
The color change accompanying the reaction of phenolphthalein with strong base is illustrated below. The change in concentration of the dye can be followed by spectrophotometry (Section 4.9), and some data collected by that approach are given below. The initial concentrations were [phenolphthalein] = 0.0050 mol/L and [OH‒] = 0.61 mol/L. (Data are taken from review materials for kinetics at chemed.chem.purdue.edu.) (For more details on this reaction see L Nicholson, Journal of Chemical Education, Vol. 66, p. 725, 1989.)

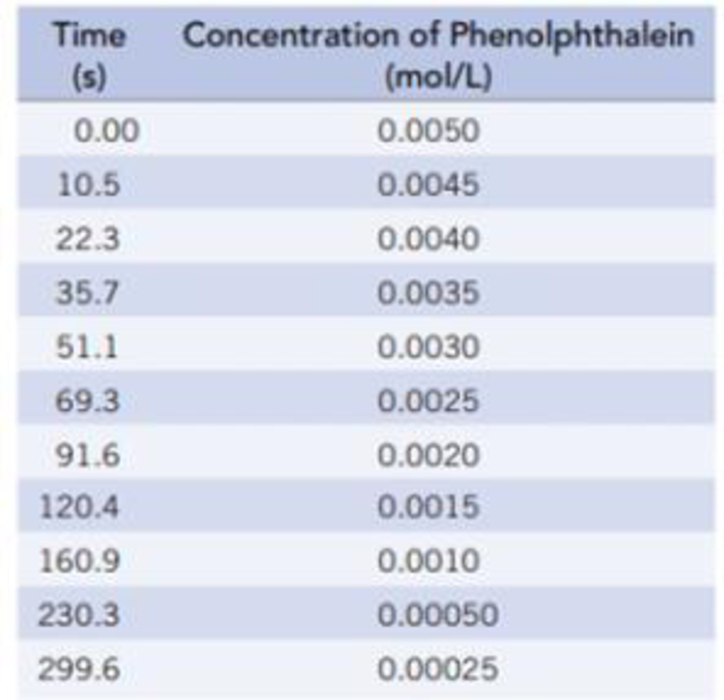
(a) Plot the data above as [phenolphthalein] versus time, and determine the average rate from t = 0 to t = 15 seconds and from t = 100 seconds to t = 125 seconds. Does the rate change? If so, why?
(b) Use a graphical method to determine the order of the reaction with respect to phenolphthalein. Write the rate law, and determine the rate constant.
(c) What is the half-life for the reaction?
Trending nowThis is a popular solution!

Chapter 14 Solutions
Chemistry & Chemical Reactivity
- 11.93 On a particular day, the ozone level in Milwaukee exceeded the EPAs 1-hour standard of 0.12 ppin by 10 ppb. How many ozone molecules would be present in 1 liter of air at the detection site?arrow_forwardWhat is the activation energy of a reaction which takes place at 385 K, where the rate constant of the reaction is 3.45 x 10-4 s-1 and A = 4.00 x 1013 ? (Please provide your answer to 3 significant figures.) Accepted format for scientific notation: Enter 99.9 as 9.99*10^1.arrow_forwardRoute 1:RME = 82.92%MI= 1.34 Route 2RME = 93.33%MI = 1.57 Route 3RME = 57.34%MI = 1.89 Compare the value of Reaction Mass Efficiency (RME) and Mass Intensity (MI) generally, and choose which metrics is more significant in green measurement. Justify your answer.arrow_forward
- но HO HO HO 0 + ½ O, → + H,0 H H но OH ascorbic acid dehydroascorbic acid Vitamin C is oxidized slowly to dehydroascorbic acid by the oxygen in air. It is catalyzed by ions such as Cu*2 and Fe*3. The reaction can be followed by measuring the ultraviolet absorbance at 243 nm. Time (hours) Absorbance (A) 1/A In A - In A 0.75 1.3 -0.29 0.29 1 0.38 2.6 -0.97 0.97 2 0.19 5.3 - 1.7 1.7 3 0.095 11 - 2.4 2.4 29. What is the hybridization of carbon 1 (far left) and carbon 2 (middle) in this hydrocarbon: CH3CH=CH2? (A) sp³, sp (B) sp?, sp? (C) sp³, sp? (D) sp, sp?arrow_forwardA student proposed the following mechanism for the gas phase reaction of fluorine with chlorine dioxide. step 1 fast: 2 CiO,=Cl,04 step 2 slow: Cl,0, + F2→2 FCIO, (1) What is the equation for the overall reaction? Use the smallest integer coefficients possible. If a box is not needed, leave it blank. (2) Enter the formula of any species that acts as a reaction intermediate? If none leave box blank: (3) Complete the rate law for the overall reaction that is consistent with this mechanism. Use the form k[A]"|B]".. , where 'l' is understood (so don't write it if it's a '1') for m, n etc. Rate =arrow_forwardConsider the reaction: H2 (g) +I2 (g) → 2 HI (g) A chemist performed an experiment and monitored the concentration of I2 during the course of the reaction. The red line in the graph below represents the results obtained. Which line in the plot would best represent how the concentration of HI changes during the course of the reaction? Time (s) Select an answer and submit. For keyboard navigation, use the up/down arrow keys to select an answer. a a b b d d е е Concentration (M)arrow_forward
- 11. The lifetime of the VOC called glyoxal in the atmosphere is controlled by its reaction with the OH radical and photolysis. The rate constants for these reactions have been measured as ko3.7x 10* cm molecule" s'and ka=12 min". What would the overall lifetime of glyoxal under these conditions with an [OH]=1.6x 10 molecule cm.arrow_forward5.) For the reactionBrO3–+ 5 Br–+ 6 H+↓ 3 Br2 + 3 H2O The rate of disappearance of BrO3–(– Δ[BrO3–]/ Δt) is 1.5 × 10–2 mol/L·s (M ·s–1) at a particular time. What isthe rate of disappearance of Br– (– Δ[Br–]/ Δt) at the same instant?arrow_forward11. The lifetime of the VOC called glyoxal in the atmosphere is controlled by its reaction with the OH radical and photolysis. The rate constants for these reactions have been measured as kos=3.7x 10* cm molecules'and ka =12 min". What would the overall lifetime of glyoxal under these conditions with an [OH] =1.6x 10 molecule cm.arrow_forward
- 2AB3 = 2A + 3B2 The half-life of the above reaction is 9 hours. Using 12 moles of AB3, calculate the moles and volume of B2 formed after 9 hours under normal conditions (half-life is independent of the amount of starting material. (physical chemistry question)arrow_forward(#28) Reaction Rates and time. A decomposition reaction in the first order has a rate constant of 1.26 x 10-4/sec. If the initial concentration of the reactant is 0.644 M. how much time will it take before 20% of the reactant has decomposed? 28a) In blank #1, type-in the working formula to solve for the time required. Enter the algebraic formula as a one-line equation and without spaces in between characters. Use lowercase for all fonts. 28b) In blank #2, type in your answer for the time it will take for 20% f the reactant to decompose. The answer is in seconds but do not include the unit. Enter your answer as a whole number with four significant figures.arrow_forwardIn a study of the decomposition of hydrogen iodide on a gold surface at 150 °C HI(g)% H2(g) + ½ I½(g) the concentration of HI was followed as a function of time. It was found that a graph of [HI] versus time in seconds gave a straight line with a slope of -1.91×10-4 Ms and a y-intercept of 0.235 M. Based on this plot, the reaction is reaction is order in HI and the rate constant for the Ms!arrow_forward
 Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning
Principles of Modern ChemistryChemistryISBN:9781305079113Author:David W. Oxtoby, H. Pat Gillis, Laurie J. ButlerPublisher:Cengage Learning Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co
Chemistry for Engineering StudentsChemistryISBN:9781337398909Author:Lawrence S. Brown, Tom HolmePublisher:Cengage LearningChemistry: Matter and ChangeChemistryISBN:9780078746376Author:Dinah Zike, Laurel Dingrando, Nicholas Hainen, Cheryl WistromPublisher:Glencoe/McGraw-Hill School Pub Co


