
Concept explainers
(a)
Interpretation: The constitutional isomer formed by the reaction of given alkene with [1]
Concept introduction: The compounds with same chemical formula but different arrangement of atoms in space are known as the constitutional isomer.
Hydration of
Answer to Problem 10.29P
The constitutional isomer formed by the reaction of given alkene with
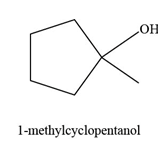
Figure 1
The constitutional isomer formed by the reaction of given alkene with
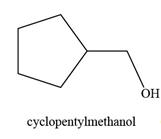
Figure 2
Explanation of Solution
The compounds with same chemical formula but different arrangement of atoms in space are known as the constitutional isomer.
Hydration of alkenes is one of the methods used for the formation of alcohol.
Hydroboration reaction is a two step reaction, which involves conversion of alkene into alcohol. This type of reaction follows Anti-Markovnikov’s rule.
In hydration reactions, the mode of addition is anti-addition, whereas in hydroboration-oxidation, the mode of addition is syn addition.
The given alkene is,
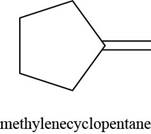
Figure 3
The product formed by the reaction of methylenecyclopentane with
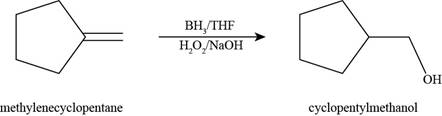
Figure 4
The general steps followed by the hydration reaction are stated below:
• First protonation of the alkene take place to generate the carbocation.
• Formation of protonated alcohol.
• Deprotonation.
The product formed by the reaction of methylenecyclopentane with
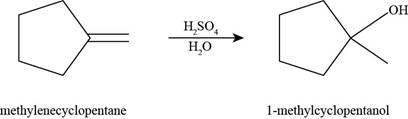
Figure 5
The constitutional isomer formed by the reaction of given alkenes with [1]
(b)
Interpretation: The constitutional isomer formed by the reaction of given alkenes with [1]
Concept introduction: The compounds with same chemical formula but different arrangement of atoms in space are known as the constitutional isomer.
Hydration of alkenes is one of the methods used for the formation of alcohol.
Answer to Problem 10.29P
The constitutional isomer formed by the reaction of given alkenes with
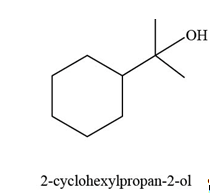
Figure 6
The constitutional isomer formed by the reaction of given alkenes with
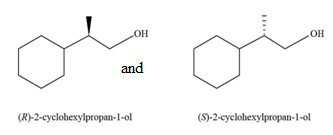
Figure 7
Explanation of Solution
The compounds with same chemical formula but different arrangement of atoms in space are known as the constitutional isomer.
Hydration of alkenes is one of the methods used for the formation of alcohol.
Hydroboration reaction is a two step reaction, which involves conversion of alkene into alcohol. This type of reaction follows Anti-Markovnikov’s rule.
In hydration reactions, the mode of addition is anti-addition, whereas in hydroboration-oxidation, the mode of addition is syn addition.
The given alkene is,
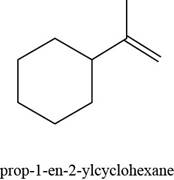
Figure 8
The product formed by the reaction of
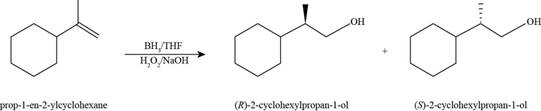
Figure 9
The general steps followed by the hydration reaction are stated below:
• First protonation of the alkene take place to generate the carbocation.
• Formation of protonated alcohol.
• Deprotonation.
The product formed by the reaction of
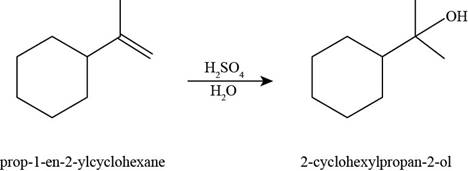
Figure 10
The constitutional isomer formed by the reaction of given alkenes with [1]
(c)
Interpretation: The constitutional isomer formed by the reaction of given alkene with [1]
Concept introduction: The compounds with same chemical formula but different arrangement of atoms in space are known as the constitutional isomer.
Hydration of alkenes is one of the methods used for the formation of alcohol.
Answer to Problem 10.29P
The constitutional isomer formed by the reaction of given alkene with
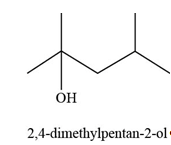
Figure 11
The constitutional isomer formed by the reaction of given alkene with
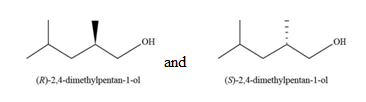
Figure 12
Explanation of Solution
The compounds with same chemical formula but different arrangement of atoms in space are known as the constitutional isomer.
Hydration of alkenes is one of the methods used for the formation of alcohol.
Hydroboration reaction is a two step reaction, which involves conversion of alkene into alcohol. This type of reaction follows Anti-Markovnikov’s rule.
In hydration reactions, the mode of addition is anti-addition, whereas in hydroboration-oxidation, the mode of addition is syn addition.
The given alkene is,
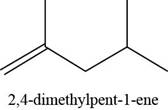
Figure 13
The product formed by the reaction of methylenecyclopentane with
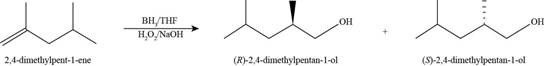
Figure 14
The general steps followed by the hydration reaction are stated below:
• First protonation of the alkene take place to generate the carbocation.
• Formation of protonated alcohol.
• Deprotonation.
The product formed by the reaction of methylenecyclopentane with
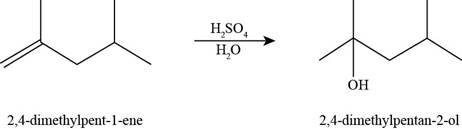
Figure 15
The constitutional isomer formed by the reaction of given alkenes with [1]
Want to see more full solutions like this?
Chapter 10 Solutions
Organic Chemistry
- Synthesize each compound from benzene. a. SO₂H 19일 g b. 00 이 Harrow_forwardDraw the products formed when the following alkynes are treated with each set of reagents: [1] H2O, H2SO4, HgSO4; or [2] R2BH followed by H2O2, −OH.arrow_forwardDraw all constitutional isomers formed when each alkene is treated with NBS + hv. -CH3 CH c. CH2=C(CH2CH3)2 a. CH,CH=CHCH3 b.arrow_forward
- 1. Ethylene glycol, a major component of antifreeze, becomes especially poisonous when it is oxidized by the liver. Draw two oxidation products that could be derived from ethylene glycol. 2.When 2-methyl-1-butanol is dehydrated in an acid medi- um to an alkene, it yelds mainly 2-methyl-2-butene rather than 2-methyl-1-butene. This indicates that the dehydra- tion to an alkene is at least a two-step reaction. Suggest a mechanism to explain the reaction.arrow_forwardWhen 2-pentene is treated with Cl, in methanol, three products are formed. Account for the formation of each product (you need not explain their relative percentages). CI OCH3 H,CO CI CI CI Cle CH;CHCHCH,CH, + CH;CHCHCH,CH3 + CH,CHČHCH,CH, CH;OH CH;CH=CHCH,CH, 50% 35% 15%arrow_forwardIdentify the structure of 3-phenylpentane 1 ΠΙ CHÍCH,CH HẠCH CH a. I IV O > d. II IV. OH СЊСЊ СНСН СН V.arrow_forward
- 3. What alcohol is formed when each alkene is treated with H2O in the presence of H2SO4 (as the catalyst)? а. СНЗСН-СНСНЗ b. CH3CH2CH=CH2 С. CH3 CH3arrow_forwardIndicate the product obtained by reacting A and B. A || H3C-S-CH₂ Na* || CH 3 B CH2. C. CH3 H3C CH3arrow_forwardWhich reaction will produce this alkene as the major product? A. A B. B C. Both D. Neither (a) (b) Br NaOCH3 Δ H₂C P(Ph)3arrow_forward
- A. the products formed when an alkene reacts with these reagents: (i) H2 in the presence of Pd catalyst (ii) HClarrow_forwardName each compound in which the benzene ring is best treated as a substituent. a. H;C-CH;-CH-CH-CH,-CH; H;C b. ČH;-CH2-CH;-CH2-C=C-CH, CH3 c. CH;-CH- CH-C=CH-CH-CH2-CH; CH3 CH,arrow_forwardDraw the products formed when alkene reacts with these reagents: (i) H2 in the presence of Pd catalyst, (ii) HCl.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





