
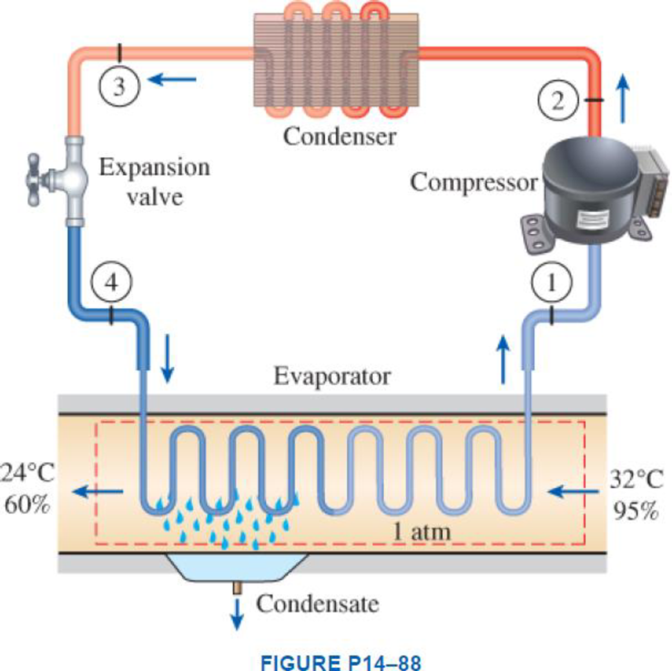
Atmospheric air at 1 atm, 32°C, and 95 percent relative humidity is cooled to 24°C and 60 percent relative humidity. A simple ideal vapor-compression refrigeration system using refrigerant-134a as the working fluid is used to provide the cooling required. It operates its evaporator at 4°C and its condenser at a saturation temperature of 39.4°C. The condenser rejects its heat to the atmospheric air. Calculate the exergy destruction, in kJ, in the total system per 1000 m3 of dry air processed.
The exergy destruction in the total system per
Answer to Problem 88P
The exergy destruction in the total system per
Explanation of Solution
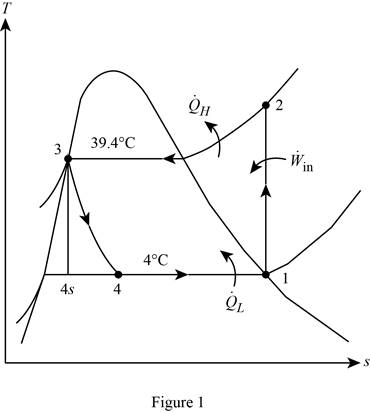
Show the T-s diagram for the simple ideal vapour-compression refrigeration system.
Express the mass of air.
Here, volume at state 1 is
Express the water mass balance and energy balance equations to the combined cooling and dehumidification section.
For water mass balance:
For energy balance:
Write the energy balance equation using steady-flow equation.
Here, the rate of total energy entering the system is
Substitute
Here, the specific enthalpy at the state 1 and 2 are
Write the formula for mass flow rate of refrigerant-134a.
Write the formula for amount of heat rejected from the condenser.
Calculate the exergy destruction in the components of the refrigeration cycle.
For the process 1-2,
Here, the process 1-2 is an isentropic.
For the process 2-3,
For the process 3-4,
Write the entropy change of water vapour in the air stream.
Write the entropy of water leaving the cooling section.
Determine the partial pressure of water vapour at state 1 for air steam.
Determine the partial pressure of dry air at state 1 for air steam.
Here, the pressure at the state 1 is
Determine the partial pressure of water vapour at state 2 for air steam.
Determine the partial pressure of dry air at state 2 for air steam.
Here, the pressure at the state 2 is
Write the formula for entropy change of dry air.
Write the formula for entropy change R-134a in the evaporator.
Write the formula for an entropy balance on the evaporator.
Write the formula for an exergy destruction in the evaporator.
Write the formula for the total exergy destruction.
Conclusion:
Refer Figure A-31, “psychometric chart at
Refer Table A-4, “saturated water-temperature table”, and write the specific enthalpy of condensate water at temperature of
Here, entropy of saturation liquid at temperature of
Write the formula of interpolation method of two variables.
Here, the variables denote by x and y is temperature and specific enthalpy of condensate water at state 2 respectively.
Show the specific enthalpy of condensate water at corresponding to temperature as in Table (1).
|
Temperature |
Specific enthalpy of condensate water |
| 25 | 104.83 |
| 28 | |
| 30 | 125.74 |
Substitute
Substitute
Substitute
Substitute 1105 kg for
Substitute 1105 kg for
From the Table A-11 “Saturated Refrigerant-134a-Pressure Table”, obtain the value of the specific enthalpy and entropy at state 1 of
Refer Table A-13 “Saturated Refrigerant-134a-Pressure Table”, and write the specific enthalpy at state 2 in 1 MPa of pressure and entropy of
From the Table A-12 “Saturated Refrigerant-134a-Pressure Table”, obtain the value of the specific enthalpy and entropy at state 3 of
Here, the specific enthalpy at the state 4 and 3 are equal in the throttling.
Calculate the value of
From the Table A-11 “Saturated Refrigerant-134a-Pressure Table”, obtain the value of the specific enthalpy and entropy of saturated liquid and change upon vaporization at state 4 of
Substitute
Calculate the value of specific entropy of the state 4.
Substitute
Substitute
Substitute
Substitute
Substitute
Refer Table A-4 “Saturated water-temperature Table”, and write the specific entropy at state 1 at
Refer Table A-4 “Saturated water-temperature Table”, and write the specific entropy at state 1 at
Substitute
Substitute
Substitute
Substitute
Substitute
Substitute
Substitute
Substitute
Substitute
Substitute
Substitute 0 for
Thus, the exergy destruction in the total system per
Want to see more full solutions like this?
Chapter 14 Solutions
Thermodynamics: An Engineering Approach
- What are higher-efficiency refrigerant blends that can be used as long-term replacements for R-22 in residential and light-commercial air-conditioning applications?arrow_forwardIndonesia Inggris A single effect evaporator was used to concentrate 8000 kg / hr of tomato juice from 5% total solids to 30% total solids. The juice enters the evaporator at 25 ° C. The evaporator is operated on Steam (85% quality) at 143.27 kPa. The vacuum in the evaporator allows the juice to boil at 75 ° C. The specific heat of the dilute material is 4.1 kJ / (kg ° C) and the concentrate product is 3.1 kJ / (kg ° C). Count it (a) The rate of demand for steam? b. Steam economy if condensate temperature is released at 75 ° C.?arrow_forwardIn an uncomfortable summer day, the air is at 87OF and 80% relative humidity. A laboratory air conditioner is to deliver 1.00 x 103ft3/min of air at 55OF in order to maintain the interior air at an average temperature of 75OF and relative humidity of 40%.a. If the vent switch on the air conditioner is turned to the open position, outside air enters the unit as shown belown the air conditioner, the air is cooled to a temperature low enough to condense the necessary amount of water and reheated to 55OF, at which point it has the same absolute humidity as the room air. Determine the rate (lb/min) at which water is condensed, the temperature to which the air must be cooled to condense water at this rate, and the net tons of cooling required (Q), where 1 ton of cooling = -12,000 Btu/h. Assume A = 87 οF, B = 80 %, C = 55 οF.arrow_forward
- 1. A refrigeration system using R-22 has a capacity of 320 kW of refrigeration. The evaporating temperature is -10°C and the condensing temperature is 40°C. Calculate the fraction of vapor in the mixture before the evaporator. Properties of R-22: At -10°C; hg = 401.60 kJ/kg, hf = 188.43 kJ/kg At 40°C; hf 249.69 kJ/kgarrow_forwardConsider the vapor-compression refrigeration cycle shown below with tetrafluoroethane as the refrigerant. If the evaporation temperature is -12°C, determine the coefficients of performance for condensation temperatures of 16°C, 28°C, and 40°C. Use the table containing the properties of saturated 1,1,1,2- tetrafluoroethane (R134A) and the PH diagram for tetrafluoroethane (HFC-134a). T S Vapor-compression refrigeration cycle. 4 Throttle valve Condenser Compressor Evaporator Assume isentropic compression of the vapor. The coefficient of performance for a condensation temperature of 16°C is The coefficient of performance for a condensation temperature of 28°C is 3 The coefficient of performance for a condensation temperature of 40°C is 2arrow_forwardAdditional Instructions: Define each enthalpy obtained from tables and chart and draw the p-h diagram labeling the respective evaporating and condensing pressures and temperatures in each problem. (2) A standard vapor compression system produces 20 tons of refrigeration using R-12 as a refrigerant while operating between a condenser temperature of 41.6 C and an evaporator temperature of -25°C. Determine (a) the refrigerating effect in kJ/kg, (b) the circulating rate in kg/s, (c) the power supplied, (d) the COP, (e) the heat rejected in kW, and (f the volume flow rate in L/s. Ans. (a) 100.26 kJ/kg, (b) 0.7022 kg/s, (c) 25.69 kW, (d) 2.74, (e) 96.09 kW, (f) 92.18 L/sarrow_forward
- A refrigeration system using refrigerant 12 is to have a refrigerating capacity of 80 kW. The cycle is a standard vapor-compression cycle in which the evaporating temperature is -15°C and the condensing temperature 30°C. At the entrance to the evaporator, what is the fraction of vapor in the mixture expressed on a mass basis? 0.97 0.29 0.92 0.27arrow_forwardA space to be conditioned has a sensible heat load of 80 kw and a latent heat load of 20 kw and is to be maintained at 20°C and 50% relative humidity. Outside air is at 35°C and 60% RH. The conditioned air will enter the space at 15°C. If 40% of the supply air is fresh and the rest is recirculated, the refrigeration capacity of the system is nearest to what value? 430 kw 455 kw 428 kw Ⓒ333 kwarrow_forwardAn ammonia system with a 158-kW evaporator operating at -1°C and a 35-kW evaporator operatingat -40°C uses flash gas removal and intercooling. The condensing temperature is 32°C. Thearrangement is as shown. How many kilograms per minute of ammonia are compressed by the high-pressure compressor?arrow_forward
- Can you also show the plot on the chart provided? The mass flow rate of the refrigerant in a Single Stage Vapor Compression (SSVC) refrigeration system is 0.015 kg/s and the evaporating and condensing temperatures shall be -20°C and 50°C, respectively. Determine the following: (1) cooling capacity in tons, (2) work of compressor in kW, (3) COP of the system, (4) EER of the system, and (5) refrigerating efficiency. Can you also show the plot on the chart provided?arrow_forwardAir enters a humidifier at 34.25°C and 30% RH. If the air leaves the humidifier at 70% RH. Determine: The exit dry-bulb temperature of the air in °C.The saturation effectiveness in %.arrow_forwardGaisano Mall uses an atmospheric cooling tower to provide cooling for the jacket water of a four-stroke 309 KW diesel generator. The cooling tower efficiency is 53% at a temperature of approach of 11 deg. C. if ambient air has a relative humidity of 60% and dry bulb temperature of 31 deg. C, determine the cooling water supplied to the diesel engine in liters/hour.arrow_forward
 Refrigeration and Air Conditioning Technology (Mi...Mechanical EngineeringISBN:9781305578296Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill JohnsonPublisher:Cengage Learning
Refrigeration and Air Conditioning Technology (Mi...Mechanical EngineeringISBN:9781305578296Author:John Tomczyk, Eugene Silberstein, Bill Whitman, Bill JohnsonPublisher:Cengage Learning
