
Concept explainers
Assign formal charges to each carbon atom in the given species. All lone pairs have been drawn in.
a.
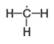 d.
d. 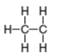
(a)
Interpretation: The formal charge on each carbon atom in the given species is to be assigned.
Concept introduction: The formal charge on an atom is calculated by the formula,
Answer to Problem 1.39P
In the given species, the formal charge on carbon atom of
Explanation of Solution
The given species is
The formal charge on an atom is calculated by the formula,
Substitute these values in the above equation to calculate the formal charge.
For the carbon present in
Substitute these values in above equation to calculate the formal charge on the carbon of
Thus, in the given species, the formal charge on carbon atom of
(a) In the given species, the formal charge on carbon atom of
(b)
Interpretation: The formal charge on each carbon atom in the given species is to be assigned.
Concept introduction: The formal charge on an atom is calculated by the formula,
Answer to Problem 1.39P
In the given species, the formal charge on carbon atom is zero as shown below.
Explanation of Solution
The given species is
The formal charge on an atom is calculated by the formula,
Substitute these values in above equation to calculate the formal charge on carbon atom.
Thus, in the given species, the formal charge on carbon atom is zero.
In the given species, the formal charge on carbon atom is zero.
(c)
Interpretation: The formal charge on each carbon atom in the given species is to be assigned.
Concept introduction: The formal charge on an atom is calculated by the formula,
Answer to Problem 1.39P
In the given species, the formal charge on carbon atom is zero as shown below.
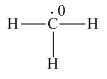
Explanation of Solution
The given species is,
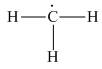
Figure 1
The formal charge on an atom is calculated by the formula,
For the given carbon atom,
Substitute these values in above equation, to calculate the formal charge on carbon atom.
Thus, in the given species, the formal charge on carbon atom is zero.

Figure 2
In the given species, the formal charge on carbon atom is zero.
(d)
Interpretation: The formal charge on each carbon atom in the given species is to be assigned.
Concept introduction: The formal charge on an atom is calculated by the formula,
Answer to Problem 1.39P
In the given species, the formal charge on carbon atom of
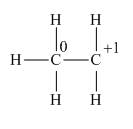
Explanation of Solution
The given species is,
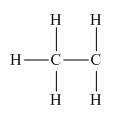
Figure 3
The formal charge on an atom is calculated by the formula,
Substitute these value in above equation to calculate the formal charge on the carbon of
For the carbon of
Substitute these value in above equation to calculate the formal charge on the carbon of
Thus, in the given species, the formal charge on carbon atom of

Figure 4
In the given species, the formal charge on carbon atom of
Want to see more full solutions like this?
Chapter 1 Solutions
Organic Chemistry
Additional Science Textbook Solutions
Chemistry: The Molecular Nature of Matter
Organic Chemistry (9th Edition)
Organic Chemistry
General, Organic, and Biological Chemistry (3rd Edition)
Principles of General, Organic, Biological Chemistry
General, Organic, and Biological Chemistry: Structures of Life (5th Edition)
- b. There is one additional resonance structure. NH₂ c. There is a total of five resonance structures (including the original structure). : OHarrow_forwardents V Soul (TV series) O REPRESENTATIONS OF ORGANIC MOLECULES Drawing a skeletal structure from a Lewis structure Convert the Lewis structure below into a skeletal structure. Н—С— Н H. H- H- Н—С—Н H. HH H. H- Н—С—С—С—С—С—С—С- Н C=C H- H. H. H. Н—С— Н H. ct+ Click and drag to start drawing a structure. HIC CIH HICarrow_forwardDraw Lewis structures for tye following molecules. a.) Atom "X" has 4 valence electrons and Atom "Y" has 6 valence electrons. Draw the Lewis structure for the molecule XY2, showing all lone pairs and formal charges when necessary b.) Atom "M" has 5 valence electrons and Atom "L" has 7 valence electrons. Draw the Lewis structure for the molecule M2L4, showing all lone pairs and formal charges when necessaryarrow_forward
- The Structure of Hydrocarbons 1. Draw the four carbon bonding patterns commonly seen in hydrocarbons and all other organic compounds. Construct the Lewis structures of the following compounds by combining the carbon atoms with the appropriate bonding patterns and the required number of hydrogen atoms. 2. a. hexane: CH,CH,CH,CH,CH,CH, b. 3-hexene: CH,CH,CH=CHCH,CH, c. 2-hexyne: CH,C=CCH,CH,CH, 3. There are three distinct bond angles formed in the four bonding patterns of carbon. What are they and in which bonding pattern(s) is each seen?arrow_forward4. The following are valid Lewis structures for CH3SOCH3. Label the formal charges and circle the best Lewis structure. H :ö: H H :0 H a) H-C=S-C-H b) Н—С—S— С-н H. H. H. H. Н :0: Н H :ö: H d) H-C=$-C-H c) Н—С—$—С-н H. H. H Harrow_forwardDraw the Lewis structure for each compound.a. Cl2O7 (no Cl¬Cl bond)b. H3PO3 (two OH bonds)c. H3AsO4arrow_forward
- Choose the Lewis structure of NH4. H-N-H H. H-N-H H-N=Harrow_forward1. What is the relationship between the following compounds? H :0: H H:0: H H-C=C-C-C-C-H H-C-C-C=Ċ-H H HH H. H-C-H B. constitutional isomers C. the same structure A. isotopes D. composed of different elements E. no relationship 2. What is the correct Lewis structure for HN3, including the formal charges? CIHarrow_forwardAssign formal charges to each carbon atom in the given species. All lone pairs have been drawn in. нн a. CH2=CH b. H-с-н с. Н-с-н d. H-С-С H H e. -N: f. g. h. :N=N=N: -N=O:arrow_forward
- 5. Draw Lewis structures for each molecular formula. H A. C₂H4Cl₂ (two isomers) B. C3H8O (three isomers) C. C6H14 (five isomers) C-H-C-H-CI-CI C-H-C-H-CI-CIarrow_forward1. Complete the Lewis structure by adding double bonds, triple bonds, and/or lone pairs. There are no formal charges on any atom in this structure. H. H H. H エーOarrow_forwardWrite Lewis structures for each molecule or ion. Include reso- nance structures if necessary and assign formal charges to all atoms. If necessary, expand the octet on the central atom to lower formal charge. a. SO,2- b. HSO, c. SO3 d. BrOzarrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning

