
Concept explainers
(a)
Interpretation:
The electronic configuration of lithium has to be given.
The given orbital diagram is,
Concept Introduction:
Aufbau’s principle:
Aufbau’s principle states that electrons are filled into atomic orbitals in the increasing order of orbital energy level. According to the Aufbau principle the available atomic orbitals with the lowest energy levels are occupied before those with higher energy levels.
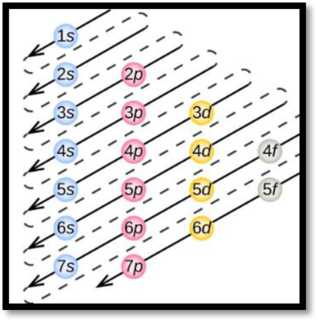
Figure 1
(a)
Explanation of Solution
The given orbital diagram is,
Lithium is a chemical element with chemical symbol
(b)
Interpretation:
The electronic configuration of phosphorous has to be given.
The given orbital diagram is,
Concept Introduction:
Refer to part (a).
(b)
Explanation of Solution
The given orbital diagram is,
The electronic configuration of Phosphorous is
(c)
Interpretation:
The electronic configuration of zinc has to be given.
The given orbital diagram is,
Concept Introduction:
Refer to part (a).
(c)
Explanation of Solution
The given orbital diagram is,
The electronic configuration of Zinc is
(d)
Interpretation:
The electronic configuration of sodium has to be given.
The given orbital diagram is,
Concept Introduction:
Refer to part (a).
(d)
Explanation of Solution
The given orbital diagram is,
Sodium is a chemical element with chemical symbol
(e)
Interpretation:
The electronic configuration of potassium has to be given.
The given orbital diagram is,
Concept Introduction:
Refer to part (a).
(e)
Explanation of Solution
The given orbital diagram is,
Potassium is a chemical element with chemical symbol
Want to see more full solutions like this?
Chapter 10 Solutions
EBK FOUNDATIONS OF COLLEGE CHEMISTRY
- What is the mole fraction of calcium chloride in 3.35 m CaCl2 (aq)? The molar mass of CaCl2 is 111.0 g/mol and the molar mass of water is 18.02 g/mol.arrow_forwardThe universal gas constant, R, is defined as the following: kg · m² s2 · K• mol R = 8.314 What would be its value in the following unit? lb· cm? day2 · K• molarrow_forwardThe pressure P of a sample of oxygen gas that is compressed at a constant temperature is related to the volume V of gas by a reciprocal function of the form P = k V . (a) A sample of oxygen gas that occupies 0.674 m3 exerts a pressure of 39 kPa at a temperature of 293 K (absolute temperature measured on the Kelvin scale). Find the value of k (in kPa · m3) in the given model.arrow_forward
- What volume of carbon dioxide gas (in mL) at 834 mm Hg and 25 °C can be produced when 2.3 g of MgCO3(s) are put into an Erlenmeyer flask containing 289 mL of 0.171 M HCl(aq) ?1 MgCO3(s) + 2 HCl(aq) → MgCl2(aq) + CO2(g) + H2O(l)arrow_forwardAt 1.00 atm and 0 °C, a 5.04 L mixture of methane (CH) and propane (C₂H₂) was burned, producing 16.5 g CO₂. What was the mole fraction of each gas in the mixture? Assume complete combustion. Xmethane = Xpropanearrow_forwardwhat is the mole fraction of 4.31 m KBr (aq)?arrow_forward
- Effervescent tablets contain both citric acid (C6H8O7) and sodium bicarbonate (NaHCO3) and release carbon dioxide gas when dissolved in water as well as forming trisodium citrate (Na3C6H5O7) and water. A typical effervescent tablet contains 1.00 g of citric acid and 1.92 g sodium bicarbonate. (a) Assuming that carbon dioxide acts as an ideal gas, determine the work done due to the evolution of carbon dioxide by the dissolution of one effervescent tablet in water at 25.0 °C and atmospheric pressure (1 atm i.e. 101325 Pa).arrow_forwardSulfur dioxide reacts with oxygen in the presence of plati- num to give sulfur trioxide: 2 SO2(g) + O2(g) → 2 SO3(g) Suppose that at one stage in the reaction, 26.0 mol SO2, 83.0 mol O2, and 17.0 mol SO3 are present in the reaction vessel at a total pressure of 0.950 atm. Calculate the mole fraction of SO3 and its partial pressure.arrow_forwardA metal cylinder with a capacity of 6.0 L is filled with compressed propane (C3H8). The pressure and temperature of the cylinder when it was initially filled were 120 atm and 75 ◦C, respectively. The molar mass of carbon is 12 g·mol−1 and the molar mass of hydrogen is 1 g·mol−1. a) How many moles of propane are in the cylinder? b) What is the mass of the propane inside the cylinder? c) After some time, the cylinder and its contents cool to 25 ◦C. What is the pressure in the cylinder after it has cooled?arrow_forward
- At 1.00 atm and 0 °C, a 5.04 L mixture of methane (CH4) and propane (C₂H₂) was burned, producing 16.5 g CO₂. What was the mole fraction of each gas in the mixture? Assume complete combustion. Xmethane = Xpropanearrow_forwardThe ideal gas equation is PV = nRT where P is pressure, V is volume, n is the number of moles, R is a constant, and T is temperature. You are told that a sample of gas has a pressure of P = 873 torr, a volume of V = 7280 mL, and a temperature of T = 310 K . If you use R = 8.206×10−² L · atm/(K · mol), which of the following conversions would be necessary before you could find the number of moles of gas, n, in this sample? Check all that apply. ► View Available Hint(s) Convert the pressure to atmospheres (atm). Convert the pressure to pascals (Pa). Convert the volume to cubic meters (m³). Convert the volume to liters (L). Convert the temperature to degrees Celsius (°C). Convert the temperature to degrees Fahrenheit (°F).arrow_forwardSuppose you want to calculate the number of moles of a gas released from a chemical reaction based on the ideal gas law equation. In the lab, you observe that 17.4 mL of gas are released at a temperature of 25.4°C and a pressure of 991 Torr. You begin calculations using the value for R as 0.08206 Latm mol·K Next, convert the remaining values to units suitable for use in this equation. V = T = K P = atmarrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
