
(a)
Interpretation:
The reaction,
Concept introduction:
A redox reaction is a type of reaction in which one reacting species gets oxidize and other reacting species is reduced. The exchange of electron takes place between the reacting species. Water molecules and protons can be used to
Answer to Problem 10.26P
The reaction,
Explanation of Solution
The given reaction is shown below.
The reactant and products with an oxidation number of atoms are shown below.
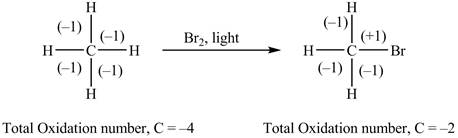
Figure 1
The oxidation number of reactant is lower than the oxidation number of the product. Therefore, the reaction is an oxidation reaction.
The number of electrons transfer in the reaction is given by the expression as shown below.
Where,
•
•
Substitute the values of
Therefore, the number of electrons transferred in the reaction is
The number of electrons transferred in the oxidation reaction is
(b)
Interpretation:
The given reaction is to be classified as an oxidation, a reduction, or neither. The number of electrons transferred in the reaction is to be stated when the corresponding reaction is oxidation or reduction.
Concept introduction:
A redox reaction is a type of reaction in which one reacting species gets oxidize and other reacting species is reduced. The exchange of electron takes place between the reacting species. Water molecules and protons can be used to balance a redox
Answer to Problem 10.26P
The given reaction is classified as oxidation reaction. The number of electrons transferred in the reaction is
Explanation of Solution
The given reaction is shown below.
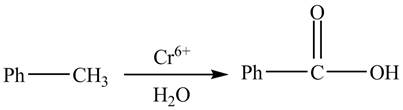
Figure 2
The reactant and products with oxidation number of atoms are shown below.
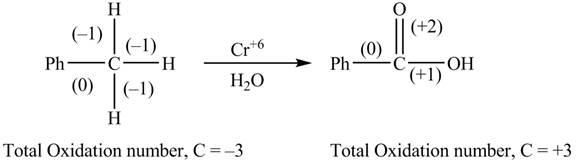
Figure 3
The oxidation number of reactant is lower than the oxidation number of the product. Therefore, the reaction is an oxidation reaction.
The number of electrons transfer in the reaction is given by the expression as shown below.
Where,
•
•
Substitute the values of
Therefore, the number of electrons transferred in the reaction is
The number of electrons transferred in the oxidation reaction is
(c)
Interpretation:
The reaction,
Concept introduction:
A redox reaction is a type of reaction in which one reacting species gets oxidize and other reacting species is reduced. The exchange of electron takes place between the reacting species. Water molecules and protons can be used to balance a redox chemical reaction in the acidic medium.
Answer to Problem 10.26P
The reaction,
Explanation of Solution
The given reaction is shown below.
The reactant and products with oxidation number of atoms are shown below.
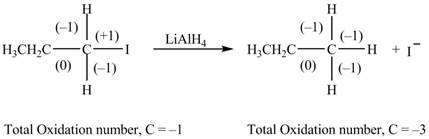
Figure 4
The oxidation number of reactant is higher than the oxidation number of the product. Therefore, the reaction is the reduction reaction.
The number of electrons transfer in the reaction is given by the expression as shown below.
Where,
•
•
Substitute the values of
Therefore, the number of electrons transferred in the reaction is
The number of electrons transferred in the reduction reaction is
(d)
Interpretation:
The given reaction is to be classified as an oxidation, a reduction, or neither. The number of electrons transferred in the reaction is to be stated when the corresponding reaction is oxidation or reduction.
Concept introduction:
A redox reaction is a type of reaction in which one reacting species gets oxidize and other reacting species is reduced. The exchange of electron takes place between the reacting species. Water molecules and protons can be used to balance a redox chemical reaction in the acidic medium.
Answer to Problem 10.26P
The given reaction is to be classified as oxidation reaction. The number of electrons transferred in the reaction is
Explanation of Solution
The given reaction is shown below.
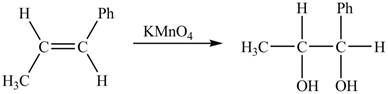
Figure 5
The reactant and products with oxidation number of atoms are shown below.
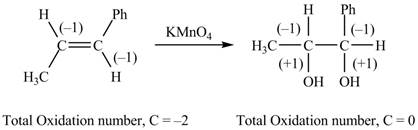
Figure 6
The oxidation number of reactant is lower than the oxidation number of the product. Therefore, the reaction is an oxidation reaction.
The number of electrons transfer in the reaction is given by the expression as shown below.
Where,
•
•
Substitute the values of
Therefore, the number of electrons transferred in the reaction is
The number of electrons transferred in the oxidation reaction is
(e)
Interpretation:
The given reaction is to be classified as an oxidation, a reduction, or neither. The number of electrons transferred in the reaction is to be stated when the corresponding reaction is oxidation or reduction.
Concept introduction:
A redox reaction is a type of reaction in which one reacting species gets oxidize and other reacting species is reduced. The exchange of electron takes place between the reacting species. Water molecules and protons can be used to balance a redox chemical reaction in the acidic medium.
Answer to Problem 10.26P
The given reaction is to be classified as oxidation reaction. The number of electrons transferred in the reaction is
Explanation of Solution
The given reaction is shown below.
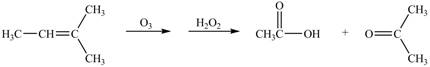
Figure 7
The reactant and products with oxidation number of atoms are shown below.
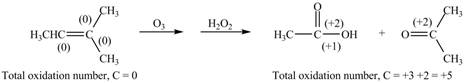
Figure 8
The oxidation number of reactant is lower than the oxidation number of product. Therefore, the reaction is oxidation reaction.
The number of electrons transfer in the reaction is given by the expression as shown below.
Where,
•
•
Substitute the values of
Therefore, the number of electrons transferred in the reaction is
The number of electrons transferred in the oxidation reaction is
(f)
Interpretation:
The given reaction is to be classified as an oxidation, a reduction, or neither. The number of electrons transferred in the reaction is to be stated when the corresponding reaction is oxidation or reduction.
Concept introduction:
A redox reaction is a type of reaction in which one reacting species gets oxidize and other reacting species is reduced. The exchange of electron takes place between the reacting species. Water molecules and protons can be used to balance a redox chemical reaction in the acidic medium.
Answer to Problem 10.26P
The given reaction is to be classified as neither
Explanation of Solution
The given reaction is shown below.
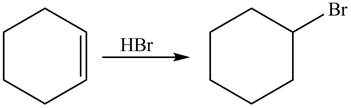
Figure 9
The reactant and products with an oxidation number of atoms are shown below.
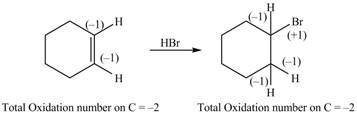
Figure 10
The oxidation number of reactant is equal to the oxidation number of the product. Therefore, the reaction is neither oxidation reaction nor reduction reaction.
The compound neither oxidized nor reduced.
(g)
Interpretation:
The given reaction is to be classified as an oxidation, a reduction, or neither. The number of electrons transferred in the reaction is to be stated when the corresponding reaction is oxidation or reduction.
Concept introduction:
A redox reaction is a type of reaction in which one reacting species gets oxidize and other reacting species is reduced. The exchange of electron takes place between the reacting species. Water molecules and protons can be used to balance a redox chemical reaction in the acidic medium.
Answer to Problem 10.26P
The given reaction is to be classified as a reduction reaction. The number of electrons transferred in the reaction is
Explanation of Solution
The given reaction is shown below.
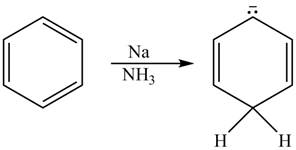
Figure 11
The reactant and products with oxidation number of atoms are shown below.
\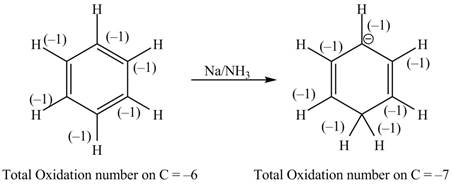
Figure 12
The oxidation number of reactant is higher than the oxidation number of the product. Therefore, the reaction is reduction reaction.
The number of electrons transfer in the reaction is given by the expression as shown below.
Where,
•
•
Substitute the values of
Therefore, the number of electrons transferred in the reaction is
The number of electrons transferred in the reduction reaction is
Want to see more full solutions like this?
