
Concept explainers
(a)
Interpretation:
The principal organic compound that is expected when
Concept introduction:
The addition reaction of the
Answer to Problem 17.22AP
The principal organic compound that is formed when
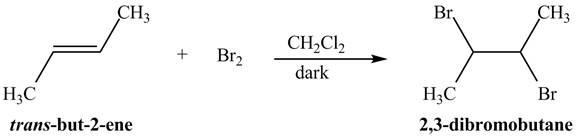
Explanation of Solution
The reaction of
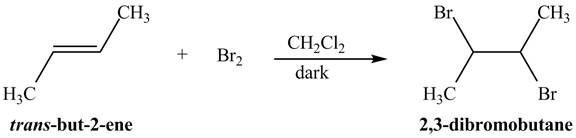
Figure 1
The addition of the bromine molecule on the double bond of the alkene takes place to give the dibromo product. The addition of bromine to the double bond is the anti-addition.
The principal organic compound that is formed when
(b)
Interpretation:
The principal organic compound that is expected when
Concept introduction:
The addition reactions of the alkene are very well known reactions. The electron density on the alkene double bond makes it nucleophilic. The alkene undergoes varieties of addition reaction via different mechanisms.
Answer to Problem 17.22AP
The principal organic compound that is formed when
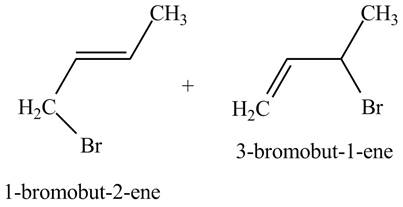
Explanation of Solution
The reaction of
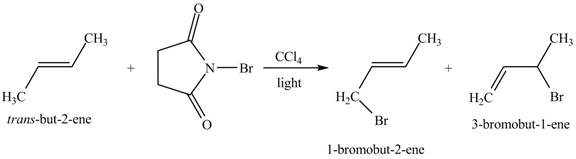
Figure 2
The reaction of an alkene with N-bromosuccinimide in the presence is the free-radical reaction. In this reaction, allylic bromination is observed instead of an addition to the alkene. The N-bromosuccinimide acts as radical initiator. This molecule breaks the
The principal organic compound that is formed when
(c)
Interpretation:
The principal organic compound that is expected when product(s) of part (b) undergo solvolysis in aqueous acetone is to be stated.
Concept introduction:
The nucleophilic substitution reactions are the reactions in which a group is substituted by a nucleophile. The rate of reaction depends upon the nucleophilicity and concentration of the nucleophile.
The
Answer to Problem 17.22AP
The principal organic compounds that are formed when product(s) of part (b) undergo solvolysis in aqueous acetone are shown below.
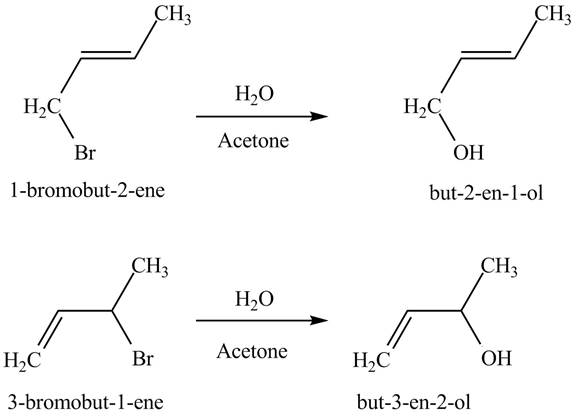
Explanation of Solution
The products of part (b) are shown below.
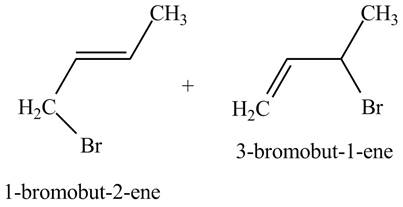
Figure 3
The reaction when product(s) of part (b) undergo solvolysis in aqueous acetone is shown below.
