
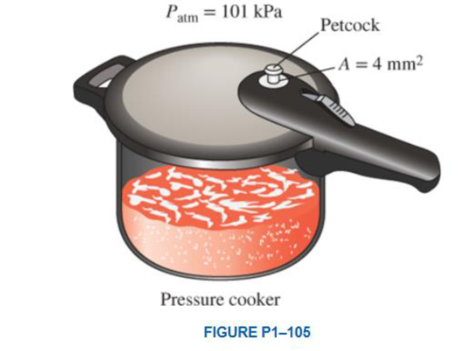
A pressure cooker cooks a lot faster than an ordinary pan by maintaining a higher pressure and temperature inside. The lid of a pressure cooker is well sealed, and steam can escape only through an opening in the middle of the lid. A separate metal piece, the petcock. sits on top of this opening and prevents steam from escaping until the pressure force overcomes the weight of the petcock. The periodic escape of the steam in this manner prevents any potentially dangerous pressure buildup and keeps the pressure inside at a constant value. Determine the mass of the petcock of a pressure cooker whose operation pressure is 100 kPa gage and has an opening cross-sectional area of 4 mm2. Assume an atmospheric pressure of 101 kPa, and draw the free-body diagram of the petcock. Answer: 40.8 g
Want to see the full answer?
Check out a sample textbook solution
Chapter 1 Solutions
THERMODYNAMICS(SI UNITS,INTL.ED)EBOOK>I
Additional Engineering Textbook Solutions
Heat and Mass Transfer: Fundamentals and Applications
Fluid Mechanics Fundamentals And Applications
Fundamentals of Aerodynamics
Applied Statics and Strength of Materials (6th Edition)
Fundamentals Of Thermodynamics
EBK FUNDAMENTALS OF THERMODYNAMICS, ENH
- (b) Hairdryer is an electromechanical device that blows ambient or hot air over damp hair to speed the evaporation of water to dry the hair. To design a hairdryer, it should contain the following basic components: a duct of constant diameter with a few layers of electric coils, a small fan to pull the air in and force it through the heating coils. If the density of air is 1.18 kg/m³ at the inlet and 0.90 kg/m³ at the exit, calculate the percent increase in the velocity of air as it flows through the dryer.arrow_forwardThe water level required in a tank to obtain velocity of 10 m/s at bottom isarrow_forwardA weighted piston (total mass of 16.8 kg), resting on stops in a cylinder (radius of 0.05 m) divides a container into two regions. 0.075 kg of saturated liquid water at 50 °C is contained beneath the piston. The rest of the cylinder and container is evacuated. Heat is applied to the water until the piston is lifted off of the stops and moved a distance (y) of 0.6 m. As the piston reaches the mouth of the cylinder, the hermetic seal is broken and the cylinder contents can now expand to fill the remainder of the evacuated space. At this instant, no additional heat is added to or removed from the system. When the system fully equilibrates, its absolute pressure is measured to be 0.01 MPa. vacuum y (cc) BY-NC-SA Niel Crews. 2013 a) What is the state of the water when the piston begins to rise off of the stops? compressed liquid b) What is the water temperature when the piston reaches the mouth of the cylinder? 61.02432 °C c) What is the final volume of the water (after the seal is broken)?…arrow_forward
- 2. If 37 kJ of heat is applied to a non-flow system while 54 kJ of work is done by thesystem, determine the magnitude of the change in the internal energy and explain thedirection of this change. Explain the relationships between system constants for a perfect gasarrow_forwardWater is to be pumped from the bottom of a well 20 ft deep with a specific heat of 1 BTU/lbm F is pumped using a 1-hp jet pump. Heat is lost from the whole system at the constant rate of 500 BTU/min. What is the temperature (℉) of the water as it enters the storage tank assuming that the well water is at 35℉?arrow_forwardA fluid, contained in a horizontal cylinder fitted with a frictionless leak proof piston, is continuously agitated by means of a stirrer passing through the cylinder cover. The cylinder diameter is 0.40 m. During the stirring process lasting 10 minutes, the piston slowly moves out a distance of 0.485 m against the atmosphere. The net work done by the fluid during the process is 2 kJ. The speed of the electric motor drivingthe stirrer is 840 rpm. Determine the torque in the shaft and the poweroutput of the motor.arrow_forward
- Water having a mass of 1800 lb is cooled from 70ºF to a final temperature of 32ºF. Calculate the amount of heat removed from water.arrow_forwardQuestion # 3 - Water of 2 kg fills a part of the chamber with P = 300 kPa. The other part of the chamber is separated by a partition from the water part, and it is empty and evacuated. The total chamber volume is 1.5 m³; the part filled with water has 1/3 of the total volume. If the partition between the two parts is removed and water expands inside the entire chamber, determine the following: a) The water temperature before removing the partition b) The pressure after removing the partition if T = 3 °C c) For part (b), find the specific enthalpy waterarrow_forwardIn a heat engine, water vapor flows into a turbine and then it enters a condenser for rejecting thermal energy. The system requires maintaining the gauge pressure of the steam at 1400 kPa at the turbine inlet and at a vacuum of 71 cmHg in the condenser. Having that the barometric pressure is measured as 77.2 cmHg, calculate the absolute pressures (in kPa) of the steam at the turbine inlet and in the condenser. (Density of mercury is 13600 kg/m3)arrow_forward
- Q.6.B. Four g of air is contained in the cylinder. The air is heated until the piston raises 30 mm. The spring just touches the piston initially. Calculate (a) the temperature when the piston leaves the stops and (b) the work done by the air on the piston. 400 kN/m Frictionjess piston 300 kg Air 150 mm 200 mm ITarrow_forwardDESCRIBE THE ENERGY ANALYSIS OF STEADY-FLOW SYSTEMSarrow_forwardIn the piston-cylinder system shown in the figure, in the first state, the piston is located on the stoppers and the volume below it is 0.15 m3. There is 2 kg of water at a temperature of 40 oC in the cylinder. The cross-sectional area of the piston is 0.075 m2 and its mass is 35 kg. Atmospheric pressure is 100 kPa and gravitational acceleration is 9.76 m/s2. Then, heat is given to the water until it becomes dry saturated water vapor. Calculate the temperature of the water when the piston starts to move over the stoppers and the work done during the whole process.arrow_forward
 Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press
Elements Of ElectromagneticsMechanical EngineeringISBN:9780190698614Author:Sadiku, Matthew N. O.Publisher:Oxford University Press Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON
Mechanics of Materials (10th Edition)Mechanical EngineeringISBN:9780134319650Author:Russell C. HibbelerPublisher:PEARSON Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education
Thermodynamics: An Engineering ApproachMechanical EngineeringISBN:9781259822674Author:Yunus A. Cengel Dr., Michael A. BolesPublisher:McGraw-Hill Education Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY
Control Systems EngineeringMechanical EngineeringISBN:9781118170519Author:Norman S. NisePublisher:WILEY Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning
Mechanics of Materials (MindTap Course List)Mechanical EngineeringISBN:9781337093347Author:Barry J. Goodno, James M. GerePublisher:Cengage Learning Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY
Engineering Mechanics: StaticsMechanical EngineeringISBN:9781118807330Author:James L. Meriam, L. G. Kraige, J. N. BoltonPublisher:WILEY





