
Concept explainers
(a)
Interpretation:
The possible structures of the starting materials of the given Diels-Alder product are to be given.
Concept introduction:
In order to find the starting materials of the given compounds follows the rules are given below.
First, label the double bond carbons as
In order to complete the diene structure, eliminate the double bonds between
In order to complete the dienophile structure, add the double bonds between its carbons.
Also, dienophile is substituted with electron withdrawing groups.
Answer to Problem 15.20P
The possible structures of starting materials of given compound can be as follows.
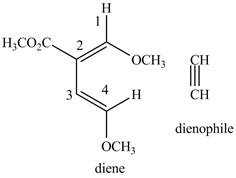
Explanation of Solution
First designate the double bond carbon atoms as
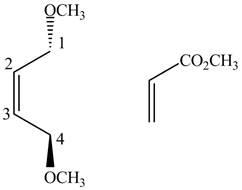
Figure 1
In order to complete the structure of diene, remove the double bonds present between carbons
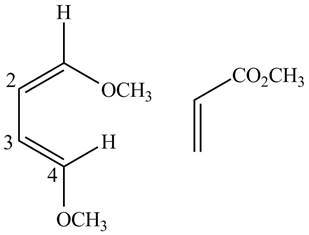
Figure 2
Finally, complete the dienophile structure by adding the double bonds between carbons.
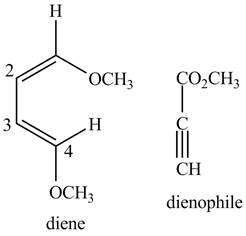
Figure 3
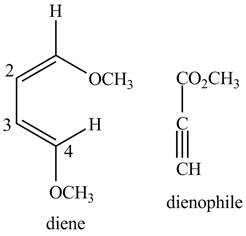
Similarly, second possibility on the basis of above mentioned steps will be as follows.
First designate the double bond carbons as
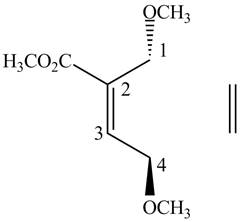
Figure 4
In order to complete the structure of diene, remove the double bonds present between carbons
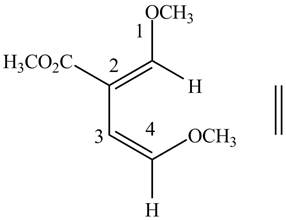
Figure 5
Finally, complete the dienophile structure by adding the double bonds between carbons
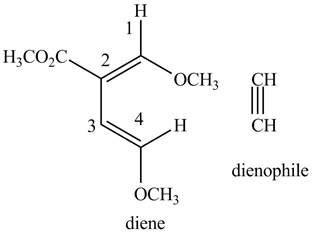
Figure 6
The possible starting materials of the given compound are shown in Figure 3 and Figure 6.
(b)
Interpretation:
The possible structures of the starting materials of the given Diels-Alder product are to be given.
Concept introduction:
First, label the double bond carbons as
In order to complete the diene structure, eliminate the double bonds between
In order to complete the dienophile structure, add the double bonds between its carbons.
Also, dienophile is substituted with electron withdrawing groups.
Answer to Problem 15.20P
The possible structures of starting materials of given compound are shown below.
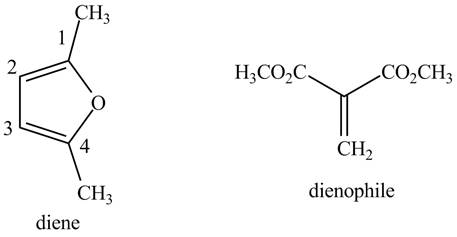
Explanation of Solution
First designate the double bond carbons as
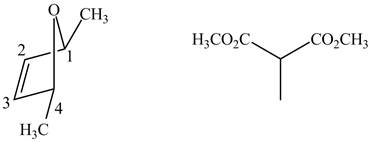
Figure 7
In order to complete the structure of diene, remove the double bonds present between carbons
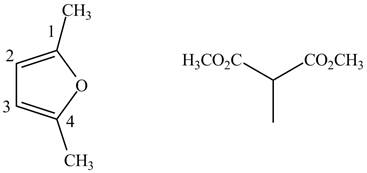
Figure 8
Finally, complete the dienophile structure by adding the double bonds between carbon atoms.