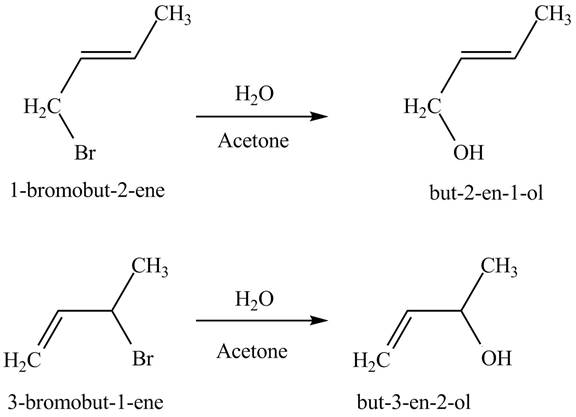
Figure 4
The nucleophilic substitution reaction takes place when solvolysis of products of part (b) takes place. The bromine group is substituted by the hydroxyl group giving rise to the formation of an allylic alcohol.
The principal organic compound that is formed when product(s) of part (b) undergo solvolysis in aqueous acetone is shown in Figure 4.
(d)
Interpretation:
The principal organic compound that is expected when product(s) of part (b) are reacted with
Concept introduction:
The reaction of an
Answer to Problem 17.22AP
The principal organic compounds that are formed when product(s) of part (b) are reacted with
Explanation of Solution
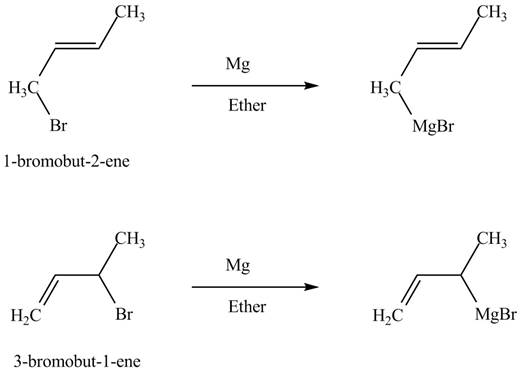
The products of part (b) are shown below.
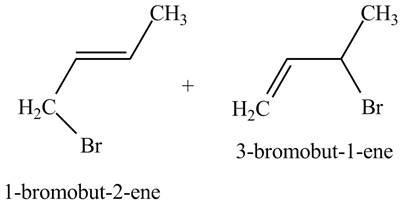
Figure 3
The reaction that occurs when product(s) of part (b) are reacted with
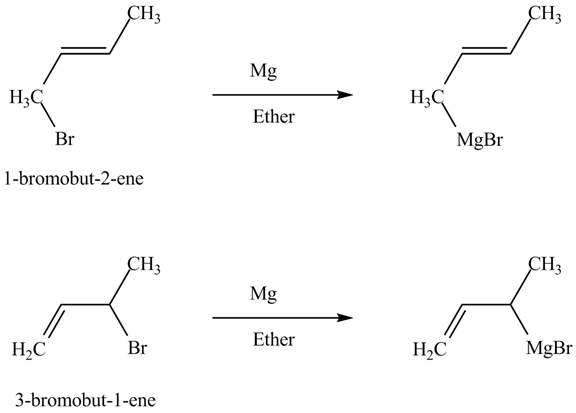
Figure 5
The allylic halide also undergoes the same kind of reactions are do the alkyl halide with magnesium metal in dry ether. They are also lead to the formation of Grignard reagent but this time with the allylic group.
The principal organic compound that is formed when product(s) of part (b) undergo solvolysis in aqueous acetone is shown in Figure 5.
(e)
Interpretation:
The principal organic compound that is expected when product(s) of part (d) are reacted with
Concept introduction:
The reaction of an alkyl halide with a metal-like magnesium in the presence of dry ether leads to the formation of
Answer to Problem 17.22AP
The principal organic compound that is formed when products of part (d) react with
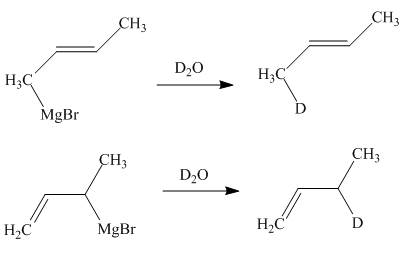
Explanation of Solution
The products of part (d) are shown below.
