
(a)
Interpretation:
The main group elements on the periodic table has to be located.
Concept introduction:
Main group elements
Main group elements are
(a)
Explanation of Solution
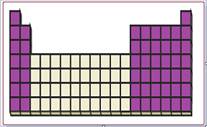
Figure 1
The location of s and p-block elements in the periodic table are shown using violet colored boxes in the above given figure 1.
(b)
Interpretation:
The
Concept introduction:
The s-block elements are (group 1) alkali metals and (group 2) alkaline earth metals.
(b)
Explanation of Solution
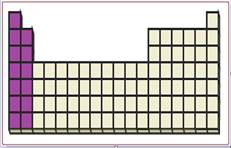
Figure 2
The s-block elements are (group 1) alkali metals and (group 2) alkaline earth metals. S-block elements have valence electrons in the
The location of s-block elements in the periodic table are shown using violet colored boxes in the above given figure 2.
(c)
Interpretation:
The
Concept introduction:
The p-block elements are groups
(c)
Explanation of Solution
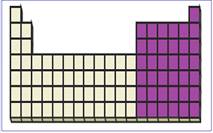
Figure 3
P-block elements have valence electrons in the
The location of p-block elements in the periodic table are shown using violet colored boxes in the above given figure 3.
(d)
Interpretation:
The main group metal elements on the periodic table has to be located.
Concept introduction:
Main group metals all are solid except mercury which is a liquid
Most have a silvery shine, good conductors of heat and electricity.
Relatively low ionization energies.
Relatively low electronegative.
(d)
Explanation of Solution
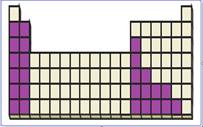
Figure 4
The location of main group metals in the periodic table are shown using violet colored boxes in the above given figure 1.
(e)
Interpretation:
The non-metal elements on the periodic table has to be located.
Concept introduction:
Non-metals all are gases at
Most lack a metallic luster
Relatively high ionization energies.
Relatively high electronegative.
Gain electrons to form anions, share electrons to form oxyanions
(e)
Explanation of Solution
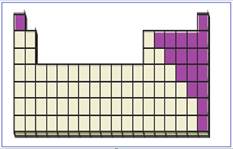
Figure 5
The location of main group non-metals in the periodic table are shown using violet colored boxes in the above given figure 5.
(f)
Interpretation:
The semimetals elements on the periodic table has to be located.
Concept introduction:
Carbon is a non-metal, silicon and germanium are semimetals, and tin and lead are metals. The horizontal and vertical periodic trends combine to locate the element with the most metallic character in the lower left of the periodic table, the element with the most non-metallic character in the upper right, and the semimetals along the diagonal stair step that stretches across the middle.
(f)
Explanation of Solution
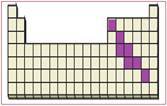
Figure 6
The location of semimetals in the periodic table are shown using violet colored boxes in the above given figure 6.
Want to see more full solutions like this?
Chapter 19 Solutions
General Chemistry: Atoms First
- 4. This question concerns the d-block elements, Sulphur, Nitrogen and Group IV elements,. (a) (i) Distinguish between a d-block element and a transition metal. (ii) Write down the symbols for the elements of the first transition series in increasing order of atomic number. (iii) State the property of the transition element which enables them to act as catalyst in many industrial processes.arrow_forwardGive TWO reasons why aluminium metal was not commonly used till last century, even though it is one of the most common elements on earth. (b) Why are copper and lithium such important elements today?arrow_forwardUntil the early 1960s the group 8A elements were called the inert gases; before that they were called the rare gases. The term rare gases was dropped after it was discovered that argon accounts for roughly 1% of Earth’s atmosphere. (a) Why was the term inert gases dropped? (b) What discovery triggered this change in name? (c)What name is applied to the group now?arrow_forward
- Which element is a row 5 transition element?(a) Sr(b) Pd(c) P(d) Varrow_forwardElement X is a metal with a valency 2. Element Y is a non-metal with a valency 3 (a) Write equations to show how X and Y form ions. (b) If Y is a diatomic gas, write the equation for the direct combination of X and Y to form a compound.arrow_forwardIn general terms, how does each of the following atomic properties influence the metallic character of themain-group elements in a period?(a) Ionization energy(b) Atomic radius(c) Number of outer electrons(d) Effective nuclear chargearrow_forward
- In general terms, how does each of the following atomic properties influence the metallic character of the main-group elements in a period?(a) Ionization energy(b) Atomic radius(c) Number of outer electrons(d) Effective nuclear chargearrow_forwardUse an orbital diagram to describe the electron configuration of the valence shell of each of the followingatoms:(a) N(b) Si(c) Fe(d) Te(e) Moarrow_forwardAccording to general trends in the periodic table, predict which element in each of the following pairs has the larger atomic radius. (a) Rb or Sr (b) As or Se (c) Pb or Bi (d) I or Xearrow_forward
- Explain how the following properties change across the periodic Table from group 1 to group 2, and give reasons for the increase or decrease thereof:d)Melting pointe)Electropositive characterarrow_forwardThe oxidation state of chromium in CrAsO4 is: (a) 0 (b) +1 (c) +2 (d) +3 (e) -3arrow_forward(a) Why is calcium generally more reactive than magnesium?(b) Why is calcium generally less reactive than potassium?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





