
Concept explainers
Draw the products formed when phenol
a.
b.
c.
d.
(a)
Interpretation: The products formed by the treatment of phenol with given reagents are to be drawn.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.36P
The products formed by the treatment of phenol with given reagents are shown below:
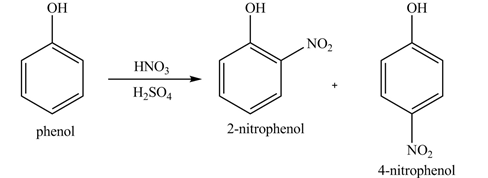
Explanation of Solution
Phenol undergoes nitration reaction on treatment with

Figure 1
The products formed by the treatment of phenol with given reagents are shown in Figure 1.
(b)
Interpretation: The products formed by the treatment of phenol with given reagents are to be drawn.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.36P
The products formed by the treatment of phenol with given reagents are shown below:
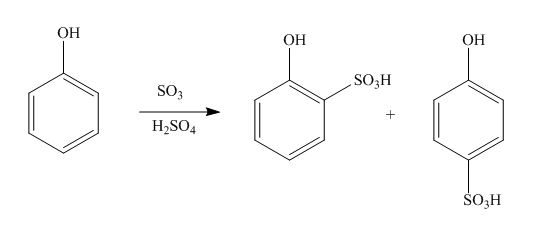
Explanation of Solution
An aromatic compound undergoes sulphonation on reaction with

Figure 2
The products formed by the treatment of phenol with given reagents are shown in Figure 2.
(c)
Interpretation: The products formed by the treatment of phenol with given reagents are to be drawn.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.36P
The products formed by the treatment of phenol with given reagents are shown below:
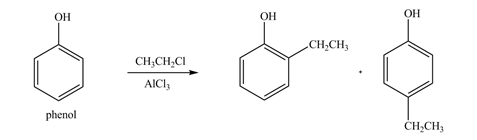
Explanation of Solution
An aromatic compound undergoes Friedel-Craft alkylation on reaction with

Figure 3
The products formed by the treatment of phenol with given reagents are shown in Figure 3.
(d)
Interpretation: The products formed by the treatment of phenol with given reagents are to be drawn.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.36P
The products formed by the treatment of phenol with given reagents are shown below:
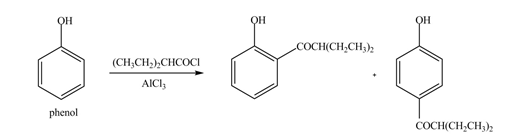
Explanation of Solution
An aromatic compound undergoes Friedel-Craft acylation on reaction with

Figure 4
The products formed by the treatment of phenol with given reagents are shown in Figure 4.
(e)
Interpretation: The products formed by the treatment of phenol with given reagents are to be drawn.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.36P
The products formed by the treatment of phenol with given reagents are shown below:
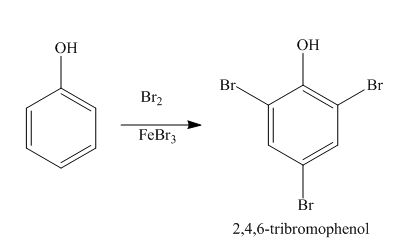
Explanation of Solution
The benzene ring is activated in phenol due to the presence of strong electron donating group. In bromination, bromonium acts as electrophile and produces

Figure 5
The products formed by the treatment of phenol with given reagents are shown in Figure 5.
(f)
Interpretation: The products formed by the treatment of phenol with given reagents are to be drawn.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.36P
The products formed by the treatment of phenol with given reagents are shown below:
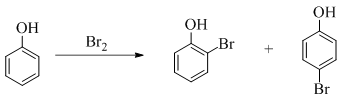
Explanation of Solution
Phenol undergoes mono bromination with bromine without catalyst. The corresponding chemical reaction is shown below.

Figure 6
The products formed by the treatment of phenol with given reagents are shown in Figure 6.
(g)
Interpretation: The products formed by the treatment of phenol with given reagents are to be drawn.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.36P
The products formed by the treatment of phenol with given reagents are shown below:
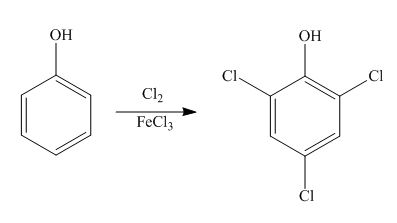
Explanation of Solution
The benzene ring is activated in phenol due to the presence of strong electron donating group. In chlorination, chloronium acts as electrophile and produces

Figure 7
The products formed by the treatment of phenol with given reagents are shown in Figure 7.
(h)
Interpretation: The products formed by the treatment of phenol with given reagents are to be drawn.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.36P
The products formed by the treatment of phenol with given reagents are shown below:
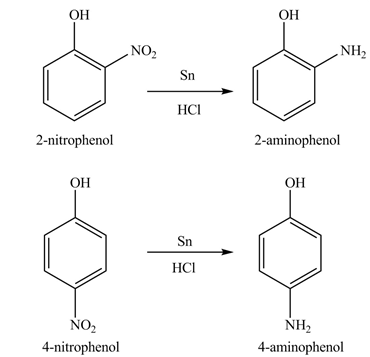
Explanation of Solution
The

Figure 8
The products formed by the treatment of phenol with given reagents are shown in Figure 8.
(i)
Interpretation: The products formed by the treatment of phenol with given reagents are to be drawn.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.36P
The products formed by the treatment of phenol with given reagents are shown below:
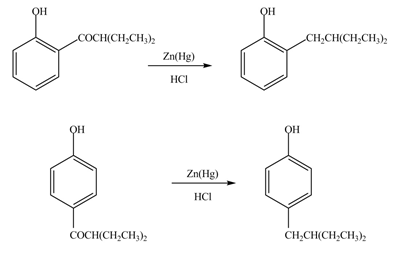
Explanation of Solution
The carbonyl group

Figure 9
The products formed by the treatment of phenol with given reagents are shown in Figure 9.
(j)
Interpretation: The products formed by the treatment of phenol with given reagents are to be drawn.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.36P
The products formed by the treatment of phenol with given reagents are shown below:
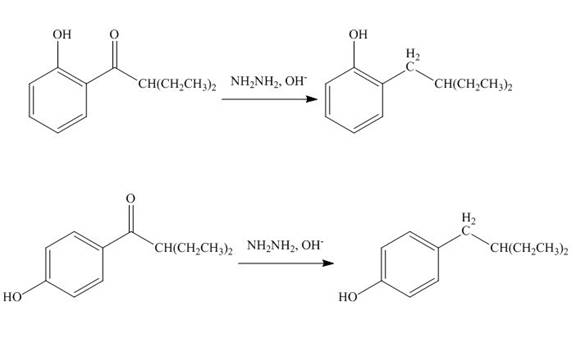
Explanation of Solution
The carbonyl group
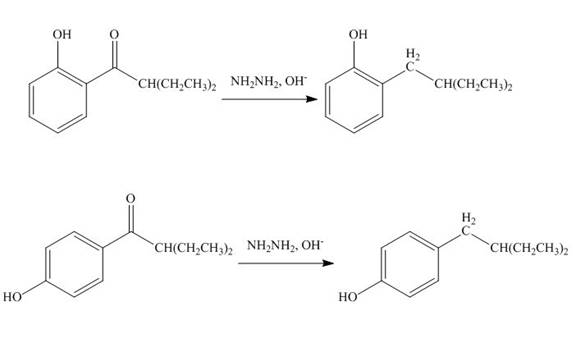
Figure 10
The products formed by the treatment of phenol with given reagents are shown in Figure 10.
(k)
Interpretation: The products formed by the treatment of phenol with given reagents are to be drawn.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.36P
The products formed by the treatment of phenol with given reagents are shown below:
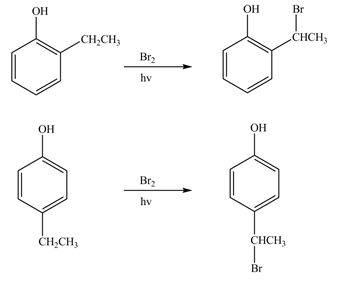
Explanation of Solution
The bromine group attaches to the side chain of the aromatic compound on reaction with

Figure 11
The products formed by the treatment of phenol with given reagents are shown in Figure 11.
(l)
Interpretation: The products formed by the treatment of phenol with given reagents are to be drawn.
Concept introduction: The replacement or substitution of one functional group with another different functional group in any chemical reaction is termed as substitution reaction. The electron deficient chemical species that contains positive charge are known as electrophile. In electrophilic aromatic substitution reaction, electrophile takes the position of hydrogen atom by attacking the electron rich carbon atom of benzene.
Answer to Problem 18.36P
The products formed by the treatment of phenol with given reagents are shown below:
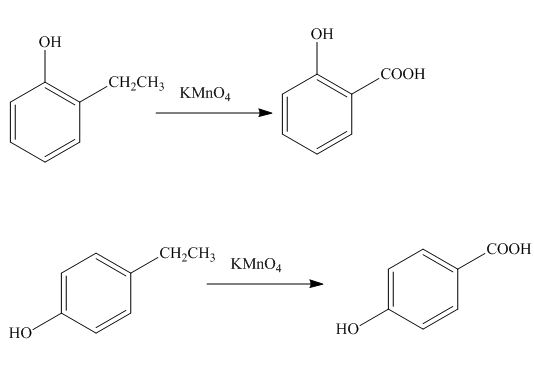
Explanation of Solution
The alkyl group oxidizes to

Figure 12
The products formed by the treatment of phenol with given reagents are shown in Figure 12.
Want to see more full solutions like this?
Chapter 18 Solutions
Organic Chemistry
- 0 NaBH₁ Ethanol A. Product A B. Product B C. Product C D. Product D E. Product E major product A OH B OH BH₁ FFF HO D HO E 12:42 AMarrow_forwardDraw the products of each reaction. NANH2 NaOH KNH2 b. CH,0- -CI a. NH3 H20 NH3 Aarrow_forwardShow how to bring about each conversion in good yield. a. b. C6H5 Cl OH COOH C6H5 COOHarrow_forward
- What is the product of the following reaction? 1 a. b. C. d. e. BH₂ 3 only 1 only 5 only 4 only 2 only 2 NaBH4 OH then HCI 3 ? gonد 5 OHarrow_forwardWhat is the product of this reaction? 1) Hg(OAc), H2o 2. NABH4 -OPr A. OH B. C. D. HOHarrow_forward6S. What is the major product of this reaction? 65. NaOH a C d.arrow_forward
- What is the major product of this reaction? 1 equiv HCI -80 °C 身arrow_forward1. Which two reactions will give the same major product? 1. 3. OH CH3OH A. 1 and 2 H* 2. CH3OH 1. NaH B. 1 and 3 Br C. 1 and 4 2. D. 2 and 3 4. Br CH3Br E. 2 and 4 H+ CH3Br F. 3 and 4arrow_forwardChoose the correct product for the given reaction. CF3 HBr ? CF3 CF3 Br A. Br Br CF3 Br CF3 D. Br С. Br A B B. ооо оarrow_forward
- What is the best choice as the major product for this reaction? NaCN ų th OH CH2 a. b. OH он NH d. O a O b Oc O d earrow_forwardWhat is the major product of the following reaction? 1. LDA. THE 2. CHC(O)H 3.11,0 II IV %3Darrow_forward4. OH Write reagents and product(s) for the each reaction. HBr ? Brarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





