
(a)
Interpretation:
Line angle formula to be identified for 2,2,4-trimethylhexane.
Concept Introduction:
Example of
In line angle formula each line indicates 2 carbon are linked to each other through a single bond.
Answer to Problem 19P
The line angle formula for 2,2,4-trimethylhexane.
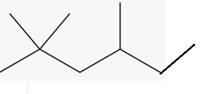
Explanation of Solution
The line angle formula for 2,2,4-trimethylhexane.
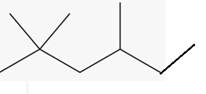
(b)
Interpretation:
Line angle formula to be identified for 2,2-dimethylpropane.
Concept Introduction:
Example of alkane are propane having formula C3 H8, butane having formula C4 H10 etc. Their molecular formula indicate the number of hydrogen is 2 more than twice the number of carbon in the structure and thus the derived formula for alkane is Cn H2n +2.
In line angle formula each line indicates 2 carbon are linked to each other through a single bond.
Answer to Problem 19P
The line angle formula for 2,2-dimethylpropane.

Explanation of Solution
The line angle formula for 2,2-dimethylpropane.

(c)
Interpretation:
Line angle formula to be identified for 3-ethyl-2,4,5-trimethyloctane.
Concept Introduction:
Example of alkane are propane having formula C3 H8, butane having formula C4 H10 etc. Their molecular formula indicate the number of hydrogen is 2 more than twice the number of carbon in the structure and thus the derived formula for alkane is Cn H2n +2.
In line angle formula each line indicates 2 carbon are linked to each other through a single bond.
Answer to Problem 19P
The line angle formula for 3-ethyl-2,4,5-trimethyloctane.
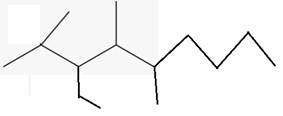
Explanation of Solution
The line angle formula for 3-ethyl-2,4,5-trimethyloctane.
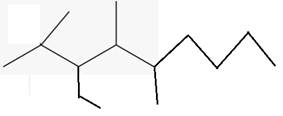
(d)
Interpretation:
Line angle formula to be identified for 5-butyl-2,2-dimethylnonane.
Concept Introduction:
Example of alkane are propane having formula C3 H8, butane having formula C4 H10 etc. Their molecular formula indicate the number of hydrogen is 2 more than twice the number of carbon in the structure and thus the derived formula for alkane is Cn H2n +2.
In line angle formula each line indicates 2 carbon are linked to each other through a single bond.
Answer to Problem 19P
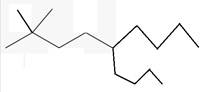
The line angle formula for 5-butyl-2,2-dimethylnonane.
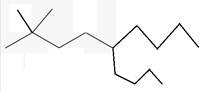
Explanation of Solution
The line angle formula for 5-butyl-2,2-dimethylnonane.
(e)
Interpretation:
Line angle formula to be identified for 4-isopropyloctane.
Concept Introduction:
Example of alkane are propane having formula C3 H8, butane having formula C4 H10 etc. Their molecular formula indicate the number of hydrogen is 2 more than twice the number of carbon in the structure and thus the derived formula for alkane is Cn H2n +2.
In line angle formula each line indicates 2 carbon are linked to each other through a single bond.
Answer to Problem 19P
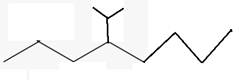
The line angle formula for 4-isopropyloctane.
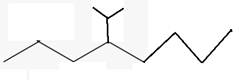
Explanation of Solution
The line angle formula for 4-isopropyloctane.
(f)
Interpretation:
Line angle formula to be identified for 3,3-dimethylpentane.
Concept Introduction:
Example of alkane are propane having formula C3 H8, butane having formula C4 H10 etc. Their molecular formula indicate the number of hydrogen is 2 more than twice the number of carbon in the structure and thus the derived formula for alkane is Cn H2n +2.
In line angle formula each line indicates 2 carbon are linked to each other through a single bond.
Answer to Problem 19P
The line angle formula for 3,3-dimethylpentane.
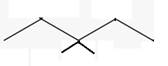
Explanation of Solution
The line angle formula for 3,3-dimethylpentane.
(g)
Interpretation:
Line angle formula to be identified for trans-1,3-dimethylcyclopentane.
Concept Introduction:
Example of alkane are propane having formula C3 H8, butane having formula C4 H10 etc. Their molecular formula indicate the number of hydrogen is 2 more than twice the number of carbon in the structure and thus the derived formula for alkane is Cn H2n +2.
In line angle formula each line indicates 2 carbon are linked to each other through a single bond.
Answer to Problem 19P
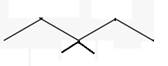
The line angle formula for trans-1,3-dimethylcyclopentane.
