
Concept explainers
(a)
Interpretation:
The Haworth projection for given molecule is to be drawn.
Concept introduction:
Structural drawings of carbohydrates in cyclic forms are called Haworth formulas. The
Answer to Problem 18.73P
The Haworth projection of
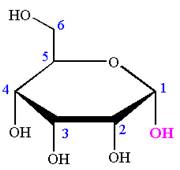
Explanation of Solution
The given molecule is
The structure of
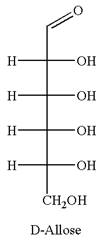
The name
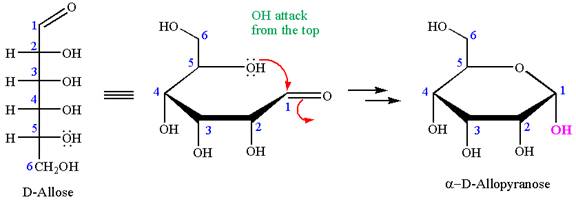
The Haworth projection of
(b)
Interpretation:
The Haworth projection for given molecule is to be drawn.
Concept introduction:
Structural drawings of carbohydrates in cyclic forms are called Haworth formulas. The
Answer to Problem 18.73P
The Haworth projection of
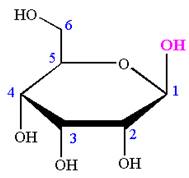
Explanation of Solution
The given molecule is
The structure of
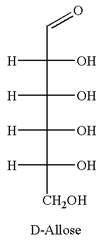
The name
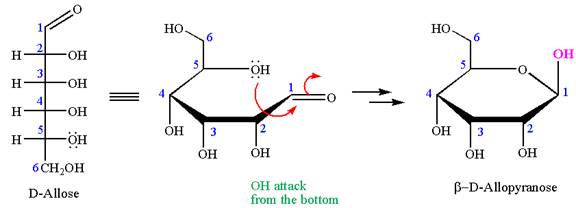
The Haworth projection of
(c)
Interpretation:
The Haworth projection for given molecule is to be drawn.
Concept introduction:
Structural drawings of carbohydrates in cyclic forms are called Haworth formulas. The
Answer to Problem 18.73P
The Haworth projection of
Explanation of Solution
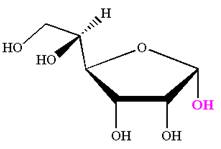
The given molecule is
The structure of
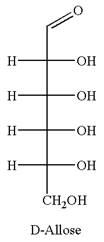
The name
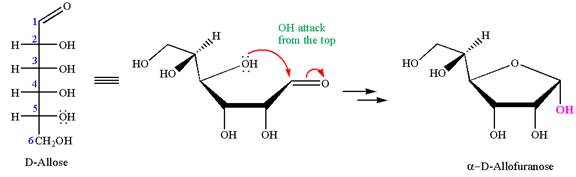
The Haworth projection of
(d)
Interpretation:
The Haworth projection for given molecule is to be drawn.
Concept introduction:
Structural drawings of carbohydrates in cyclic forms are called Haworth formulas. The
Answer to Problem 18.73P
The Haworth projection of
Explanation of Solution
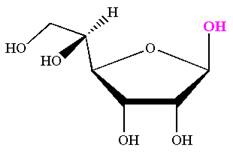
The given molecule is
The structure of

The name
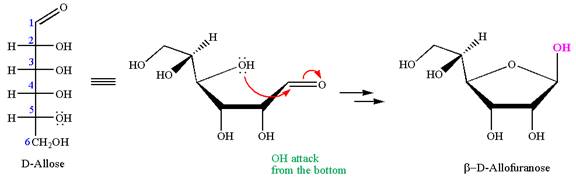
The Haworth projection of
Want to see more full solutions like this?
Chapter 18 Solutions
Organic Chemistry: Principles and Mechanisms (Second Edition)
- (a) Which compounds (B–F) are identical to A? (b) Which compounds (B–F) represent an isomer of A?arrow_forwardCan someone please label the H1, H2 etc into its respective regions (ie. aliphatic and aromatic regions)? Thank you!arrow_forwardFor each pair of compounds, a state which compound is the better SN2 substrate. (a) 2-bromobutane or isopropyl bromidearrow_forward
- Draw the structure of each of the following molecules. (a) (R)-1-chloro-1-fluorobutane; (b) (S)-2-chloropentane; (c) (R)-2-chloro-2-methoxypentane;(d) (R)-2,2,3-trichlorobutane; (e) (S)-3-methylhexane; (f) (S)-2-bromo-1-nitropentanearrow_forwardDraw the structure of each molecule. (a) (E)-4-carbamoylbut-3-enoic acid; (b) 3-carbamoylpentanediamide; (c) 4,6-dioxohexanenitrile; (d) (1S,2S)-2-methoxycyclopent-3-ene-1-carbonitrile; (e) cyclohexa-3,6-diene-1,3-dicarboxylic acidarrow_forwardCan you please answer what the missing compound A and B are?arrow_forward
- Identify A, B, C, and D in the preceding problem if D is oxidized to an optically inactive aldaric acid; if A, B, and C are oxidized to optically active aldaric acids; and if interchanging the aldehyde and alcohol groups of A leads to a different sugar.arrow_forwardThe saccharide shown here is present in some plant-derived foods. (a) Indicate the anomeric carbon atom(s) in this structure by drawing a circle around the atom(s) or by drawing an arrow pointing to the atom(s). (b) Would this saccharide give a positive result with Benedict’s reagent? Why or why not? (c) Would this saccharide give a positive result with Barfoed’s reagent? Why or why not? (d) Would this saccharide give a positive result with Seliwanoff’s reagent? Why or why not? (e) In a separate set of experiments, the saccharide solution was treated with a reagent that breaks glycosidic bonds. After this treatment, would any of the three assays give different results? Be sure to indicate which assay results would be different and give a reason.arrow_forwardPossesses a tautomer in equilibrium..... Benzaldehyde Benzophenone Both Neitherarrow_forward
- Draw the complete, detailed SN1 mechanism for each of the following reactions. (a) (b) CI -Br CH3OH -OCH3 Br NaCIarrow_forwardPlease help answer all part of the following question..thank you! Provide the reagent(s) needed to accomplish the following transformations. Some conversions may require more than one step. If more than one step is required, be sure to enumerate each step.arrow_forwardMenthone and menthol are both isolated from mint. Explain why mentholis a solid at room temperature but menthone is a liquid. (see attached)arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





