
Chemistry In Context
9th Edition
ISBN: 9781259638145
Author: Fahlman, Bradley D., Purvis-roberts, Kathleen, Kirk, John S., Bentley, Anne K., Daubenmire, Patrick L., ELLIS, Jamie P., Mury, Michael T., American Chemical Society
Publisher: Mcgraw-hill Education,
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 12.9, Problem 12.26YT
Skill Building Ester Formation
Draw structural formulas for the esters that form when these alcohol and acid pairs react.
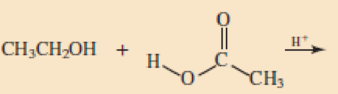
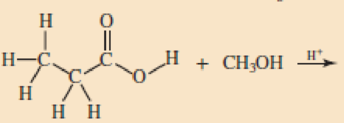
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
E Knowledge Check
Question 5
Hannal
For each compound in the table below, decide whether there would be any hydrogen-bonding force between molecules of the compound, or between molecules
of the compound and molecules of water.
compound
hydrogen-bonding force
Between
molecules of the
formula or Lewis
Between molecules of
name
the compound and
molecules of water?
structure
compound?
hydrogen bromide
O yes
H Br
O yes
O no
O no
hypobromous acid
H BrO
O yes
O yes
O no
O no
H :ċi:
N,N-
O yes
O yes
H - C-N- Ci:
dichloromethylamine
O no
O no
H.
I Don't Know
Submit
MacBook Air
DD
DI
F10
F9
F8
888
F7
F6
F5
F4
F3
F2
Draw the condensed structure of 6,8-dimethyl-4-propyl-1-nonanol.
Explanation
Check
Click anywhere to draw the first atom
of your structure.
Q Search
© 2023 McGraw Hill LL
Initial Knowledge Check
Decide whether each molecule or polyatomic ion is polar or nonpolar.
If the molecule or polyatomic ion is polar, write the chemical symbol of the atom closest to the negative side. For example, if the molecule were HCI
decided the hydrogen atom was closest to the negative side of the molecule, you'd enter "H" in the last column of the table.
molecule or
polar or
nonpolar?
atom closest to
polyatomic ion
negative side
polar
CH,F
nonpolar
polar
Br,
nonpolar
polar
H,S
O nonpolar
I Don't Know
2021 McGraw Hill LLC, ARights Reserve
Chapter 12 Solutions
Chemistry In Context
Ch. 12.1 - Skill Building Finding Equilibrium Glucose and...Ch. 12.1 - Prob. 12.3YTCh. 12.1 - Prob. 12.4YTCh. 12.2 - Prob. 12.5YTCh. 12.2 - Prob. 12.6YTCh. 12.2 - A practicing scientist must Judge a potential...Ch. 12.3 - Prob. 12.8YTCh. 12.3 - Skill Building Checking on Carbon a. Examine the...Ch. 12.3 - Prob. 12.10YTCh. 12.3 - Prob. 12.11YT
Ch. 12.3 - Prob. 12.12YTCh. 12.4 - Prob. 12.13YTCh. 12.4 - Skill Building Functional Groups in Dopamine Draw...Ch. 12.4 - Prob. 12.15YTCh. 12.5 - Prob. 12.16YTCh. 12.5 - Prob. 12.17YTCh. 12.6 - Prob. 12.18YTCh. 12.6 - Prob. 12.19YTCh. 12.6 - The structures of proteins, such as the ones shown...Ch. 12.7 - Reconsider your work in past chapters. List three...Ch. 12.7 - Prob. 12.22YTCh. 12.7 - Prob. 12.23YTCh. 12.8 - Prob. 12.24YTCh. 12.8 - Prob. 12.25YTCh. 12.9 - Skill Building Ester Formation Draw structural...Ch. 12.9 - Prob. 12.27YTCh. 12.9 - You Decide Supersize My Aspirin A friend who...Ch. 12.9 - Modern methods of drug discovery involve...Ch. 12.10 - Make two lists of drugs for each of the two...Ch. 12.10 - See for yourself the shapes of drug molecules by...Ch. 12.10 - Prob. 12.33YTCh. 12.10 - Prob. 12.34YTCh. 12 - Scientific Practices Follow the Hormone Using the...Ch. 12 - The field of chemistry has many sub-disciplines....Ch. 12 - Prob. 2QCh. 12 - Prob. 4QCh. 12 - Nitrous acid (HNO2) has a Ka value of 4.0 10 4,...Ch. 12 - Use the Henderson-Hasselbalch equation and Table...Ch. 12 - Write the structural formula and line-angle...Ch. 12 - Prob. 8QCh. 12 - Prob. 9QCh. 12 - Prob. 10QCh. 12 - Prob. 11QCh. 12 - Prob. 12QCh. 12 - Estradiol is relatively insoluble in water but...Ch. 12 - Prob. 14QCh. 12 - Prob. 15QCh. 12 - Prob. 16QCh. 12 - Define and relate the two terms: hormone and...Ch. 12 - Refer to Figure 11.17. Select two examples of...Ch. 12 - Prob. 19QCh. 12 - Molecules as diverse as cholesterol, sex hormones,...Ch. 12 - Prob. 21QCh. 12 - Prob. 22QCh. 12 - Prob. 23QCh. 12 - Sulfanilamide is the simplest sulfa drug, a type...Ch. 12 - Explain why an equilibrium constant cannot tell...Ch. 12 - Use the information in Table 12.1 to redraw Figure...Ch. 12 - Draw structural formulas for each of these...Ch. 12 - In Your Turn 12.12, you were asked to draw...Ch. 12 - Prob. 29QCh. 12 - Prob. 30QCh. 12 - Prob. 31QCh. 12 - Prob. 32QCh. 12 - Prob. 34QCh. 12 - Prob. 35QCh. 12 - Prob. 36QCh. 12 - Prob. 37QCh. 12 - Prob. 38QCh. 12 - The text states that some racemic mixtures contain...Ch. 12 - Prob. 40QCh. 12 - Prob. 41QCh. 12 - Prob. 44QCh. 12 - Prob. 47QCh. 12 - Prob. 49QCh. 12 - Dorothy Crowfoot Hodgkin first determined the...Ch. 12 - Prob. 52Q