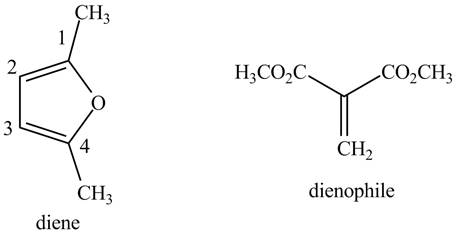
Figure 9
The possible starting materials of given compound are shown in Figure 9.
(c)
Interpretation:
The possible structures of the starting materials of the given Diels-Alder product are to be given.
Concept introduction:
First, label the double bond carbons as
In order to complete the diene structure, eliminate the double bonds between
In order to complete the dienophile structure, add the double bonds between its carbons.
Also, dienophile is substituted with electron withdrawing groups.
Answer to Problem 15.20P
The possible structures of starting materials of given compound are as follows.
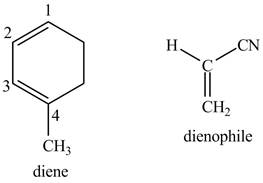
Explanation of Solution
First designate the double bond carbons as
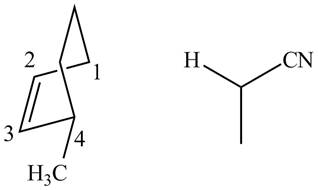
Figure 10
In order to complete the structure of diene, remove the double bonds present between carbons
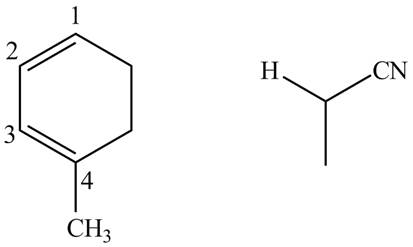
Figure 11
Finally, complete the dienophile structure by adding the double bonds between carbons.
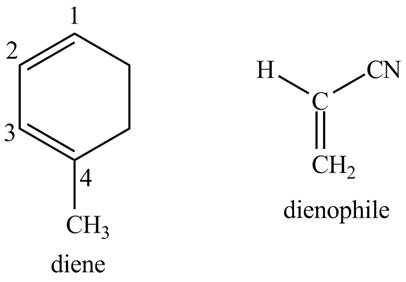
Figure 12
The possible starting materials of given compound are shown in Figure 12.
(d)
Interpretation:
The possible structures of the starting materials of the given Diels-Alder product are to be given.
Concept introduction:
First, label the double bond carbons as
In order to complete the diene structure, eliminate the double bonds between
In order to complete the dienophile structure, add the double bonds between its carbons.
Also, dienophile is substituted with electron withdrawing groups.
Answer to Problem 15.20P
The possible structures of starting materials of given compound are as follows.
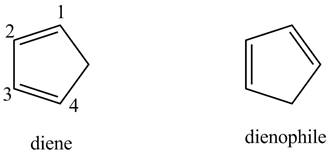
Explanation of Solution
First designate the double bond carbons as
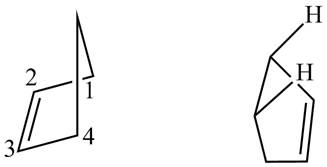
Figure 13
In order to complete the structure of diene, remove the double bonds present between carbons

Figure 14
Finally, complete the dienophile structure by adding the double bonds between carbons.
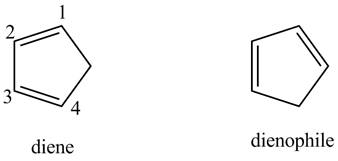
Figure 15
The possible starting materials of given compound are shown in 15.
Want to see more full solutions like this?
Chapter 15 Solutions
Organic Chemistry
- Many variations of ring-closing metathesis have now been reported. Tandem ring–opening–ring-closing metathesis can occur with cyclic alkenes that contain two additional carbon–carbon double bonds. In this reaction, the cycloalkene is cleaved, and two new rings are formed. [1] What compounds are formed in this tandem reaction with the following substrates? [2] Devise a synthesis of the substrate in part (b) that uses a Diels–Alder reaction with diethyl maleate as the dienophile.arrow_forwardThe diene lactone shown in part (a) has one electron-donating group (¬OR) and one electron-withdrawing group(C“O). This diene lactone is sufficiently electron-rich to serve as the diene in a Diels–Alder reaction.(a) What product would you expect to form when this diene reacts with methyl acetylenecarboxylate, a strong dienophile?arrow_forwardMany variations of ring-closing metathesis have now been reported. Tandem ring-opening-ring-closing metathesis can occur with cyclic alkenes that contain two additional carbon-carbon double bonds. In this reaction, the cycloalkene is cleaved, and two new rings are formed. [1] What compounds are formed in this tandem reaction with the following substrates? [2] Devise a synthesis of the substrate in part (b) that uses a Diels–Alder reaction with diethyl maleate as the dienophile.arrow_forward
- Aromaticity On the basis of Huckel's rule, label the following molecules as aromatic, nonaromatic or antiaromatic. Share your answers and comment on two other students' postings - have they correctly labeled the molecules? (a) (c) (e) anthracene [12]annulene azulene (b) (d) [10]annulene [14]annulene S-indacenearrow_forwardDiscuss the hybridization, aromaticity, and stability of the following organic intermediate. Also, arrange them in the increasing order of stability. CHarrow_forward(a) Which of the following phenols is the least acidic? (b) Which of the following phenols is the most acidic?arrow_forward
- One step in the synthesis of dodecahedrane involves reaction of the tetraene C with dimethylacetylene dicarboxylate (D) to afford two compounds having molecular formula C16H16O4. This reaction has been called a domino Diels–Alder reaction. Identify the two products formed.arrow_forwardOne step in the synthesis of dodecahedrane (Section 4.11) involves reaction of the tetraene C with dimethylacetylene dicarboxylate (D) to afford two compounds having molecular formula C16H16O4. This reaction has been called a domino Diels–Alder reaction. Identify the two products formed.arrow_forward(c) Diels-Alder reactions are a highly effective way to synthesise stereospecifically fused cyclic structures. (1) Using suitable diagrams, curly arrows and/or reaction schemes, explain why the reaction between cyclopentadiene and maleic anhydride favours formation of the endo product. (ii) If the reaction in part (i) were conducted using furan instead of cyclopentadiene, what difference would you observe in the product/s, with respect to their stereochemistry? (No need to draw reaction mechanisms) (iii) Draw the stereospecific 3D structure of the product formed during the Diels-Alder reaction below. (No need to show the reaction mechanism) Нeat Br Br (Figure Q11ciii)arrow_forward
- Following is an example of a type of reaction known as a Diels-Alder reaction 1,3-Pentadiene Ethylene 3-Methylcyclohexene (a racemic mixture) The Diels-Alder reaction between a diene and an alkene is quite remarkable in that it is one of the few ways that chemists have to form two new carbon-carbon bonds in a single reaction. Given what you know about the relative strengths of carbon-carbon sigma and pi bonds, would you predict the Diels-Alder reaction to be exothermic or endothermic? Explain your reasoning.arrow_forward8. (a) Benzene derivatives exhibit medium to strong absorption in UV-region. Explain why aniline and phenoxide ion have strong UV-absorptions.arrow_forwardConsider the tetracyclic aromatic compound drawn below, with rings labeled as A, B, C, and D. (a) Which of the four rings is most reactive in electrophilic aromatic substitution? (b) Which of the four rings is least reactive in electrophilic aromatic substitution? (c) What are the major product(s) formed when this compound is treated with one equivalent of Br2?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