Chapter 10 Solutions
Organic Chemistry
- The carbon dioxide exhaled in the breath of astronauts is often removed from the spacecraft by reaction with lithium hydroxide 2LiOH(s)+CO2(g)Li2CO3(s)+H2O(l) Estimate the grams of lithium hydroxide required per astronaut per day. Assume that each astronaut requires 2.50 103 kcal of energy per day. Further assume that this energy can be equated to the heat of combustion of a quantity of glucose, C6H12O6, to CO2(g) and H2O(l). From the amount of glucose required to give 2.50 103 kcal of heat, calculate the amount of CO2 produced and hence the amount of LiOH required. The H for glucose(s) is 1273 kJ/mol.arrow_forwardEthanol, C2H5OH, is a gasoline additive that can be produced by fermentation of glucose. C6H12O62C2H5OH+2CO2 (a) Calculate the mass (g) of ethanol produced by the fermentation of 1.000 lb glucose. (b) Gasohol is a mixture of 10.00 mL ethanol per 90.00 mL gasoline. Calculate the mass (in g) of glucose required to produce the ethanol in 1.00 gal gasohol. Density of ethanol = 0.785 g/mL. (c) By 2022, the U. S. Energy Independence and Security Act calls for annual production of 3.6 1010 gal of ethanol, no more than 40% of it produced by fermentation of corn. Fermentation of 1 ton (2.2 103 lb) of corn yields approximately 106 gal of ethanol. The average corn yield in the United States is about 2.1 105 lb per 1.0 105 m2. Calculate the acreage (in m2) required to raise corn solely for ethanol production in 2022 in the United States.arrow_forwardA rebreathing gas mask contains potassium superoxide, KO2, which reacts with moisture in the breath to give oxygen. 4KO2(s)+2H2O(l)4KOH(s)+3O2(g) Estimate the grams of potassium superoxide required to supply a persons oxygen needs for one hour. Assume a person requires 1.00 102 kcal of energy for this time period. Further assume that this energy can be equated to the heat of combustion of a quantity of glucose, C6H12O6, to CO2(g) and H2O(l). From the amount of glucose required to give 1.00 102 kcal of heat, calculate the amount of oxygen consumed and hence the amount of KO2 required. The ff0 for glucose(s) is 1273 kJ/mol.arrow_forward
- Identify the type (synthesis, decomposition, single displacement, double displacement, or combustion) of each of the following reactions: (a) magnesium + oxygen → magnesium oxide (b) copper (II) chloride + aluminum → aluminum chloride + copper (c) propane + oxygen → carbon dioxide + water_ (d) calcium carbonate → calcium oxide + carbon dioxide (e) sulfuric acid + sodium hydroxide → sodium sulfate + water (f) FeCl3 + Mg → MgCl₂ + Fe (g) NaN3 → Na + N₂_ (h)Mg(OH)₂ + AgNO3 → Mg(NO3)2 + AgOH (i) H₂ + O₂ → H₂O (j) CH4 + O₂ → CO₂ + H₂Oarrow_forward21. (a) Write the balanced chemical equation for the combustion of propane (C3H3). How does the potential energy change between the reactants and products in this reaction? (b) Draw a potential energy diagram for this chemical reaction. (c) What is the relationship between these energy changes and the breakdown and/or formation of covalent bonds? Explain your ideas in at least two (2) sentences and remember to use vocabulary terms.arrow_forwardFor each of the following reactions, suggest two soluble ionic compounds that, when mixed together in water, result in the net ionic equation given: (a) 2 Ag+ (aq) + CO3²¯ (aq) → Ag₂CO3(s) (b) Mg²+ (aq) + 2 OH¯(aq) → Mg(OH)₂(s), the suspension present in milk of magnesia 3+ (c) 3 Ca³+ (aq) + 2 PO2 (aq) → Ca3(PO4)2(s), gypsum, a component of concretearrow_forward
- 4NO2(g)+O2(g)+2H2O(l) → 4HNO3(aq) The generation of the electricity used in a medium-sized home produces about 19 kg of NO2 per year. Assuming that there is adequate O2 and H2O, what mass of HNO3, in kg, can form from this amount of NO2 pollutant?arrow_forwardEthyl alcohol (C2H5OH), also called grain alcohol, can be made by the fermentation of glucose: C6H12O6→ 2 C2H5OH + 2 CO2If a 84.6% yield of ethyl alcohol is obtained, What mass of ethyl alcohol will be produced from 750 g of glucose? What mass (in kg) of glucose should be used to produce 475 g of C2H5OH?arrow_forwardWhen tin comes into contact with oxygen in the air, tin (IV) oxide, SnO2 is formed. Sn (s) + O2 (g) SnO2 (s) A piece of tin foil, 8.25 cm x 21.5 cm x 0.600 mm (d = 7.28 g / cm), exposed to oxygen. (A) Assuming the reaction of all tin, what is the mass of the oxidized tin foil? (B) Air is about 21% oxygen by volume (d = 1.309 g / L at 25 ° C, 1 atm). How many liters of air is required to fully react with tin foil?arrow_forward
- There are two steps in the extraction of copper metal from chalcocite, a copper ore. In the first step, copper(I) sulfide and oxygen react to form copper(I) oxide and sulfur dioxide: 2 Cu,S(s)+3 0,(g)→2 Cu,O(s)+2 SO,(g) In the second step, copper(I) oxide and carbon react to form copper and carbon monoxide: 圖 Cu, O(s)+C(s)-2 Cu(s)+CO(g) Write the net chemical equation for the production of copper from copper(I) sulfide, oxygen and carbon. Be sure your equation is balanced. 山因arrow_forwardBalance each of the following equations. Classify each reaction as synthesis, decomposition, single-displacement, or double-displacement. (a) H2 + Br2→HBr (b) BaO2s + H2SO4aq→BaSO4s + H2O2aq (c) BaClO32→ΔBaCl2 + O2 (d) CrCl3 + AgNO3→CrNO33 + AgCl (e) H2O2→H2O + O2arrow_forward2) au Fill in reactants, products or conditions needed to complete the following reactions. a) /T b) () مر d) Br Br H Br ott Mecht Tscarrow_forward
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning


