
(a)
Interpretation:
The set of reagents satisfying the given conditions used for the synthesis of the given
Concept introduction:
An
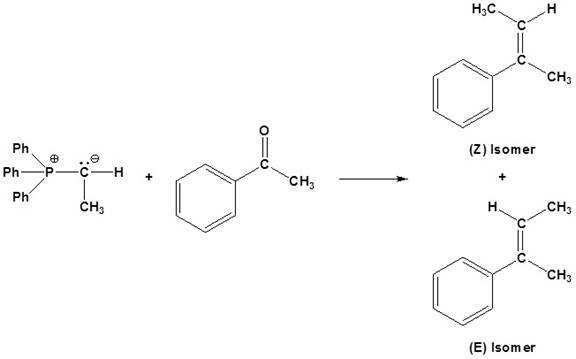
(b)
Interpretation:
The
Concept introduction:
An aldehyde or a ketone reacts with phosphonium ylide to form an alkene. This reaction is known as wittig reaction.
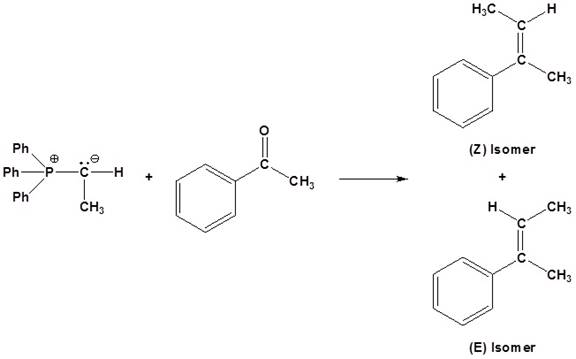
(c)
Interpretation:
The reagents used for the synthesis of given set of alkenes should be determined.
Concept introduction:
An aldehyde or a ketone reacts with phosphonium ylide to form an alkene. This reaction is known as wittig reaction.
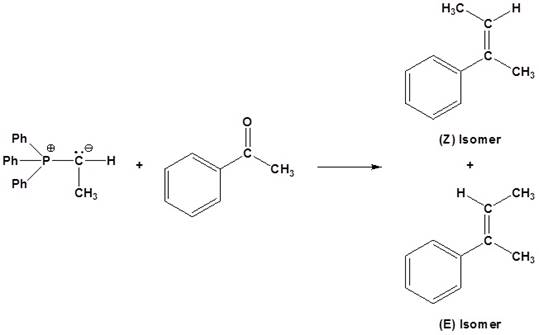
Want to see the full answer?
Check out a sample textbook solution
Chapter 16 Solutions
Organic Chemistry (8th Edition)
- Consider the following nucleophilc substitution. LOCH3 Br OCH3 в A a. Identify the reaction conditions A and specify the reaction mechanism for the nucleophilic substitution. Explain your choice. b. Explain why the transformation does not give rise to significant elimination. c. Which product(s) B is formed in the reaction? Please specify the stereochemistry clearly with reference to the reaction mechanism.arrow_forwarda. HO b. NH 33. Which is a better nucleophile? a. H₂O or HO c. H₂S b. NH3 or NH₂ C. CH, CO in a better leaving group. or CH3CH₂O d. -0 or -0arrow_forwardWhich of the following compounds cannot be prepared by a Heck reaction? a. For those compounds that can be prepared by a Heck reaction, what starting materials are required?arrow_forward
- 12. What alkyl halide and nucleophile are needed to prepare the following compounds? a. b. OHarrow_forwardEnols are quite reactive toward electrophiles than alkenes because: а. The OH group has a powerful electron-donating O resonance effect b. A resonance structure can be drawn that places a negative charge on one of the carbon atoms, making this carbon electrophilic С. The enol can react with nucleophilic carbon to form a new bond to carbon d. None of the options are correctarrow_forwardDevise a synthesis of each compound using CH3CH₂CH=CH₂ as the starting material. You may use any other organic compounds or inorganic reagents. a. b. d. e. Br Br Br OH OH (+ enantiomer)arrow_forward
- a. Which of the following compounds cannot be prepared by a Heck reaction?b. For those compounds that can be prepared by a Heck reaction, what starting materials are required?arrow_forwardThe major product that would form from the presented reaction scheme is? .C. CH3 H2/Pd NO2arrow_forwardIf the alkyl halide in each of these reactions was an alkyl chloride instead of thebromide,A. the reaction rate would decrease.B. a better leaving group would be involved. C. a polar aprotic solvent would be needed.D. ΔG would be decreased.arrow_forward
- Select all compounds capable of keto-enol tautomerism. он H.C. он H.C. H.C. H.C. CH, CH, CH, H.C H.C a b darrow_forwardWhat is the major organic product of the following sequence of reactions? Yot A. B. ZI NH₂ H₂SO4 C. D. ZI ZIarrow_forward2. Provide reagents/conditions to accomplish the following syntheses. More than one step is required in some cases. a. b. CN Br Br CH₂ Revised F Farrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





