
Consider the interconversion of A molecules (red spheres) and B molecules (blue spheres) according to the reaction A ⇌ B. Each of the following series of pictures represents a separate experiment in which time increases from left to right:
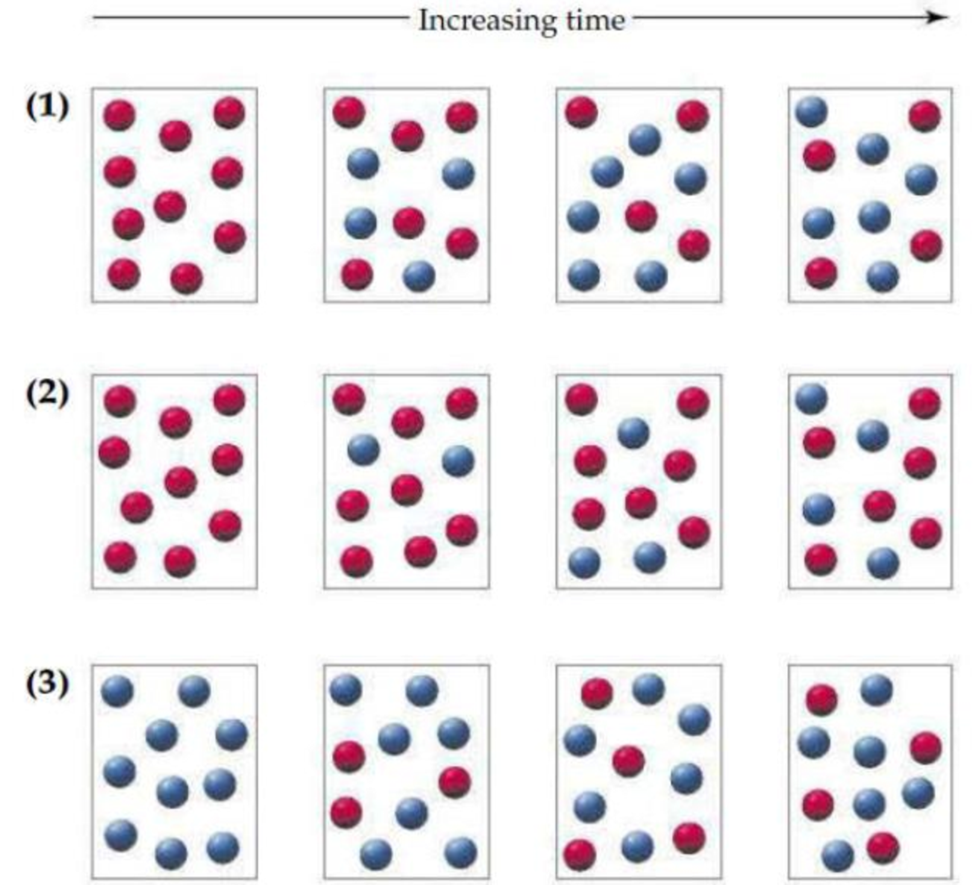
- (a) Which of the experiments has resulted in an equilibrium state?
- (b) What is the value of the equilibrium constant Kc for the reaction A ⇌ B?
- (c) Explain why you calculate Kc without knowing the volume of the reaction vessel.
(a)
Interpretation:
From the given experiments, the one resulted in equilibrium state has to be identified.
Concept introduction:
Chemical equilibrium:
It is defined as a state in which concentrations of reactants and products are same over a period of time. An equilibrium mixture is a mixture in which reactants and products are in equilibrium.
Answer to Problem 13.26CP
Experiment one and three are in equilibrium state.
Explanation of Solution
Reason correct options:
Given,

Figure 1
In experiment one and three, the number of red spheres A and blue spheres B are same over a period of time. In both experiments third and fourth box have same number of A and B. Hence, experiment one and three are in equilibrium state.
Reason incorrect options:
In experiment two, the number of A and B are not same over a period of time. Hence, experiment two is not in equilibrium.
(b)
Interpretation:
The equilibrium constant for given reaction has to be calculated.
Concept introduction:
Equilibrium constant
Equilibrium constant
Consider the reaction where A reacts to give B.
On rearranging,
Where,
Answer to Problem 13.26CP
The value of equilibrium constant is
Explanation of Solution
Given,
The equilibrium constant is calculated by substituting concentrations of reactants and products in equilibrium constant expression.
Therefore, the value of equilibrium constant is
(c)
Interpretation:
The reason for calculating equilibrium constant without knowing volume of the reaction vessel has to be explained.
Concept introduction:
Equilibrium constant
Equilibrium constant
Consider the reaction where A reacts to give B.
On rearranging,
Where,
Explanation of Solution
At equilibrium the number of molecules on reactant and product side remains same and volume gets cancelled while substituting in equilibrium constant expression. Hence, equilibrium constant
Want to see more full solutions like this?
Chapter 13 Solutions
General Chemistry: Atoms First
- Write equilibrium-constant expressions Kp for each of the following reactions: a H2(g)+Br2(g)2HBr(g) b CS2(g)+4H2(g)CH4(g)+2H2S(g) c 4HCl(g)+O2(g)2H2O(g)+2Cl2(g) d CO(g)+2H2(g)CH3OH(g)arrow_forwardCyclohexane, C6H12, a hydrocarbon, can isomerize or change into methylcyclopentane, a compound of the same formula (C5H9CH3) but with a different molecular structure. sssss The equilibrium constant has been estimated to be 0.12 at 25 C. If you had originally placed 0.045 mol of cyclohexane in a 2.8-L flask, what would be the concentrations of cyclohexane and methylcyclopentane when equilibrium is established?arrow_forwardFor the reaction N2(g)+3H2(g)2NH3(g) show that Kc = Kp(RT)2 Do not use the formula Kp = Kc(RT)5n given in the text. Start from the fact that Pi = [i]RT, where Pi is the partial pressure of substance i and [i] is its molar concentration. Substitute into Kc.arrow_forward
- 12.103 Methanol, CH3OH, can be produced by the reaction of CO with H2, with the liberation of heat. All species in the reaction are gaseous. What effect will each of the following have on the equilibrium concentration of CO? (a) Pressure is increased, (b) volume of the reaction container is decreased, (c) heat is added, (d) the concentration of CO is increased, (e) some methanol is removed from the container, and (f) H2 is added.arrow_forwardSuppose a reaction has the equilibrium constant K = 1.3 108. What does the magnitude of this constant tell you about the relative concentrations of products and reactants that will be present once equilibrium is reached? Is this reaction likely to be a good source of the products?arrow_forwardThe equilibrium constant Kc for the synthesis of methanol, CH3OH. CO(g)+2H2(g)CH3OH(g) is 4.3 at 250C and 1.8 at 275C. Is this reaction endothermic or exothermic?arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
Introductory Chemistry: A FoundationChemistryISBN:9781337399425Author:Steven S. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning





