
Concept explainers
Draw the major product obtained when each of the following
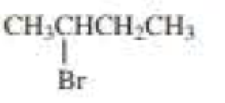
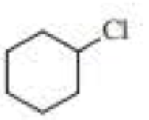
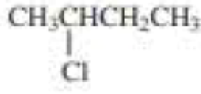
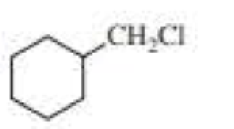
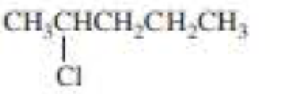
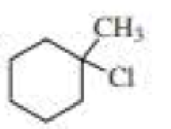
(a)
Interpretation:
Major product formed when given alkyl halides undergoes E2 reaction has to be drawn.
Concept Introduction:
An E2 reaction is a concerted, one-step reaction in which the proton and the halide ion are removed in the same step. A strong base favours an E2 reaction. A high concentration of a base favours an E2 reaction. If one of the reactants is charged, an E2 reaction will be favoured by the least polar solvent that will dissolve the reactant. If neither of the reactant is charged an E2 reaction is favoured by the protic polar solvent. The major product is a stable alkene, unless the reactants are sterically hindered.
Explanation of Solution
The product when given compound undergo E2 addition,
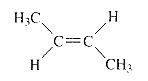
For an E2 reaction major product is the more stable alkene. The reactant is a secondary alkyl halide, undergo E2 reactions.
(b)
Interpretation:
Major product formed when given alkyl halides undergoes E2 reaction is to be identified.
Concept Introduction:
An E2 reaction is a concerted, one-step reaction in which the proton and the halide ion are removed in the same step.
Explanation of Solution
Chlorocyclohexane is a less stable conformer undergo E2 reaction as
The reactant is a secondary alkyl halide, undergo E2 reactions.
(c)
Interpretation:
Major product formed when given alkyl halides undergoes E2 reaction is to be identified.
Concept Introduction:
An E2 reaction is a concerted, one-step reaction in which the proton and the halide ion are removed in the same step. A strong base favours an E2 reaction. A high concentration of a base favours an E2 reaction. If one of the reactants is charged, an E2 reaction will be favoured by the least polar solvent that will dissolve the reactant. If neither of the reactant is charged an E2 reaction is favoured by the protic polar solvent. The major product is a stable alkene, unless the reactants are sterically hindered.
Explanation of Solution
Product of given compound when undergo E2 reaction,
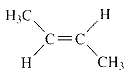
More substituted alkene is more stable therefore should be the major product.
(d)
Interpretation:
Major product formed when given alkyl halides undergoes E2 reaction is to be identified.
Concept Introduction:
An E2 reaction is a concerted, one-step reaction in which the proton and the halide ion are removed in the same step. A strong base favours an E2 reaction. A high concentration of a base favours an E2 reaction. If one of the reactants is charged, an E2 reaction will be favoured by the least polar solvent that will dissolve the reactant. If neither of the reactant is charged an E2 reaction is favoured by the protic polar solvent.
The major product is a stable alkene, unless the reactants are sterically hindered.
Explanation of Solution
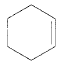
Product of given compound when undergo E2 reaction,
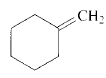
Chloromethyl cyclohexane on E2 reaction gives methylene cyclohexane. And E2 reaction is regioselective: major product is the more substituted stable alkene.
(e)
Interpretation:
Major product formed when given alkyl halides undergoes E2 reaction is to be identified.
Concept Introduction:
An E2 reaction is a concerted, one-step reaction in which the proton and the halide ion are removed in the same step. A strong base favours an E2 reaction. A high concentration of a base favours an E2 reaction. If one of the reactants is charged, an E2 reaction will be favoured by the least polar solvent that will dissolve the reactant. If neither of the reactant is charged an E2 reaction is favoured by the protic polar solvent.
The major product is a stable alkene, unless the reactants are sterically hindered.
Explanation of Solution
Product of given compound when undergo E2 reaction,
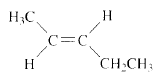
The reactant is a secondary alkyl halide, undergo E2 reactions.
(f)
Interpretation:
Major product formed when given alkyl halides undergoes E2 reaction is to be identified.
Concept Introduction:
An E2 reaction is a concerted, one-step reaction in which the proton and the halide ion are removed in the same step. A strong base favours an E2 reaction. A high concentration of a base favours an E2 reaction. If one of the reactants is charged, an E2 reaction will be favoured by the least polar solvent that will dissolve the reactant. If neither of the reactant is charged an E2 reaction is favoured by the protic polar solvent.
The major product is a stable alkene, unless the reactants are sterically hindered.
Explanation of Solution
Product of given compound when undergo E2 reaction,
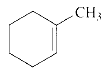
An E2 reaction is regioselective: major product is the more substituted stable alkene.
Want to see more full solutions like this?
Chapter 10 Solutions
Organic Chemistry
- What is the major product formed when the following compound undergoes an E1 reaction?arrow_forwardWhich of the following compounds will not undergo keto-enol tautomerism? ОН OH ОН OH ОНarrow_forwardRank the following carbonyl compounds in order of increasing reactivity in nucleophilic substitution reactions: H I II III IV ZIarrow_forward
- Draw the structures of the initially formed enol tautomers in the reactions of propyne and dicyclohexylethyne with dicyclohexylborane followed by NaOHNaOH and H2O2H2O2arrow_forwardWhich of the following sequences of reactions would convert toluene to 2-bromo-4-cyanotoluene? ? Br CN Nitration, bromination, reduction, diazotization, reaction with cyanide anion Bromination, nitration, reduction, diazotization, reaction with cyanide anion Bromination, nitration, diazotization, reduction , reaction with cyanide anion Nitration, bromination, diazotization, reduction, reaction with cyanide anionarrow_forwardDraw the starting structure that would lead to the major product shown under the provided conditions. Drawing H3O+ HOarrow_forward
- Determine a stepwise mechanism for the following reaction that illustrates why two substitution products are formed. Explain why 1-bromohex-2-ene reacts rapidly with a weak nucleophile (CH3OH) under Syl reaction conditions, even though it is a 1° alkyl halide. 1-bromohex-2-ene Part 1: Br CH₂OH The first step in the reaction proceeds according to which mechanism? CH₂CH₂CH₂CH=CHCH₂ + CH₂OH CH₂CH₂CH₂CH: Part 2: Br CHCH₂ Draw the missing resonance contributor. OCH3 + CH3CH₂CH₂CH=CHCH₂ CH3CH₂CH₂CH=CH-CH₂ + Br Br OCH 3 H₂CH₂CH₂CH=CH₂ view structure + + Br HBr Xarrow_forwardIdentify the dominant elimination mechanism (E1 or E2) for the following reaction. NaOH Neither Е2 Е1arrow_forwardWhich of the following species is attacked by benzene in the electrophilic nitration reaction? NO+ NO₂ NO₂ HNO3 N3™ +arrow_forward
- Draw the major organic product formed in each of the following transition-metal catalyzed reactions:arrow_forwardPropose a plausible mechanism for the following transformation: HO [H3O+] EtOH 19.55 The first three steps of the mechanism involve the formation of The first step is The second step is The third step is eTextbook and Media Save for Later Attemarrow_forwardProvide the missing reagents required for the following transformation: OH HO-arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