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Dinitrogen difluoride (see Example 10.5) exists as cis and trans isomers. Write structural formulas for these isomers and explain (in terms of the valence bond theory of the double bond) why they exist.arrow_forwardExercise Sketch the following simple structures using Chemdraw software 1. Lewis dot structure of methane (2D drawing) Hint: use solid bond and text tools. Hints: H 2. Chemical structure of methane (3D drawing) 3. Chemical structure of methanol (3D drawing) 4. Chemical structure of benzene 5. Chemical structure of acetone 6. Chemical structure of methyl acetate Tools →→ Template → stereocenters, Click on the tetrahedron shape H H :0: H :0: H3C CH3 WITH -H H3C OCH3 H CH Harrow_forwardModule Knowledge Check Determine the structures of the missing organic molecules in the following reaction: got I Don't Know + H₂O Submit H+ Y H+ Note: Molecules that share the same letter have the exact same structure. In the drawing area below, draw the skeletal ("line") structures of the missing organic molecules X, Y, and Z. You may draw the structures in any arrangement that you like, so long as they aren't touching. Molecule X shows up in multiple steps, but you only have to draw its structure once. Q Search Question 3 Z Click and drag to start drawing a structure. с с X 3 C* Allie V ? FEED allo Ar © 2023 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center Accessibility ?arrow_forward
- Chemistry can you do me A-G? this is organic chemarrow_forwardTime left The C=O bond in carbon dioxide is a polar bond, therefore, the overall polarity of the molecule is also polar. Select one: O True False Nexarrow_forwardWhich of the following atoms do NOT have 5 valence electrons? Select all that applyarrow_forward
- O ORGANIC FUNCTIONAL GROUPS Naming and drawing carboxylic acids Draw the skeletal ("line") structure of 6-chloro-3,5-dimethylhexanoic acid. Click and drag to start drawing a structure. C™ X A Ċ Sarrow_forwardQUESTION 21 Which of the following solvents is polar? O C6H4(CH3)2 O C2H5OH O C5H12 C3H8arrow_forwardTwo molecules, each with the general formula AX3, have different dipole moments. Molecule Y has a dipole moment of zero, whereas molecule Z has a nonzero dipole moment. From this information, what can you say about the geometries of Y and Z?arrow_forward
- EXERCISE Resonance Structures I Clear O S Below is one of the Lewis structures of SO3, which has two other resonance structures that do not violate the octet rule. :0 s=ö :ö: Complete one of the other resonance structures by dragging bonds and electron lone pairs to their appropriate positions. Then click Check.arrow_forwardDashboard O PRINCIPLES OF ORGANIC CHEMISTRY Identifying hydrophobic and hydrophilic parts of an organic... ALEKS Explanation Check Highlight the atoms or groups that are in hydrophobic portions of this molecule in blue, and the atoms or groups that are in hydrophilic portions of this molecule in red. CH3-CH₂-0-CH₂-CH3 A ALEKS-Kimberly Cedillos - Learn MacBook Pro X G Write down the oxidation state 3 OD 1/3 Kimberly © 2023 McGraw Hill LLC. All Rights Reserved. Terms of Use | Privacy Center | Accessibility ol E Ar 0 Marrow_forwardQUESTION 16 Which describes the functional group(s) of the following molecule? :OH CH;CHCHCH; CH3 O aldehyde O ketone O neither aldehyde nor ketone O both aldehyde and ketone QUESTION 17 Click Save and Submit to save and submit. Click Save All Answers to save all ansuwers. MacBook Proarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER
Living By Chemistry: First Edition TextbookChemistryISBN:9781559539418Author:Angelica StacyPublisher:MAC HIGHER

General Chemistry - Standalone book (MindTap Cour...
Chemistry
ISBN:9781305580343
Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; Darrell
Publisher:Cengage Learning

Living By Chemistry: First Edition Textbook
Chemistry
ISBN:9781559539418
Author:Angelica Stacy
Publisher:MAC HIGHER
Quantum Mechanics - Part 1: Crash Course Physics #43; Author: CrashCourse;https://www.youtube.com/watch?v=7kb1VT0J3DE;License: Standard YouTube License, CC-BY