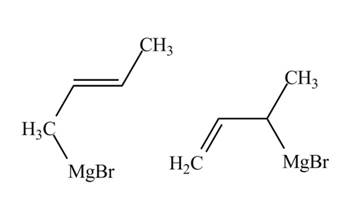
Figure 6
The Gringnard reagents react with

Figure 7
The principal organic compound that is formed when products of part (d) react with
Want to see more full solutions like this?
Chapter 17 Solutions
Organic Chemistry
- Name the reagents used in the following reactions: (i) Nitration of phenol to 2, 4, 6-trinitrophenol (ii) Butanal to Butanol . (iii) Friedel – Crafts acetylation of anisole (iv) Oxidation of primary alcohol to aldehydearrow_forward4 Predict the products of reactions of ethers and epoxides, including the following:(a) Cleavage and autoxidation of ethers(b) Acid- and base-promoted opening of epoxides(c) Reactions of epoxides with organometallic reagents(d) Cleavage of silyl ethersarrow_forwardn-Pentanol (CH3CH2CH2CH2CH2OH) and 2-methylbutan-2-ol (CH3CH2C(CH3)2OH) are converted to their corresponding alkyl chorides on being reacted with hydrogen chloride. (a) Write out an equation for each reaction (b) Assign each the appropriate symbol (SN1 or SN2) (c) Write a suitable mechanism for each reactionarrow_forward
- Consider the following structural formulas. OH H;C CH3 CH3 F G H (a) Which of the compounds F-l can easily be subjected to an elimination reaction? (b) Provide the reaction mechanism for all eliminations under (a).arrow_forward(b) Describe the hazards of (i) trioxygen: (ii) hydroxide ion; (ii) hydrogen sulfide. (c) Explain why an aqueous solution of sodium sulfide has an odor of hydrogen sulfide. (d) (11) Reduction of H,CCHCHO with NABH4 gives a product different from that of catalytic hydrogenation (H2 /Ni). What are the products?arrow_forward(a) The Friedel-Crafts reaction of benzene with 2-chloro-3-methylbutane in the presence of AlCl3 occurs with a carbocation rearrangement. Give mechanistic explanation and the product formed. (b) Predict the product(s) will be formed from the following reactions: (i) Bromination of p-methylbenzoic acid (ii) Sulphonation of m-bromoanisole (iii) Friedel-craft acylation of o-bromonitrobenzenearrow_forward
- Predict the major product or the necessary reagent or reactant to complete each of the following reactions. In the box before each reaction, indicate the mechanism followed by the reaction. (Free radical (FR), SN2, SN1). (b) (a) Cl₂ CH₂CH₂OH (a) 1 hv 2 (b) H Br H₂O Na+ SCH3 (c) (d) CI "CH3arrow_forwardThe reaction of 1-bromopropane and sodium hydroxide in ethanol occurs by an SN2mechanism. What happens to the rate of this reaction under the following conditions?(a) The concentration of NaOH is doubled.(b) The concentrations of both NaOH and 1-bromopropane are doubled.(c) The volume of the solution in which the reaction is carried out is doubled.arrow_forward4 Which of the following alcohols could not be made selectively by hydroboration-oxidation of an alkene? Explain. about the (b) (a) (c) OH | CH3CH₂CH₂CHCH3 In H OH (CH3)2CHC(CH3)2 (d) CH3 vino zbiory 1 OH o no atou OH vollot edt evig H noituloa siblos V H но но CHO SHO=3₂HƆ m² CH3s 10 out of ST bewollot giaylonoso no WBTⱭ 12-8 His to edustrie 5 wode, $2-8 OnMX dilw osviel vitabarrow_forward
- 6) Which is the organic product for the following reaction? (a) (b) (c) (d) LOH OH COOH OH OH COOH COOH KMnO4 H₂O (e) None of the above products will be formedarrow_forward(b) State the reagents needed to convert benzoic acid into the following compounds. (i) C6H§COCI (ii) C,H$CH2OH (iii) C6H$CONHCH3arrow_forwardPredict the major products (including stereochemistry) when cis-3-methylcyclohexanol reacts with the following reagents. (a) concentrated HBr (b) TsCl/pyridine, then NaBrarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





