
(a)
Interpretation:
Structural formula of the compound whose
Concept-Introduction:
Nuclear Magnetic Resonance Spectroscopy:
Nuclear Magnetic Resonance Spectroscopy or NMR Spectroscopy is a technique used to find the purity, content and molecular structure of the compound. Resonance transition between energy levels happens if
It is also known as Proton Nuclear Magnetic Resonance. Hydrogen nuclei with in the molecule is considered here.
The NMR signals for the
(a)
Explanation of Solution
Splitting in
In
In
.94ppm will the shift of
13.69ppm will the shift of
So the given values of the
Structural formula of the compound is;
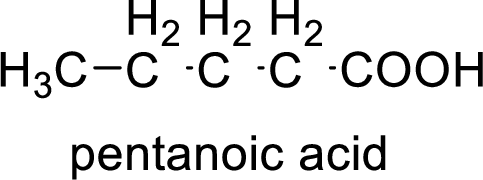
(b)
Interpretation:
Structural formula of the compound whose
Concept-Introduction:
Nuclear Magnetic Resonance Spectroscopy:
Nuclear Magnetic Resonance Spectroscopy or NMR Spectroscopy is a technique used to find the purity, content and molecular structure of the compound. Resonance transition between energy levels happens if electromagnetic radiation with specific frequency is applied on an atomic nuclei placed in an electromagnetic field. This transition happens only if the frequency of applied radiation matches with the frequency of magnetic field, then it is to be in resonance condition. If an external magnetic field is applied spin gets excited from its ground state to the excited state by absorbing energy. The absorbed radio frequency is emitted back at the same frequency level, when the spin returns to its ground state. This radio frequency will give the NMR spectra. The plot of the spectra is between Intensity of the signal VS magnetic field. Trimethyl Silane ie, TMS is used as the reference. Chemical shift is a term which refers to the position in the spectrum. It is dependent on several factors like electron density around the proton, inductive effect etc.
It is also known as Proton Nuclear Magnetic Resonance. Hydrogen nuclei with in the molecule is considered here.
The NMR signals for the
(b)
Explanation of Solution
Splitting in
In
In
1.08ppm will the shift of
30.69ppm will the shift of
So the given values of the
Structure formula of the compound is:
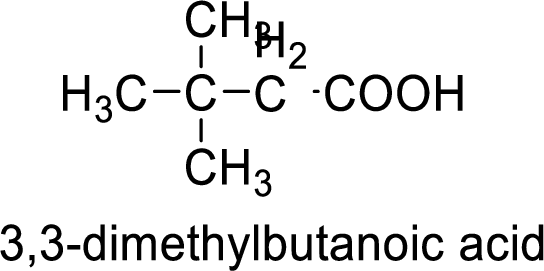
(c)
Interpretation:
Structural formula of the compound whose
Concept-Introduction:
Nuclear Magnetic Resonance Spectroscopy:
Nuclear Magnetic Resonance Spectroscopy or NMR Spectroscopy is a technique used to find the purity, content and molecular structure of the compound. Resonance transition between energy levels happens if electromagnetic radiation with specific frequency is applied on atomic nuclei placed in an electromagnetic field. This transition happens only if the frequency of applied radiation matches with the frequency of magnetic field, then it is to be in resonance condition. If an external magnetic field is applied spin gets excited from its ground state to the excited state by absorbing energy. The absorbed radio frequency is emitted back at the same frequency level, when the spin returns to its ground state. This radio frequency will give the NMR spectra. The plot of the spectra is between Intensity of the signal VS magnetic field. Trimethyl Silane ie, TMS is used as the reference. Chemical shift is a term which refers to the position in the spectrum. It is dependent on several factors like electron density around the proton, inductive effect etc.
It is also known as Proton Nuclear Magnetic Resonance. Hydrogen nuclei with in the molecule are considered here.
The NMR signals for the
(c)
Explanation of Solution
Splitting in
In
In
.93ppm will the shift of
11.81ppm will be the shift of
So the given values of the
Structural formula of the product is;
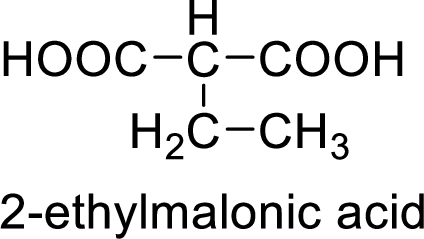
(d)
Interpretation:
Structural formula of the compound whose
Concept-Introduction:
Nuclear Magnetic Resonance Spectroscopy:
Nuclear Magnetic Resonance Spectroscopy or NMR Spectroscopy is a technique used to find the purity, content and molecular structure of the compound. Resonance transition between energy levels happens if electromagnetic radiation with specific frequency is applied on an atomic nuclei placed in an electromagnetic field. This transition happens only if the frequency of applied radiation matches with the frequency of magnetic field, then it is to be in resonance condition. If an external magnetic field is applied spin gets excited from its ground state to the excited state by absorbing energy. The absorbed radio frequency is emitted back at the same frequency level, when the spin returns to its ground state. This radio frequency will give the NMR spectra. The plot of the spectra is between Intensity of the signal VS magnetic field. Trimethyl Silane ie, TMS is used as the reference. Chemical shift is a term which refers to the position in the spectrum. It is dependent on several factors like electron density around the proton, inductive effect etc.
It is also known as Proton Nuclear Magnetic Resonance. Hydrogen nuclei with in the molecule are considered here.
The NMR signals for the
(d)
Explanation of Solution
Splitting in
In
In
1.29ppm will the shift of
22.56ppm will the shift of
So the given values of
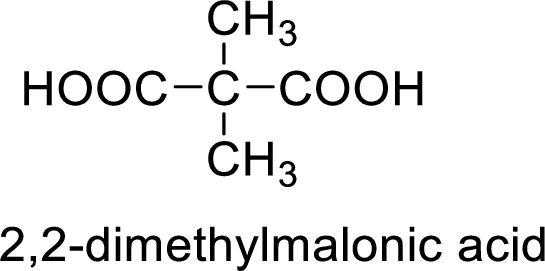
(e)
Interpretation:
Structural formula of the compound whose
Concept-Introduction:
Nuclear Magnetic Resonance Spectroscopy:
Nuclear Magnetic Resonance Spectroscopy or NMR Spectroscopy is a technique used to find the purity, content and molecular structure of the compound. Resonance transition between energy levels happens if electromagnetic radiation with specific frequency is applied on an atomic nuclei placed in an electromagnetic field. This transition happens only if the frequency of applied radiation matches with the frequency of magnetic field, then it is to be in resonance condition. If an external magnetic field is applied spin gets excited from its ground state to the excited state by absorbing energy. The absorbed radio frequency is emitted back at the same frequency level, when the spin returns to its ground state. This radio frequency will give the NMR spectra. The plot of the spectra is between Intensity of the signal VS magnetic field. Trimethyl Silane ie, TMS is used as the reference. Chemical shift is a term which refers to the position in the spectrum. It is dependent on several factors like electron density around the proton, inductive effect etc.
It is also known as Proton Nuclear Magnetic Resonance. Hydrogen nuclei with in the molecule are considered here.
The NMR signals for the
(e)
Explanation of Solution
Splitting in
In
In
1.91ppm will the shift of
18.11ppm will the shift of
So the given values of
Structural formula of the product;
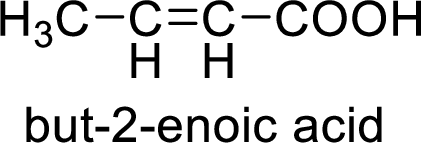
(f)
Interpretation:
Structural formula of the compound whose
Concept-Introduction:
Nuclear Magnetic Resonance Spectroscopy:
Nuclear Magnetic Resonance Spectroscopy or NMR Spectroscopy is a technique used to find the purity, content and molecular structure of the compound. Resonance transition between energy levels happens if electromagnetic radiation with specific frequency is applied on an atomic nuclei placed in an electromagnetic field. This transition happens only if the frequency of applied radiation matches with the frequency of magnetic field, then it is to be in resonance condition. If an external magnetic field is applied spin gets excited from its ground state to the excited state by absorbing energy. The absorbed radio frequency is emitted back at the same frequency level, when the spin returns to its ground state. This radio frequency will give the NMR spectra. The plot of the spectra is between Intensity of the signal VS magnetic field. Trimethyl Silane ie, TMS is used as the reference. Chemical shift is a term which refers to the position in the spectrum. It is dependent on several factors like electron density around the proton, inductive effect etc.
It is also known as Proton Nuclear Magnetic Resonance. Hydrogen nuclei with in the molecule are considered here.
The NMR signals for the
(f)
Explanation of Solution
Splitting in
In
In
2.34ppm will the shift of
34.02ppm will the shift of
So the given values of
Structure of the product
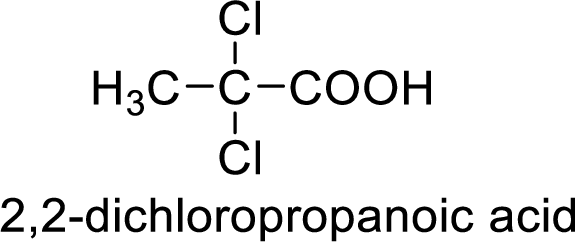
(g)
Interpretation:
Structural formula of the compound whose
Concept-Introduction:
Nuclear Magnetic Resonance Spectroscopy:
Nuclear Magnetic Resonance Spectroscopy or NMR Spectroscopy is a technique used to find the purity, content and molecular structure of the compound. Resonance transition between energy levels happens if electromagnetic radiation with specific frequency is applied on an atomic nuclei placed in an electromagnetic field. This transition happens only if the frequency of applied radiation matches with the frequency of magnetic field, then it is to be in resonance condition. If an external magnetic field is applied spin gets excited from its ground state to the excited state by absorbing energy. The absorbed radio frequency is emitted back at the same frequency level, when the spin returns to its ground state. This radio frequency will give the NMR spectra. The plot of the spectra is between Intensity of the signal VS magnetic field. Trimethyl Silane ie, TMS is used as the reference. Chemical shift is a term which refers to the position in the spectrum. It is dependent on several factors like electron density around the proton, inductive effect etc.
It is also known as Proton Nuclear Magnetic Resonance. Hydrogen nuclei with in the molecule are considered here.
The NMR signals for the
(g)
Explanation of Solution
Splitting in
In
In
1.42ppm will the shift of
13.69ppm will the shift of
So the given values of
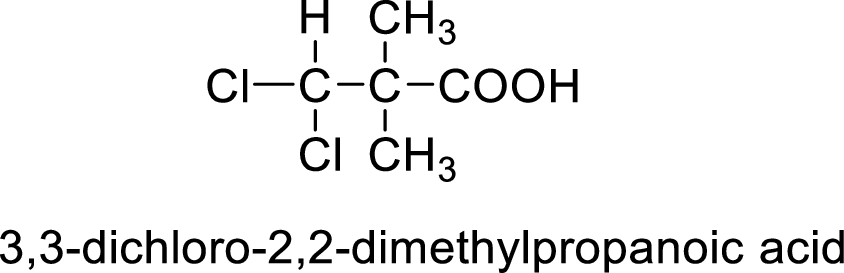
(h)
Interpretation:
Structural formula of the compound whose
Concept-Introduction:
Nuclear Magnetic Resonance Spectroscopy:
Nuclear Magnetic Resonance Spectroscopy or NMR Spectroscopy is a technique used to find the purity, content and molecular structure of the compound. Resonance transition between energy levels happens if electromagnetic radiation with specific frequency is applied on an atomic nuclei placed in an electromagnetic field. This transition happens only if the frequency of applied radiation matches with the frequency of magnetic field, then it is to be in resonance condition. If an external magnetic field is applied spin gets excited from its ground state to the excited state by absorbing energy. The absorbed radio frequency is emitted back at the same frequency level, when the spin returns to its ground state. This radio frequency will give the NMR spectra. The plot of the spectra is between Intensity of the signal VS magnetic field. Trimethyl Silane ie, TMS is used as the reference. Chemical shift is a term which refers to the position in the spectrum. It is dependent on several factors like electron density around the proton, inductive effect etc.
It is also known as Proton Nuclear Magnetic Resonance. Hydrogen nuclei with in the molecule are considered here.
The NMR signals for the
(h)
Explanation of Solution
Splitting in
In
In
97ppm will the shift of
13.24ppm will the shift of
So the given values of
Structural formula of the compound is;
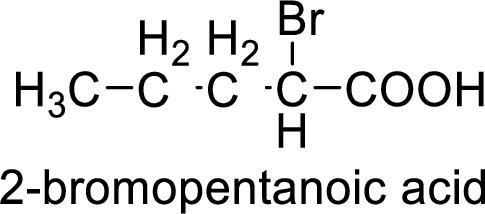
(i)
Interpretation:
Structural formula of the compound whose
Concept-Introduction:
Nuclear Magnetic Resonance Spectroscopy:
Nuclear Magnetic Resonance Spectroscopy or NMR Spectroscopy is a technique used to find the purity, content and molecular structure of the compound. Resonance transition between energy levels happens if electromagnetic radiation with specific frequency is applied on atomic nuclei placed in an electromagnetic field. This transition happens only if the frequency of applied radiation matches with the frequency of magnetic field, then it is to be in resonance condition. If an external magnetic field is applied spin gets excited from its ground state to the excited state by absorbing energy. The absorbed radio frequency is emitted back at the same frequency level, when the spin returns to its ground state. This radio frequency will give the NMR spectra. The plot of the spectra is between Intensity of the signal VS magnetic field. Trimethyl Silane ie, TMS is used as the reference. Chemical shift is a term which refers to the position in the spectrum. It is dependent on several factors like electron density around the proton, inductive effect etc.
It is also known as Proton Nuclear Magnetic Resonance. A hydrogen nucleus with in the molecule is considered here.
The NMR signals for the
(i)
Explanation of Solution
Splitting in
In
In
3.38ppm will the shift of
34.75ppm will the shift of
So the given values of
So the structure of the product is;
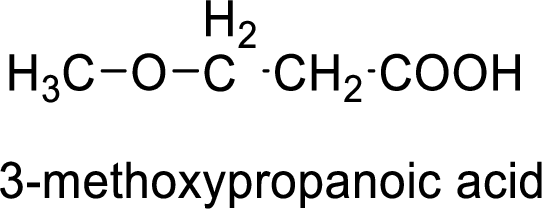
Want to see more full solutions like this?
Chapter 17 Solutions
Organic Chemistry
- Compound I (C11H14O2) is insoluble in water, aqueous acid, and aqueous NaHCO3, but dissolves readily in 10% Na2CO3 and 10% NaOH. When these alkaline solutions are acidified with 10% HCl, compound I is recovered unchanged. Given this information and its 1H-NMR spectrum, deduce the structure of compound I.arrow_forwardCompound A has molecular formula C5H10O. It shows three signals in the 1H-NMR spectrum - a doublet of integral 6 at 1.1 ppm, a singlet of integral 3 at 2.14 ppm, and a quintet of integral 1 at 2.58 ppm. Suggest a structure for A and explain your reasoning.arrow_forwardCompound B of molecular formula C9H19N shows a noteworthy infrared absorption at 3300 cm-1. Its 1H-NMR spectrum shows three singlets – δ 1.0 (6H), 1.1 (12H), 1.4 (1H) ppm. Its 13C-NMR spectrum has four signals – δ 25, 28, 41, 64 ppm. Suggest a structure for this compound. Please show work.arrow_forward
- A and B are isomeric dicarbonyl compounds of the molecular formula C5H&O2. The 'H NMR spectrum of A contains a singlet at 2.05 ppm and another singlet at 5.40 ppm. The 'H NMR spectrum of B contains three signals: a singlet at 2.3 ppm, a triplet at 1.10 ppm and a quartet at 2.70 ppm. Suggest structures for A and B and draw them in their respective boxes below. 1st attemptarrow_forwardCompound P has molecular formula C5H9ClO2. Deduce the structure of P from its 1H and 13C NMR spectra.arrow_forwardCompounds A and B have molecular formula C9H10O. Identify their structures from the 1H NMR and IR spectra given.arrow_forward
- An unknown compound has a molecular formula of C4H6O2. Its IR spectrum shows absorptions at 3095, 1762, 1254, and 1118 cm -1. It exhibits the following signals in its 1H NMR spectrum (ppm): 2.12 (singlet,3H), 4.55 (doublets of doublets, 1H), 4.85 (doublet of doublets, 1H), 7.25 (doublets of doublets, 1H); and the following signals in its 13C NMR spectrum (ppm): 20.8, 100.4, 141.2, 168.0. Draw the structure of the unknown compoundarrow_forwardCompounds A and B are isomers having the molecular formula C4H8O3. Identify A and B on the basis of their 1H NMR spectra.Compound A: δ 1.3 (3H, triplet); 3.6 (2H, quartet); 4.1 (2H, singlet); 11.1 (1H, broad singlet)Compound B: δ 2.6 (2H, triplet); 3.4 (3H, singlet); 3.7 (2H triplet); 11.3 (1H, broad singlet)arrow_forwardA hydrocarbon, compound B, has molecular formula C6H6, and gave an NMR spectrum with two signals: delta 6.55 pm and delta 3.84 pm with peak ratio of 2:1. When warmed in pyridine for three hr, compound B quantitatively converts to benzene. Mild hydrogenation of B yielded another compound C with mass spectrum of m/z 82. Infrared spectrum showed no double bonds; NMR spectrum showed one broad peak at delta 2.34 ppm. With this information, address the following questions. a) How many rings are in compound C? b) How many rings are probably in B? How many double bonds are in B? c) Can you suggest a structure for compounds B and C? d) In the NMR spectrum of B, the up-field signal was a quintet, and the down field signal was a triplet. How must you account for these splitting patterns?arrow_forward
- Compound B has molecular formula C9H10. The IR spectrum is shown below. The 1H-NMR spectrum shows a multiplet at 7.2 ppm integrating to 4H, a triplet at 2.9 ppm integrating to 4H, and a triplet at 2.1 ppm integrating to 2 H. Suggest a structure for B and explain your reasoningarrow_forwardA compound has the molecular formula C6H12O2. Its IR spectrum shows a strong absorption band near 1740 cm-1. Its 1H NMR spectrum consists of two singlets at δ 1.2 and δ 3.6. Which is the most likely structure of the compound?arrow_forwardCompounds Y and Z are isomers with the molecular formula C10H12O. The IR spectrum of each compound shows a strong absorption band near 1710 cm−1 . The 1H NMR spectra of Y and Z are given below. Propose structures for Y and Z.arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
