
Concept explainers
(a)
Interpretation:
The product of the given reaction should be given, respect with the given conditions.
Concept introduction:
Chromic Acid:
Chromic Acid (
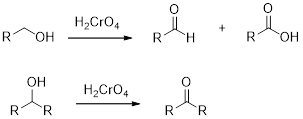
(b)
Interpretation:
The product of the given reaction should be given, respect with the given conditions.
Concept introduction:
Hypochlorous acid
Hypochlorous acid (
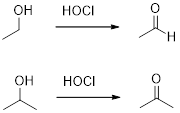
(c)
Interpretation:
The product of the given reaction should be given, respect with the given conditions.
Concept introduction:
Swern oxidation:
Primary and secondary alcohols undergoes oxidation reaction using dimethyl sulfoxide, oxalyl chloride and triethyl
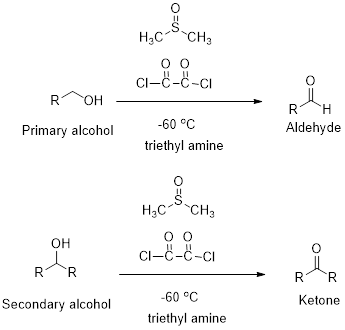
Want to see the full answer?
Check out a sample textbook solution
Chapter 10 Solutions
Organic Chemistry (8th Edition)
- Which product(s) will be formed in the following reaction? xs HI а. b. но С. OH d. e. Both alcohols f. Both alkyl iodides g. One alcohol and one alkyl iodidearrow_forwardWhich of the following alcohols can be prepared from a Grignard reagent and ethylene oxide? A. only 1 B. only 1 and 2 C. only 1, 2 and 3 D. 1, 2, 3 and 4arrow_forwardThe mechanism of dehydration of a secondary alcohol to give an alkene can involve the formation of Select one: O a. carbocations Ob. free radicals Oc. pentavalent transition state O d. carbanionsarrow_forward
- cis-Cyclohexane-1,2-diol can be synthesized from cyclohexene by using which reagent? a.O3 b.OsO4 c.H2SO4 d.mCPBAarrow_forward10. Which of the following is the structure of the rearranged carbocation in the following dehydration? OH H₂SO4 A. B. C. D.arrow_forwardWhich one is the major organic product obtained from the following reaction? LİALH4, Ether H30 а. 4-hydroxy-2-pentanone b. 2-pentanone С. 2-pentanol d. 4-penten-2-olarrow_forward
- 5. What reagents are needed to convert toluene (C,H,CH,) to each compound? a. C.H.COOH b. C.H₂CH₂Br c. p-bromotoluene d. o-nitrotoluene e. p-ethyltoluene f.arrow_forward8. Which of the following compounds would be expected to give a positive Tollens' test? a. 3-Pentanone b. Cyclohexanone c. 3-Methylbutanal d. Cyclopentanol e. 2,2-Dimethyl-1-pentanol f. Acetaldehydearrow_forward10. Write chemical equations for the reaction of ethyl propanoate with each reagent below. a) LIAIH4/ H3O* b) NH3 d) NaOH c) C2H5NH2arrow_forward
- Consider the structure of pent-2-ene, if it undergoes ozonolysis, which of the following final product is formed? a.Ethanal and Propanal b.Ethanal and Propanone c.CO2 and Propanal d.Ethanal and CO2arrow_forwardSpecify both the alcohol starting material and the reagents you would use in each step in a synthesis of the compound shown. If the synthesis requires only two steps enter "none" for step 3. r Alcohol Starting Materials 1. methanol 2. ethanol 3. 1-propanol 4. 2-propanol 5. cyclohexanol Reagents available a. LiAlH₁ f. PBr3 b. H₂SO4 c. HCI d. HBr i. CH3 MgBr; then H3O+ e. SOC1₂ j. CH3 CH₂ MgBr; then H3O+ g. CrO3, H₂ SO4, H₂O h. NaH k. CH3 CH₂ CH₂ MgBr; then H3O+ I. C6H5 MgBr (phenylmagnesium bromide); then H3O+ m. (CH3)2 CHMgBr: then H3O+t n. Dess-Martin periodinane (DMP) Write the number/letters of the alchol/reagents in the boxes below. Alcohol starting material Reagent for step 1 Reagent for step 2 Reagent for step 3arrow_forwardDraw the structure of the product formed on KMnO4 oxidation of the following alcohols. a. CH3-CH2-CH2-CH2-OH b. C. OH OHarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





