
Concept explainers
Draw the product(s) formed when each
![]()
(a)
Interpretation:
The product(s) formed by the reaction of given alkene with
Concept introduction:
The reaction of hydrogen halide with alkene results in the formation of alkyl halide. This type of reaction is an electrophilic addition of hydrogen halide. Electrophilic addition reactions are those in which breaking of pi bond take place to form new sigma bond.
Answer to Problem 15.24P
The product formed by the reaction of given alkene with
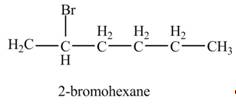
The product formed by the reaction of given alkene with
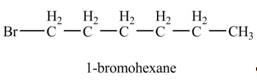
Explanation of Solution
Electrophilic addition reaction follows Markovnikov rule. According to Markovnikov rule, the positive part of halogen acid attached to that carbon atom in
The given alkene is shown below.
![]()
Figure 1
The steps followed by electrophilic addition reaction are stated below:
➢ First protonation of the alkene take place to generate the carbocation.
➢ The halide ion will attack on the carbocation to give the final product.
The product formed by the reaction of given alkene with
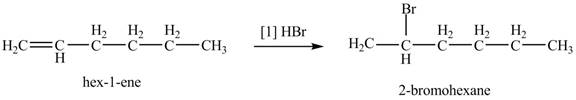
Figure 2
The product formed by the reaction of given alkene with
The addition of
The addition of
The product formed by the reaction of given alkene with
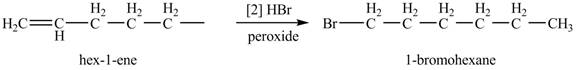
Figure 3
The product formed by the reaction of given alkene with
The product(s) formed by the reaction of given alkene with
(b)
Interpretation:
The product(s) formed by the reaction of given alkene with
Concept introduction:
The reaction of hydrogen halide with alkene results in the formation of alkyl halide. This type of reaction is an electrophilic addition of hydrogen halide. Electrophilic addition reactions are those in which breaking of pi bond take place to form new sigma bond.
Answer to Problem 15.24P
The product formed by the reaction of given alkene with
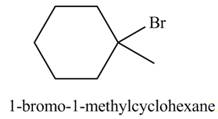
The product formed by the reaction of given alkene with
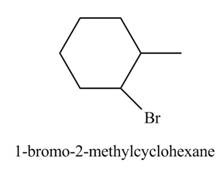
Explanation of Solution
Electrophilic addition reaction follows Markovnikov rule. According to Markovnikov rule, the positive part of halogen acid attached to that carbon atom in
The given alkene is shown below.
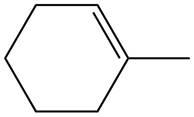
Figure 4
The steps followed by electrophilic addition reaction are stated below:
➢ First protonation of the alkene take place to generate the carbocation.
➢ The halide ion will attack on the carbocation to give the final product.
The product formed by the reaction of given alkene with
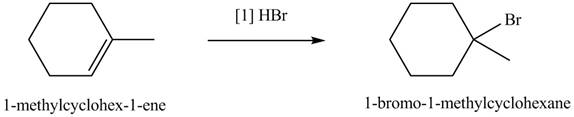
Figure 5
The product formed by the reaction of given alkene with
The addition of
The addition of
The product formed by the reaction of given alkene with
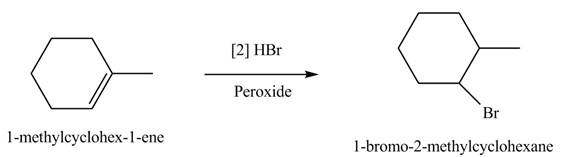
Figure 6
The product formed by the reaction of given alkene with
The product(s) formed by the reaction of given alkene with
(c)
Interpretation:
The product(s) formed by the reaction of given alkene with
Concept introduction:
The reaction of hydrogen halide with alkene results in the formation of alkyl halide. This type of reaction is an electrophilic addition of hydrogen halide. Electrophilic addition reactions are those in which breaking of pi bond take place to form new sigma bond.
Answer to Problem 15.24P
The product formed by the reaction of given alkene with
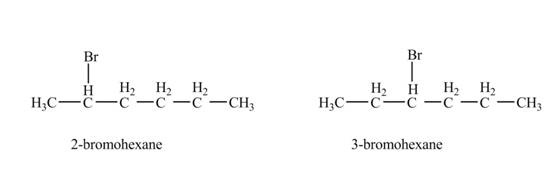
The product formed by the reaction of given alkene with

Explanation of Solution
Electrophilic addition reaction follows Markovnikov rule. According to Markovnikov rule, the positive part of halogen acid attached to that carbon atom in
The given alkene is shown below.

