
Concept explainers
(a)
Interpretation:
The probability for the particle having wavefunction
Concept introduction:
For the normalization of the wavefunction, the wavefunction is integrated as a product of its conjugate over the entire limits. It is expressed by the equation as given below.
Where,
•
•
•
Answer to Problem 10.27E
The probability for the particle having wavefunction
Explanation of Solution
For the probability of the wavefunction the expression is as follows.
Where,
•
•
•
•
Substitute the values in the above equation as follows.
The above equation is simplified as given below.
The probability for the particle having wavefunction
(b)
Interpretation:
The probability for the particle having wavefunction
Concept introduction:
For the normalization of the wavefunction, the wavefunction is integrated as a product of its conjugate over the entire limits. It is expressed by the equation as given below.
Where,
•
•
•
Answer to Problem 10.27E
The probability for the particle having wavefunction
Explanation of Solution
For the probability of the wavefunction the expression is as follows.
Where,
•
•
•
•
Substitute the values in the above equation as follows.
The above equation is simplified as follows.
The probability for the particle having wavefunction
(c)
Interpretation:
The probability for the particle having wavefunction
Concept introduction:
For the normalization of the wavefunction, the wavefunction is integrated as a product of its conjugate over the entire limits. It is expressed by the equation as given below.
Where,
•
•
•
Answer to Problem 10.27E
The probability for the particle having wavefunction
Explanation of Solution
For the probability of the wavefunction the expression is as follows.
Where,
•
•
•
•
Substitute the values in the above equation as follows.
The above equation is simplified as follows.
The probability for the particle having wavefunction
(d)
Interpretation:
The probability for the particle having wavefunction
Concept introduction:
For the normalization of the wavefunction, the wavefunction is integrated as a product of its conjugate over the entire limits. It is expressed by the equation as given below.
Where,
•
•
•
Answer to Problem 10.27E
The probability for the particle having wavefunction
Explanation of Solution
For the probability of the wavefunction the expression is as follows.
Where,
•
•
•
•
Substitute the values in the above equation as follows.
The above equation is simplified as given below.
The probability for the particle having wavefunction
(e)
Interpretation:
The probability for the particle having wavefunction
Concept introduction:
For the normalization of the wavefunction, the wavefunction is integrated as a product of its conjugate over the entire limits. It is expressed by the equation as given below.
Where,
•
•
•
Answer to Problem 10.27E
The probability for the particle having wavefunction
Explanation of Solution
For the probability of the wavefunction the expression is as follows.
Where,
•
•
•
•
Substitute the values in the above equation as follows.
The above equation is simplified as follows.
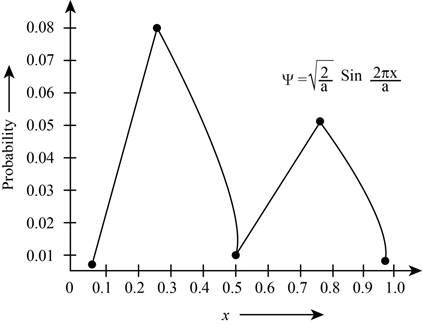
The plot the probabilities versus

Figure 1
The plot shows the probability for the given wave function. According to this plot, the probability of finding the particle is maximum in the range of
The probability for the particle having wavefunction
Want to see more full solutions like this?
Chapter 10 Solutions
Physical Chemistry
- Calculate the expectation value of x? (i.e., (x²)) of the quantum harmonic oscillator x from equilibrium when the oscillator is in the v = 0 and in the v = 1 quantum states.arrow_forwardThe ground-state wavefunction for a particle confined to a one dimensional box of length L is Ψ =(2/L)½ sin (πx/L) Suppose the box 10.0 nm long. Calculate the probability that the particle is: (a) between x = 4.95 nm and 5.05 nm (b) between 1.95 nm and 2.05 nm, (c) between x = 9.90 and 10.00 nm, (d) in the right half of the box and (e) in the central third of the box.arrow_forwardImagine a particle free to move in the x direction. Which of the following wavefunctions would be acceptable for such a particle? In eachcase, give your reasons for accepting or rejecting each function. (i) Ψ(x)=x2; (ii) Ψ(x)=1/x; (iii) Ψ(x)=e-x^2.arrow_forward
- Determine the number and location of the maxima in the plot r2R*(r) R(r) for the 2s wavefunction.arrow_forwardCalculate the value of ml for a proton constrained to rotate in a circle of radius 100 pm around a fixed point given that the rotational energy is equal to the classical average energy at 25 degrees C. (Mass of a proton = 1.6726 x 10^-27 kg, classical average energy=1/2kBT, where kBT is Boltzman constant = 1.30 x 10^ -23 J K^-1, and T is the temperature.)arrow_forwardThe wave function for the ground state of the harmonic oscillator is Vo(x) = Ce-[mw/(2ħ)]x² where C is an arbitrary constant, ħ is Planck's constant divided by 2π, m is the mass of the particle, W = ✓k/m, and k is the "spring constant" for the harmonic oscillator. Part A Normalize this wave function. What is the (positive) value of C once this wave function is normalized? You will need the formula Se -∞ Express your answer in terms of w, m, ħ, and T. ► View Available Hint(s) C = 17 ΑΣΦ xa Xh عات a √x vx 18 X> IXI -ax² X.10n X = ? wwwwwwwwww √. aarrow_forward
- Q1: Find the first excited state of harmonic oscillator using the equation: . (x) = A, (a.)"V(x), with E, (n+hw Q2: If f and T are the position and Kinetic energy operators, respectively. Where: Calculate [f ,T]. 2m axarrow_forwardWhat is the degeneracy of J=1 for a symmetric, and spherical rotor? For each rotor, give the complete set of quantum numbers for each state. (Recall that there should be a unique set of quantum numbers for each state and that the total number of states should match the degeneracy.)arrow_forwardCalculate the zero-point energy of a harmonic oscillator consisting of a particle of mass 2.33 × 10−26 kg and force constant 155 N m−1.arrow_forward
- Functions of the form sin(nπx/L), where n = 1, 2, 3 …, are wavefunctions in a region of length L (between x = 0 and x = L). Show that the wavefunctions with n = 1 and 2 are orthogonal; you will find the necessary integrals in the Resource section.arrow_forwardA normalized wavefunction for a particle confined between 0 and L in the x direction is ψ = (2/L)1/2 sin(πx/L). Suppose that L = 10.0 nm. Calculate the probability that the particle is (a) between x = 4.95 nm and 5.05 nm, (b) between x = 1.95 nm and 2.05 nm, (c) between x = 9.90 nm and 10.00 nm, (d) between x = 5.00 nm and 10.00 nm.arrow_forwardConstruct a slater determinant for the ground state He atom and show that is gives you, Ψ=1s α(2)1s β(1) - 1s α(1)1s β(2) where "1s" represents the "Ψooo" Hydrogenic 1s spatial wavefunction and α(1), β(1) represents spin up and spin down for electron 1, respectively. (Note that there is a normalization factor missing in the text. You can ignore this normalization factor also)arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





