
Concept explainers
(a)
Interpretation:
The binary hydrides shown in the Figure 1 has to be identified as ionic, covalent and interstitial.
Concept Introduction:
The compound containing hydrogen and just one other element is called as binary hydride. Type of hydride formation depends upon the element present in the group. Types of binary compounds are, ionic hydride, metallic hydride and covalent hydride.
Ionic hydride: Ionic hydrides are formed by alkali metals and heavier alkaline earth metals. They contain cations and
Covalent hydride: Covalent hydrides are formed by non-metals. These compound contains hydrogen which is bonded to another element by covalent bond. Most of the covalent hydrides consists of separate, small molecule have relatively weak intermolecular force of attraction, so they are gas or volatile liquid at normal temperature.
Metallic hydride: Metallic hydrides are formed by
(a)
Answer to Problem 18.18CP
In Figure 1,
Explanation of Solution
Given:
Given representation is shown in Figure 1.
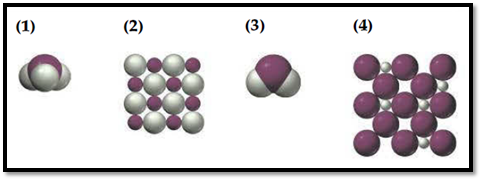
Figure 1
In this picture, ivory spheres denotes Hydrogen atoms or ions and purple spheres denotes an atom or ion of the element.
Picture (1) and (3) shows that hydrides (smaller atoms) are attached to the central atom, smaller molecules have relatively weak intermolecular force of attraction. This type of hydride are known as covalent hydride.
Picture (4) shows interstitial arrangement of hydrogen atom. They are often called as interstitial hydride because they consists of crystal lattice of metals with smaller hydrogen occupying holes or interstitial.
Picture (2) shows that it is slice of face centred arrangement. Ionic hydride only gives face centred arrangement. Thus it is concluded as ionic hydride formed by alkali metals.
So, picture
(b)
Interpretation:
The oxidation state of hydrogen and other element in compound (1), (2) and (3) has to be identified.
Concept Introduction:
Oxidation number: Oxidation number is defined as degree of oxidation (that is loss of an electrons) of an atom within the chemical compound.
There is a slight difference between oxidation state and oxidation number. In oxidation state, the electronegativity of an atom in a bond should be considered. However, while describing oxidation number, electronegativity will not be considered.
(b)
Answer to Problem 18.18CP
In compound (1), oxidation state of hydride is
In compound (2), oxidation state of hydride is
In compound (3), oxidation state of hydride is
Explanation of Solution
Given:
Given representation is shown in Figure 1.

Figure 1
In this picture, ivory spheres denotes Hydrogen atoms or ions and purple spheres denotes an atom or ion of the element.
Picture (1) illustrate a slice of the structure of nonstoichiometric interstitial hydride.
Calculation of oxidation state of hydride and other element in each compound as follows,
Compound (1):
Generally oxidation state of hydride in covalent compound is
Consider x as oxidation state of other element in the compound.
Therefore, the oxidation state of hydride in compound (1) is
Compound (2):
Generally oxidation state of hydride in ionic compound is
Consider x as oxidation state of other element in the compound.
Therefore, the oxidation state of hydride in compound (2) is
Compound (3):
Generally oxidation state of hydride in covalent compound is
Consider x as oxidation state of other element in the compound.
Therefore, the oxidation state of hydride in compound (3) is
Want to see more full solutions like this?
Chapter 18 Solutions
General Chemistry: Atoms First
- Which is the stronger acid, H2SO4 or H2SeO4? Why? You may wish to review the Chapter on acid-base equilibria.arrow_forwardThe elements sodium, aluminum, and chlorine are in the same period. (a) Which has the greatest electronegativity? (b) Which of the atoms is smallest? (c) Which is the largest possible oxidation state for each of these elements? (d) Will the oxide of each element in the highest oxidation state (write its formula) be acidic, basic, or amphoteric?arrow_forward(a) (i) (ii) (iii) With the aid of a simple schematic, explain what covalent bonding is and how it is formed. What are the differences between a covalent bond and an ionic bond? Which type of bond is found in crystalline silicon? At room temperature do you expect an ionic crystal to be a good electrical conductor or a good electrical insulator? Explain why.arrow_forward
- Write a balanced chemical equation for the reaction of an excess of oxygen with each of the following. Remember that oxygen is a strong oxidizing agent and tends to oxidize an element to its maximum oxidation state.(a) Mg(b) Rb(c) Ga(d) C2H2(e) COarrow_forwardWhich of the following statements are true?(a) Si can form an ion with six fluorine atoms, SiF62-,whereas carbon cannot.(b) Si can form three stable compounds containing two Siatoms each, Si2H2, Si2H4, and Si2H6.(c) In HNO3 and H3PO4 the central atoms, N and P, havedifferent oxidation states.(d) S is more electronegative than Se.arrow_forwardQ.5(a) The alkali metals follow the noble gases in their atomic structure. What properties of these metals can be predicted from this information? (b) Arrange the carbonates of alkaline earth metals in order of thermal stability. (c) Explain the process involved in the manufacture of NaOH, Na2CO3 and NaHCO3. (d) Identify the element X in each of the following: (i) The oxide of XO2 has a high M.P., and is very abundant in nature. (ii) X forms three oxides: XO, XO2, X3O2. (iii) X forms compounds mainly in the +2 O.S., though some compounds in +4 state do exist. (iv) X occurs as several allotropes, including a molecular one. (e) Borazine reacts with three mole equivalents of HCl to give a material with chemical composition B3N3H9C13. (i) What is the structure of product? (ii) How does the isoelectronic benzene react with HCl? (f) Explain why bond length in NO (115 pm) is longer than that in nitrosonium ion (106 pm)?arrow_forward
- The elements sodium, aluminum, and chlorine are in the same period.(a) Which has the greatest electronegativity?(b) Which of the atoms is smallest?(c) Write the Lewis structure for the simplest covalent compound that can form between aluminum and chlorine.(d) Will the oxide of each element be acidic, basic, or amphoteric?arrow_forwardWrite a balanced equation for (a) the combination reaction between lithium metal and fluorine gas?arrow_forwardWrite a balanced equation for the reaction of elemental strontium with each of the following:(a) oxygen(b) hydrogen bromide(c) hydrogen(d) phosphorus(e) waterarrow_forward
 Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax
Chemistry by OpenStax (2015-05-04)ChemistryISBN:9781938168390Author:Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark BlaserPublisher:OpenStax Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning





