
Interpretation:
The compound(s) that are more soluble in water has to be predicted from the following options –
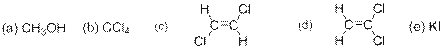
Concept Introduction:
Solubility of a substance depends on various factors like – chemical structure of the substance, temperature, pH etc,
Like dissolves like is a Universal rule which states that polar compounds dissolve more in polar solvents and non-polar compounds dissolve more in non-polar compounds.
Dipole moment is the measure of polarity in polar covalent compounds.
Answer to Problem 12.1KSP
Options (a), (d) and (e) are correct answers.
Explanation of Solution
Reason for correct answers:
The compound given in the options (a), (d) and (c) are polar compounds and so they dissolve in water which is a polar solvent.
The compound given in option (a) is methanol. It is a polar covalent compound as the
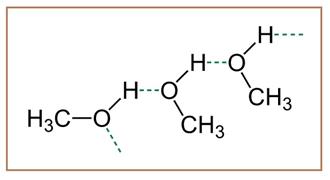
Figure 1
The compound given in option (d) is cis-1,2-dichloroethene. The
The compound given in option (e) is Potassium iodide and it is an ionic compound made of
Reasons for Incorrect answers:
The compounds given in options (
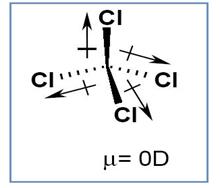
Figure 2
Though the
Therefore polarity of the compound gets cancelled.
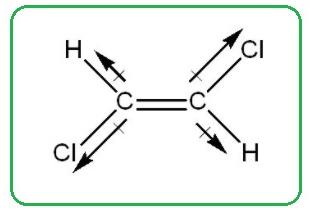
Figure 3
The compound(s) that are more soluble in water has been predicted from the given options.
Want to see more full solutions like this?
Chapter 12 Solutions
Chemistry: Atoms First
- The substance with the lowest known boiling point (4 K) is helium, an atomic element that has two electrons. Hydrogen isa diatomic molecule and also has two electrons, but its boiling point is significantly higher, at 20.28 K.(a) What is the dominant intermolecular force between a pair of helium atoms and a pair of H2 molecules?(b) Why do you think H2 has a higher boiling point?arrow_forwardWhich of the following is FALSE about the water molecule? A) The H atoms bear the partial positive charge. Intermolecular forces among water molecules are the least extensive when it is in liquid B form. (c) The atoms comprising the water molecule are covalently bonded with one another. D Its molecular geometry is not linear.arrow_forwardWhich of the following compounds would most likely have the highest melting point? NaCl NaF CaCl2 Li₂Oarrow_forward
- What kind of intermolecular forces act between a hydrogen fluoride molecule and an ammonia molecule? Note: If there is more than one type of intermolecular force that acts, be surearrow_forwardWhich is neither polar nor capable of hydrogen bonding? Explain with example?arrow_forwardwhich if the following is true about hydrogen bonding?arrow_forward
- Which of the following is FALSE about the water molecule? A The H atoms bear the partial positive charge. B The atoms comprising the water molecule are covalently bonded with one another. Its molecular geometry is not linear. D Intermolecular forces among water molecules are the least extensive when it is in liquid form.arrow_forwardWhat kind of intermolecular forces act between a helium atom and a xenon aTom? Note: if there is more than one type of intermolecular force that acts, be sure to list them all, with a comma between the name of each force. options: Dispersion, Dipole, Hydrogen Bonding, & Ion Dipolearrow_forwardWhat types of intermolecular attractive forces can be found between the molecules with the following structure? Which are the strongest one?arrow_forward
- (a) Which type of intermolecular attractive force operates between all molecules? (b) Which type ofintermolecular force operates only between polar molecules? (c) Which type of intermolecular force operatesonly between the hydrogen atom and a nearby small electronegative atom?arrow_forward= O STATES OF MATTER Identifying the important intermolecular forces in pure... Decide which intermolecular forces act between the molecules of each compound in the table below. compound F₂ fluorine nitrogen trichloride CH₂F fluoromethane hydrogen fluoride intermolecular forces (check all that apply) dispersion dipole hydrogen-bonding Uarrow_forwardFor each molecule in the table below, determine: . Could the molecule form a hydrogen bond with water? (Yes or No) What is the strongest type of intermolecular bond that the molecule could form with itself? (Dispersion, Dipole-Dipole, or Hydrogen) • Another way to describe this: If you had 2 of the molecules next to each other, what is the strongest type of intermolecular bond that could form between them? Molecule Structure: HO HO HO H-N Can this molecule form a hydrogen bond with water? ✓ [Select] Yes No [Select] [Select] [Select] [Select] [Select] What is the strongest type of intermolecular bond that this molecule could form with itself? [Select] [Select] [Select] [Select] [Select] [Select]arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning
Introductory Chemistry: An Active Learning Approa...ChemistryISBN:9781305079250Author:Mark S. Cracolice, Ed PetersPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning



