaces provided.arrow_forwardProvide the product(s) for the following reactions. Be sure to indicatestereochemistry where appropriate. No mechanismarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