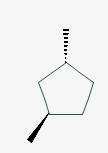
Explanation of Solution
The line angle formula for trans-1,3-dimethylcyclopentane.
(h)
Interpretation:
Line angle formula to be identified for Cis-1,2-diethylcyclobutane.
Concept Introduction:
Example of alkane are propane having formula C3 H8, butane having formula C4 H10 etc. Their molecular formula indicate the number of hydrogen is 2 more than twice the number of carbon in the structure and thus the derived formula for alkane is Cn H2n +2.
In line angle formula each line indicates 2 carbon are linked to each other through a single bond.
Answer to Problem 19P
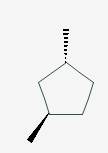
The line angle formula for Cis-1,2-diethylcyclobutane.
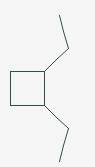
Explanation of Solution
The line angle formula for Cis-1,2-diethylcyclobutane.
Want to see more full solutions like this?
Chapter 11 Solutions
Introduction To General, Organic, And Biochemistry
- Draw condensed structural formulas for the following branched alkanes. a. 5-(sec-Butyl)decane b. 4,4-Diisopropyloctane c. 5-Isobutyl-2,3-dimethylnonane d. 4-(1,1-Dimethylethyl)octanearrow_forwardWhat is wrong with each of the following attempts name a cycloalkane using IUPAC rules? a. Dimethylcyclopropane b. 1-Methylcyclohexane c. 2,4-Dimethylcyclobutane d. 1-Propy 1-2-ethylcyclohexanearrow_forwardWrite skeletal formulas for the following alkanes and cycloalkanes. Use solid and broken lines to indicate stereochemistry. 1) 3-ethyl-2,4,5-trimethyloctane 2) 4-(1-methylethyl)octane 3) 5-butyl-2,2-dimethylnonane 4) trans-1,3-dimethylcyclopentane 5) cis-1,2-diethylcyclobutanearrow_forward
- Indicate whether each statement is true or false. Alkanes do not have any carbon-carbon multiple bonds. Cyclobutane contains a four-membered ring. Alkenes contain carbon-carbon triple bonds. Alkynes contain carbon-carbon double bonds. Pentene is a saturated hydrocarbon. 1-pentene is an unsaturated hydrocarbon. Cyclohexane is an aromatic hydrocarbon. The methyl group contains one less hydrogen atom than methane.arrow_forwardWrite the condense structure of the following: n-octane 2-methyl heptane 3-methyl heptane 4-methyl heptane 2,3-dimethyl hexane 2,4-dimethyl hexane 2,5-dimethyl hexane 3,4-dimethyl hexane 2,2-dimethyl hexane 3,3-dimethyl hexane 3-ethyl hexane 2,2,3-trimethylpentane 2,2,4-trimethylpentane 2,3,3-trimethylpentane 2,3,4-trimethylpentane 2-methyl-3-ethyl pentane 3-ethyl-3-methyl pentane 2,2,3,3-tetramethyl butanearrow_forwardDraw the structure of the following saturated hydrocarbons 3-isopropyl-2,4,5,5-tetramethylheptane 4 (1-methylpentyl)-1,2-dimethylcyclopentane 2-cyclopropyl-3,4,4-trimetnythexanearrow_forward
- Write structures for the following compounds. 3-ethyl-4-methylhexane 4-tert-butyl-2-methylheptane ) 3-ethyl-5-isobutyl-3-methylnonane D 5-isopropyl-3,3,4-trimethyloctanearrow_forwardArrange the following compounds according to increasing of boiling points Propane Methane Hexane Decane Octane Pentane 3-methyl hexane 2,2-dimethyl pentane 2,3,4-trimethyl pentane 2-methyl butanearrow_forwardGive the IUPAC name of the cycloalkane shown below. H3C. H3C *CH2CH2CH, CH3 O 4-butyl-1,2-dimethylcyclohexane O 1-butyl-3,4-dimethylcyclohexane 4-butyl-1-methyl-2-methylcyclohexane O 1, 2-dimethyl-4-butylcyclohexanearrow_forward
- Use IUPAC rules to name the following structures CH,CH3 b. C. CH3 CH3 CH3 CH,CH, CH;CH, B. Draw the structures of the following cycloalkanes 1. 2,3-dimethyl-5-propyldecane 2. 3,4,5-triethyloctane 3. 1-ethyl-3-propylcyclopentane 4. 1,2,2,4-tetramethylcyclohexanearrow_forwardWhich organic compounds is not a constitutional isomer of octane? 2,2-dimethyl hexane 2,3,4-trimethyl pentane 3-methyl heptane O cyclo octane 3-ethyl-2-methyl pentanarrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning,
Organic And Biological ChemistryChemistryISBN:9781305081079Author:STOKER, H. Stephen (howard Stephen)Publisher:Cengage Learning, General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning




