
Concept explainers
(a)
Interpretation:
Whether the given molecule is
Concept introduction:
The molecules, to be aromatic, must obey Hückel’s rule of
Answer to Problem 14.41P
The given molecule is nonaromatic due to lack of planarity.
Explanation of Solution
The given structure is:
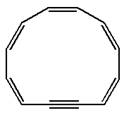
The molecule has total
The molecule is determined as nonaromatic based on the structure planarity.
(b)
Interpretation:
Whether the given molecule is aromatic, antiaromatic, or nonaromatic, is to be determined.
Concept introduction:
The molecules, to be an aromatic, must obey Hückel’s rule of
Answer to Problem 14.41P
The given molecule is nonaromatic due to lack of planarity.
Explanation of Solution
The given structure is:
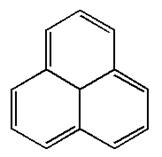
The molecule has total
The molecule is determined as nonaromatic based on the structure planarity.
(c)
Interpretation:
Whether the given molecule is aromatic, antiaromatic, or nonaromatic, is to be determined.
Concept introduction:
The molecules, to be an aromatic, must obey Hückel’s rule of
Answer to Problem 14.41P
The given molecule is aromatic as it is planar, has cyclic conjugation, and obeys Hückel’s rule.
Explanation of Solution
The given structure is:
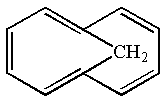
The given molecule is planar and has total
The molecule is determined as aromatic based on the Hückel’s rule.
(d)
Interpretation:
Whether the given molecule is aromatic, antiaromatic, or nonaromatic, is to be determined.
Concept introduction:
The molecules, to be an aromatic, must obey Hückel’s rule of
Answer to Problem 14.41P
The given molecule is nonaromatic due non cyclic conjugated system.
Explanation of Solution
The given structure is:
![]()
The molecule has a conjugated system of total
The molecule is determined as nonaromatic based on the non cyclic structure which lacks cyclic conjugation.
(e)
Interpretation:
Whether the given molecule is aromatic, antiaromatic, or nonaromatic, is to be determined.
Concept introduction:
The molecules, to be an aromatic, must obey Hückel’s rule of
Answer to Problem 14.41P
The given molecule is aromatic as it is planar, has cyclic conjugation, and obeys Hückel’s rule.
Explanation of Solution
The given structure is:
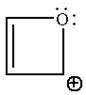
The molecule is planar and has fully cyclic conjugated system of total
The molecule is determined as aromatic based on planarity, fully conjugated system, and having Hückel’s rule of
(f)
Interpretation:
Whether the given molecule is aromatic, antiaromatic, or nonaromatic, is to be determined.
Concept introduction:
The molecules, to be an aromatic, must obey Hückel’s rule of
Answer to Problem 14.41P
The given molecule is nonaromatic as it is not conjugated and does not obey Hückel’s rule.
Explanation of Solution
The given structure is:
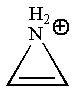
The molecule has only
The molecule is determined as nonaromatic based on the non-conjugated system of
(g)
Interpretation:
Whether the given molecule is aromatic, antiaromatic, or nonaromatic, is to be determined.
Concept introduction:
The molecules, to be an aromatic, must obey Hückel’s rule of
Answer to Problem 14.41P
The given molecule is nonaromatic as it has no cyclic conjugated system.
Explanation of Solution
The given structure is:
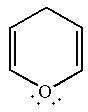
One of the lone pair on the oxygen atom participates in the resonance, thus, the molecule has
The molecule is determined as nonaromatic based on non-conjugated system of
Want to see more full solutions like this?
Chapter 14 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- 6. Determine the total number of electrons in each of the following structures. Also, circle the term that best describes the electronic configuration of each structure, aromatic, antiaromatic, or nonaromatic. total number of pi electrons aromatic antiaromatic nonaromatic HB BH HN. NH aromatic antiaromatic nonaromatic aromatic antiaromatic nonaromatic aromatic antiaromatic nonaromatic aromatic antiaromatic nonaromaticarrow_forwardDetermine if the following molecules are aromatic, non-aromatic, or anti-aromatic.arrow_forwardWhich structure is not aromatic? Please explain.arrow_forward
- which molecules should be aromaticarrow_forwardCurved arrows are used to illustrate the flow of electrons. Using the provided resonance structures, draw the curved electron-pushing arrows to show the interconversion between resonance hybrid contributors. Be sure to account for all bond-breaking and bond-making steps.arrow_forwardDetermine the molecules below are aromatic. If not, explain why.arrow_forward
- Using the curved arrow formalism, draw two major resonance contributors to the structure of the following molecule. Circle that structure that contributes most to the overall structure.arrow_forwarddraw all the resonance structures for each of the above species. be sure to include the curved arrows that indicate which pair of electrons are shifted in going from one resonance structures to the next. draw resonance hybrid of each species.arrow_forwardIn the first blank, label the molecule as either aromatic, antiaromatic, or nonaromatic. In the second, provide the number of pi electrons that would be used. Place a zero in the second blank if non aromatic.arrow_forward
- Determine whether the following molecules are aromatic (A), antiaromatic (AA),or not aromatic (N).arrow_forwardCurved arrows are used to illustrate the flow of electrons. Using the provided resonance structures, draw the curved electron- pushing arrows to show the interconversion between resonance hybrid contributors. Be sure to account for all bond- breaking and bond-making stepsarrow_forwardHow many pi electrons are in the molecule above? [Select ] Assuming that the molecule is flat, what is the aromaticity of the molecule above? SelectIarrow_forward
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
