
The amount of weight percent sulfur.
Answer to Problem 81AAP
The amount of weight percent sulfur is
Explanation of Solution
Show the chemical structure bonding of polyacrylonitrile and polybutadiene as:
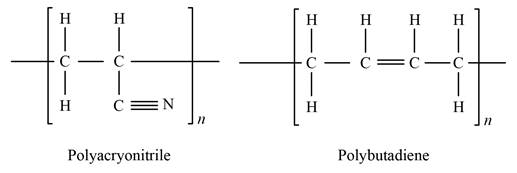
Express the weight per cent of sulfur.
Here, the molar mass of the sulfur is
Calculate the mass of sulfur at 22%.
Here, the number of mole in butadiene is
Calculate the copolymer total mass.
Here, the molar mass of the polybutadlene is
Conclusion:
The average molecular mass of the
The average molecular mass of the polybutadlene is
The average molecular mass of the sulfur is
Substitute
Calculate the fraction of butadlene.
Calculate the number of moles of butadlene using 200 kg of copolymer.
Substitute 3096.3 mol for mole butadlene and
Substitute 21,798 g for
Thus, the amount of weight percent sulfur is
Want to see more full solutions like this?
Chapter 10 Solutions
Foundations of Materials Science and Engineering
- sketch the generalized stress-strain curve for a semi-crystalline thermoplastic polymer.arrow_forwardBakelite is an example of polymer. Select one: O Thermoset O Thermoplastic O Composite O Elastomersarrow_forward1 Which one below is true for thermoplastic polymers? * O They do not soften when heated and do not harden when cooled. O Most cross-linked polymers are thermoplastics. O None of them. O They are harder and stronger than thermosetting polymers.arrow_forward
- You've been asked to select suitable candidate polymers for these applications: an ice tray with an operational temperature of -10C, a boat bumper with an operational temperature of -20C, a safety helmet, and a hairdryer with an operational temperature of 55C. Which of the below is a good choice of materials? O a. Low-density polyethylene, nylon, polypropylene, and high-density polyethylene O b. High-density polyethylene, low-density polyethylene, polypropylene, and polypropylene O c. High-density polyethylene, polypropylene, polytetrafluoroethylene, and high-density polyethylene O d. polypropylene, low-density polyethylene, poly(vinyl chloride), and polypropylene O e. Low-density polyethylene, low-density polyethylene, polytetrafluoroethylene, and high-density polyethylenearrow_forwardWhat factors influence the crystallinity ofpolymers? Explain the development androle of crystallinity in PET and nylon.arrow_forwardFor a given polymer, the activation energy for stress relaxation was measured to be 10 kJ/mol. If the stress relaxation time for this polymer at room temperature is 3,600 s, what would be the relaxation time at 100 ◦C?arrow_forward
- (b) Explain how the structural differences between the three main sub-groups of engineering polymers affect the physical and mechanical properties of the polymers. Use simple diagrams to illustrate your answer. Include examples and applications of specific polymers for each sub-group.arrow_forwardThere are 4 classifications of structural molecular characteristics of polymers - Linear, Network, Crosslinked. What is the final type? Answer:arrow_forwardWhy do thermoplastic polymers start to turn white as they are plastically deformed?arrow_forward
- Briefly explain how each of the following influences the tensile or yield strength of a semicrystalline polymer and why: (a) Molecular weight (b) Degree of crystallinity (c) Deformation by drawingarrow_forwardAn unknown polymer sample was found to have the following molecular weight distribution: Molecular weight range 8,000 - 20,000 20,000 - 32,000 32,000 - 44,000 44,000 - 56,000 56,000 - 68,000 68,000 -80,000 80,000 - 92,000 0.03 Knowing that the monomer is methyl methacrylate, which of the below is the number average degree of polymerization for this polymer sample? Select one: O a. O b. DP 476 O C. DP = 338 DP = 664 O d. DP 544 O e. DP = 801 Xi 0.05 0.15 0.21 0.28 0.18 0.10 Wi 0.02 0.08 0.17 0.29 0.23 0.16 0.05arrow_forwardWhat is the purpose of a plasticizer, and what specific effect on room temperature behavior is likely when a plasticizer is added?arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





