
Concept explainers
(a)
Interpretation:
The product for the reaction of ethylbenzene with bromine gas and
Concept introduction:
An electrophilic
Answer to Problem 16.35AP
No product will be formed in the reaction of ethylbenzene with bromine gas and
Explanation of Solution
Ethylbenzene is ortho and para directing group, the electrophile will substitute on either ortho or para position of the benzene ring. In the dark no bromine free radical will be generated, therefore, no free radical reaction will occur. Carbon tetrachloride does not facilitate the production of positively charged
No reaction takes place between ethylbenzene with bromine gas and
(b)
Interpretation:
The product for the reaction ethylbenzene with
Concept introduction:
An electrophilic aromatic substitution reaction is a type of reaction in which an electrophile substitutes a hydrogen atom of the aromatic ring. Aromatic ring does not easily undergo nucleophilic substitution reaction; however, it undergoes electrophilic substitution reaction easily.
Answer to Problem 16.35AP
The products for the reaction ethylbenzene with
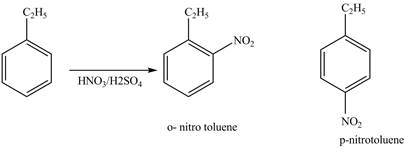
Explanation of Solution
Ethylbenzene is ortho and para directing group, the electrophile will substitute on either ortho or para position of the benzene ring. The nitronium ion
The corresponding
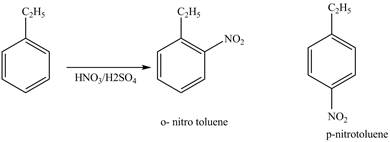
Figure 1
The products for the reaction ethylbenzene with
(c)
Interpretation:
The product for the reaction ethylbenzene with concentrated
Concept introduction:
An electrophilic aromatic substitution reaction is a type of reaction in which an electrophile substitutes a hydrogen atom of the aromatic ring. Aromatic ring does not easily undergo nucleophilic substitution reaction; however, it undergoes electrophilic substitution reaction easily.
Answer to Problem 16.35AP
The products for the reaction ethylbenzene with concentrated
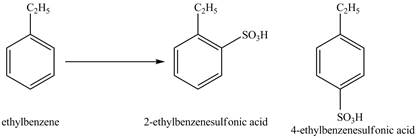
Explanation of Solution
Ethylbenzene is ortho and para directing group, the electrophile will substitute on either ortho or para position of the benzene ring. In the presence of concentrated sulphuric acid, the
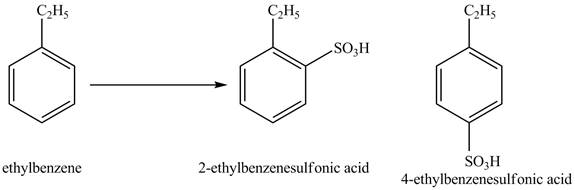
Figure 2
The products for the reaction ethylbenzene with concentrated
(d)
Interpretation:
The product for the reaction ethylbenzene with propionyl chloride and
Concept introduction:
Friedel Craft acylation is an electrophilic aromatic substitution reaction. In this reaction, the synthesis of the monoacylated product takes place from the reaction between aromatic rings and acyl chlorides.
Answer to Problem 16.35AP
The products for the reaction of ethylbenzene with propionyl chloride and
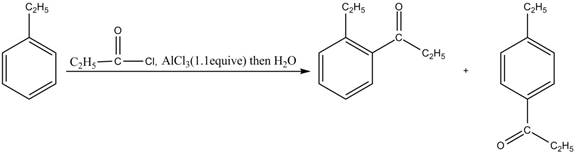
Explanation of Solution
Friedel-Crafts alkylation permits the synthesis of alkylated products by the reaction of arenes with alkyl chlorides in the presence of aluminum chloride (Lewis acid). This alkylation reaction comes under the category of electrophilic aromatic substitution. Ethylbenzene is ortho and para directing group, the electrophile will substitute on either ortho or para position of the benzene ring. The acyl group attacks either ortho or para position of ethylbenzene to form the product. The corresponding chemical reaction is shown below.
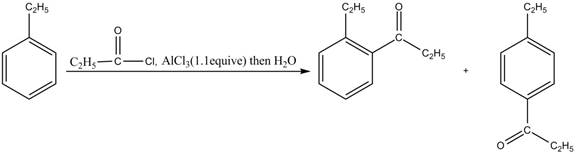
Figure 3
The products for the reaction ethylbenzene with propionyl chloride and
(e)
Interpretation:
The product for the exothermic reaction of ethylbenzene with
Concept introduction:
An electrophilic aromatic substitution reaction is a type of reaction in which an electrophile substitutes a hydrogen atom of the aromatic ring. Aromatic ring does not easily undergo nucleophilic substitution reaction; however, it undergoes electrophilic substitution reaction easily.
Answer to Problem 16.35AP
The product for the exothermic reaction of ethylbenzene with
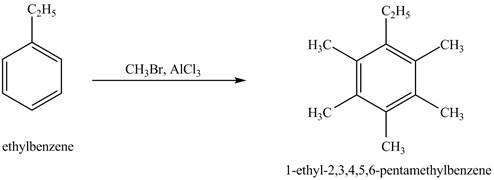
Explanation of Solution
Ethylbenzene is ortho and para directing group, the electrophile will substitute on either ortho or para position of the benzene ring. In the presence of
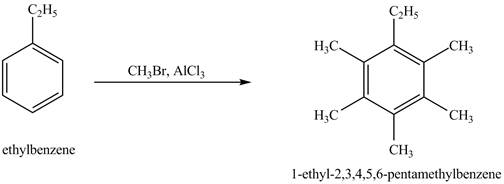
Figure 4
The product for the exothermic reaction of ethylbenzene with
(f)
Interpretation:
The product for the exothermic reaction of ethylbenzene with bromine gas and
Concept introduction:
An electrophilic aromatic substitution reaction is a type of reaction in which an electrophile substitutes a hydrogen atom of the aromatic ring. Aromatic ring does not easily undergo nucleophilic substitution reaction; however, it undergoes electrophilic substitution reaction easily.
Answer to Problem 16.35AP
The products for the exothermic reaction of ethylbenzene with bromine gas and
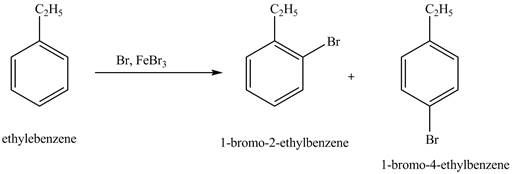
Explanation of Solution
Ethylbenzene is ortho and para directing group, the electrophile will substitute on either ortho or para position of the benzene ring. In this reaction, positively charged
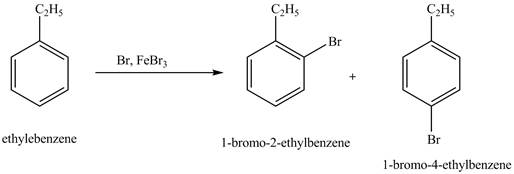
Figure 5
The products for the exothermic reaction ethylbenzene with bromine gas and
Want to see more full solutions like this?
Chapter 16 Solutions
Organic Chemistry
- (a) (b) HO N N-H Catalytic H+ (-H₂O) ? (c) NH2 HO N (d)arrow_forwardDesribe the reaction product. (q) CI ОН ОН (s) 1 H ОН ОНarrow_forward(b) Predict the suitable solvent (H2O or CH3COCH3) to increase the reaction of bromopropane (CH3CH2CH2B1) with sodium hydroxide (NaOH). Two reactions are shown below: NaOH, 55 °C CH;CH,CH,Br CH;CH,CH,OH + NaBr H,O (i) NaOH, 55 °C CH;CH,CH,Br CH;CH,CH,OH NaBr H,C CH (ii)arrow_forward
- Provide the major product for the following reaction? (1) Mg, ether (2) CO2. (3) H3O* Brarrow_forwardWhich of the following are the major products from the reaction shown below? (A) (C) + Br CH H Br H (B) (D) O Br Br Narrow_forwardCN -CN H30® /A (A); Product (A) of the reaction is : CN CN (a) (b) CO2H (c) (d) CO2Harrow_forward
- 4. Predict the product obtained from following oxidation reaction (a) он Naoa CH,COOHarrow_forwardWhat is the major product of the following reaction HINT consoder if both Sn1 and Sn2 could take place? CI (a) OH OH H₂O (b) HO OH (c) HO (d) There will be no reactionarrow_forwardWhich of the following compounds are suitable solvents for Grignard reactions?(a) n-hexane (b) CH3¬O¬CH3 (c) CHCl3(d) cyclohexane (e) benzene (f) CH3OCH2CH2OCH3arrow_forward
- provide the structure of the intermediate and product for the following reaction : (c) H CH,OH/H (C)arrow_forward(b) (d) (b) The following reactions as written will NOT give the indicated product. For each of the reactions explain (a) НО (i) (ii) (iii) OH The reason why the reaction as written will not occur; How the reaction could be modified to give the indicated product (may include the use of alternative or additional reagents); What should be the outcome for the reactions as written? OH CO,CH,CH, OH 1. CH₂OH/H* 2. Zn/Hg 3. H* 1, (CH₂CH₂)CuLi 2. PCC CrO3 Pyridine 2 x CH₂MgBr H+ ỌCH3 COH(CH3)2 = OHarrow_forwardMethionine, C5H₁1NO2S, is an essential amino acid in humans that is important in angiogenesis and the growth of new blood vessels. When methionine is burned in the presence of excess oxygen, O2, what products are formed? Select all products formed. NO₂ S₂ CO₂ No answer text provided. N₂ H₂O SO2arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