Figure 7
The steps followed by electrophilic addition reaction are stated below:
➢ First protonation of the alkene take place to generate the carbocation.
➢ The halide ion will attack on the carbocation to give the final product.
The product formed by the reaction of given alkene with
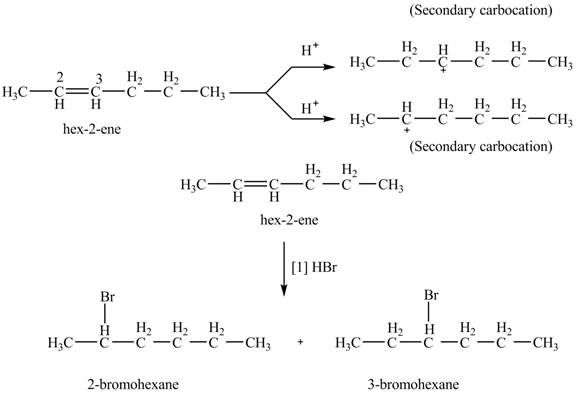
Figure 8
The reaction of given alkene with
The addition of
The addition of
The product formed by the reaction of given alkene with
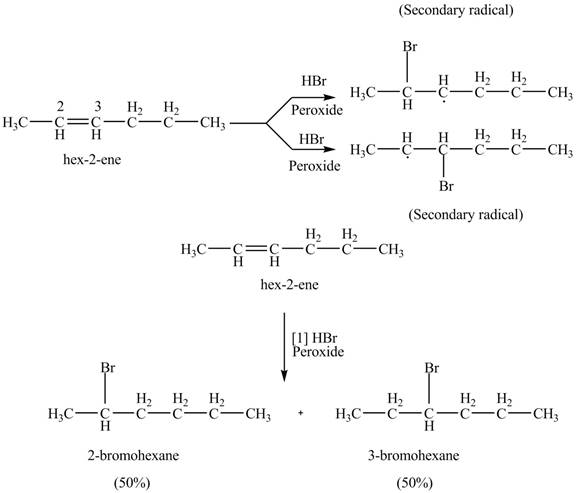
Figure 9
The reaction of given alkene with
The product(s) formed by the reaction of given alkene with
Want to see more full solutions like this?
Chapter 15 Solutions
Organic Chemistry
- a. What five-carbon alkene forms the same product whether it reacts with HBr in the presence of a peroxide or with HBr in the absence of a peroxide?b. Draw the structures of four six-carbon alkenes that form the same product, whether they react with HBr in the presence of a peroxide or with HBr inthe absence of a peroxide.arrow_forwardFor alkenes A, B, C, and D: (a) Rank A—D in order of increasing heat of hydrogenation; (b) rank A—D in order of increasing rate of reaction with H2, Pd-C; (c) draw the products formed when each alkene is treated with ozone, followed by Zn, H2O.arrow_forwardDraw the product formed when (CH3)2CHOH is treated with following reagent. H2SO4arrow_forward
- Draw the products formed when each compound is treated with HNO3 and H2SO4.State whether the reaction occurs faster or slower than a similar reaction with benzene.arrow_forwardDraw the products formed when phenol (C6H5OH) is treated with each set of reagents. a. [1] HNO3, H2SO4; [2] Sn, HCl b. [1] (CH3CH2)2CHCOCl, AlCl3; [2] Zn(Hg), HCl c. [1] CH3CH2Cl, AlCl3; [2] Br2, hν d. [1] (CH3)2CHCl, AlCl3; [2] KMnO4arrow_forwardSynthesize each compound from (CH3)3CH.arrow_forward
- For alkenes A, B, C, and D: (a) Rank A—D in order of increasing heat ofhydrogenation; (b) rank A—D in order of increasing rate of reaction withH2, Pd-C; (c) draw the products formed when each alkene is treated withozone, followed by Zn, H2O.arrow_forwardDraw the product formed from cyclohex-2-enone in each of three reactions. Reaction 1 Reaction 2 Reaction 1 1. NaCN 1. (CH3)2CuLi Reaction 2 product product 2. H3O*, H20 2. CH3CH21 A 1. EtO,CCH,CO¿Et NaOEt, HOET 2. Hао", Н-0, д Reaction 3 Reaction 3 productarrow_forwardWhat reagents ( A–D) are needed to carry out each reaction in the following sequence?arrow_forward
- Draw the products of each reaction, and state whether the reaction is faster or slower than a similar reaction with benzene.arrow_forwardDraw the organic products formed when cyclopentene is treated withfollowing reagent. [1] CH3CO3H; [2] H2O, HO−arrow_forwardDraw the products formed (including stereoisomers) in each reaction.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





