
Interpretation:
Major product has to be predicted for the given reaction.
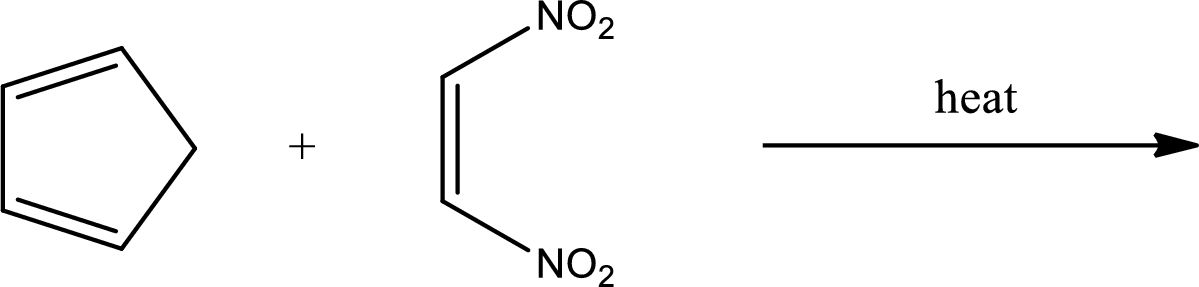
Concept Introduction:
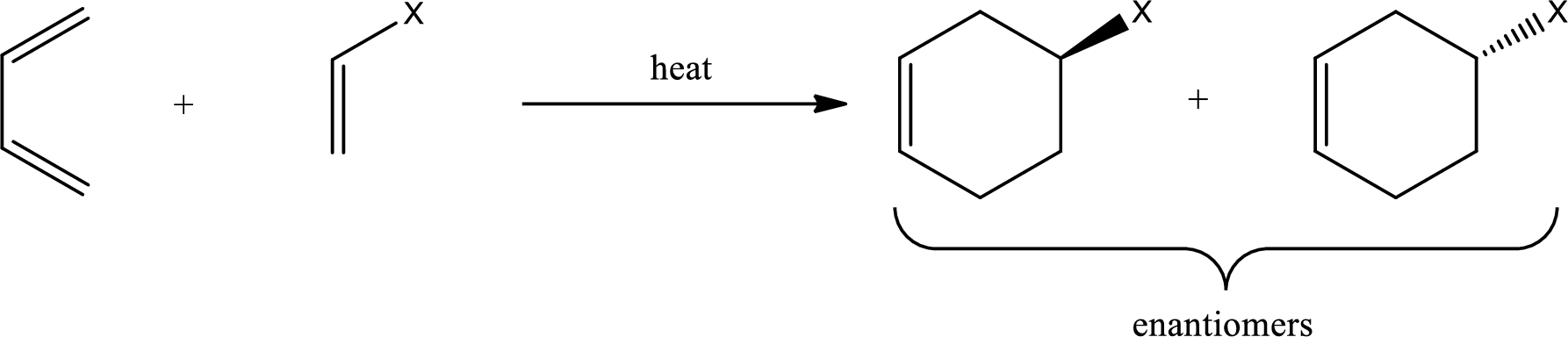
Diels-Alder reaction is the 4+2 addition reaction. Here a total of six pi bonds are involved resulting in formation of one pi bond and two single bonds. Diels-Alder reaction occurs via a single concerted step and not by involvement of ions. If a monosubstituted ethylene is used, the product obtained contains a stereocenter. This can be represented as,
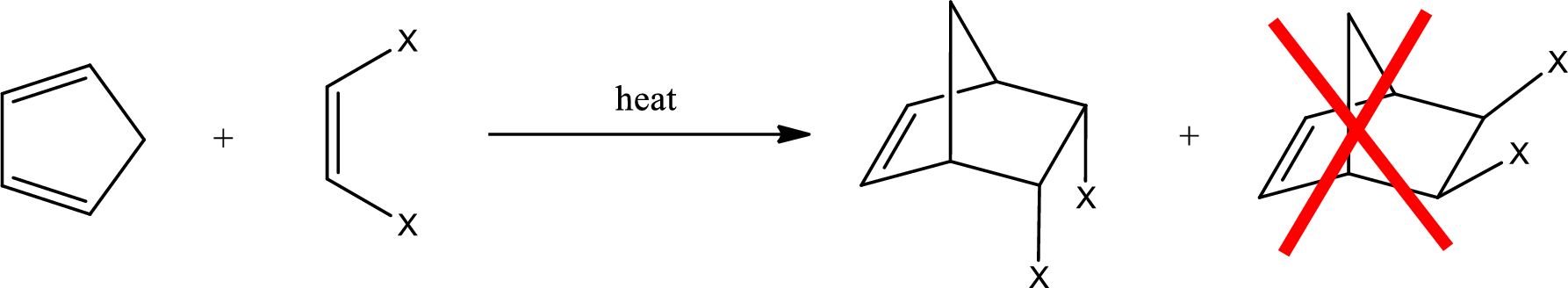
If a cyclic diene is considered, then the product obtained will be a bicyclic compound. Diels-Alder reaction that involves cyclopentadiene results in a bicyclic structure. If the dienophile has a cis configuration, then the two groups have to be in cis configuration to each other. The product obtained will have the groups in endo position. It is a meso compound.
If the dienophile has a trans configuration, then the two groups have to be in trans configuration to each other. The product obtained will have the groups as shown below,
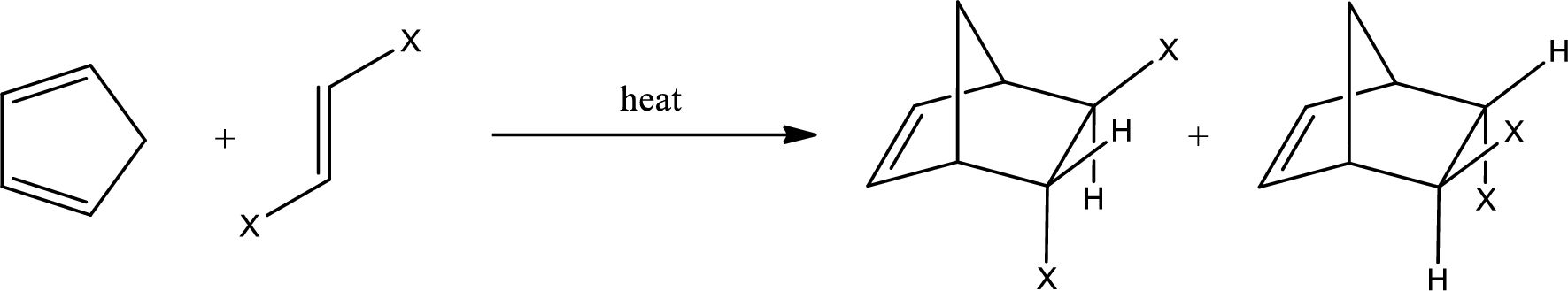
The above two structures are enantiomers.
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
- Identify the expected major product for the following reaction:arrow_forwardFor each of the following reactions predict the major product(s)arrow_forwardPlease predict the product for each of the following reactions. Make sure to clearly indicate the correct stereochemistry for each compound:arrow_forward
