
Concept explainers
(a)
Interpretation:
The product formed in the reaction of butyraldehyde and
Concept introduction:
Grignard reagents are
Answer to Problem 19.41AP
The product formed in the reaction of butyraldehyde and
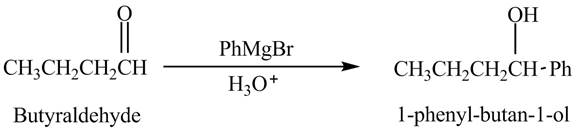
Explanation of Solution
Butyraldehyde reacts with
The reduction of butyraldehyde takes place to form the product. The reaction is shown below.

Figure 1
The product formed in the reaction of butyraldehyde and
(b)
Interpretation:
The product formed in the reaction of butyraldehyde and
Concept introduction:
The gain of electrons involved in the particular reaction is termed as reduction. The reduction process also involves the removal of an electronegative atom from a molecule followed by the hydrogen atom addition into that molecule.
Answer to Problem 19.41AP
The product formed in the reaction of butyraldehyde and
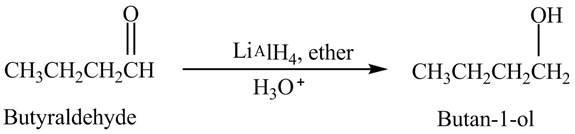
Explanation of Solution
Butyraldehyde reacts with
The reduction of butyraldehyde takes place to form the product. The reaction is shown below.
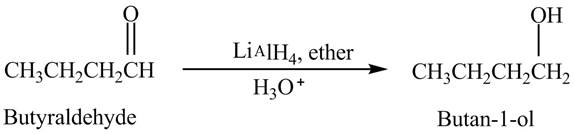
Figure 2
The product formed in the reaction of butyraldehyde and
(c)
Interpretation:
The product formed in the reaction of butyraldehyde and alkaline
Concept introduction:
Oxidation is the process of addition of oxygen. Oxidation is also defined as loss of electrons. In the oxidation process, there is increase in the oxidation state. The acid is added in the reaction for protonation.
The oxidizing agent is defined as the species which oxidizes others and itself gets reduced.
Answer to Problem 19.41AP
The product formed in the reaction of butyraldehyde and
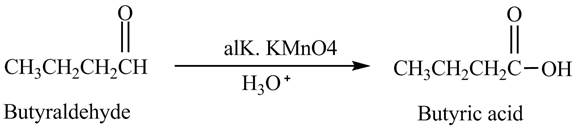
Explanation of Solution
Butyraldehyde reacts with
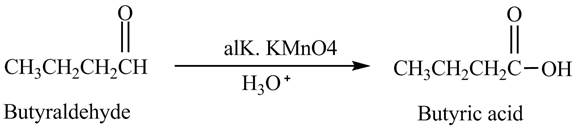
Figure 3
The product formed in the reaction of butyraldehyde and alkaline
(d)
Interpretation:
The product formed in the reaction of butyraldehyde and aqueous
Concept introduction:
Oxidation is the process of addition of oxygen. Oxidation is also defined as loss of electrons. In the oxidation process, there is increase in the oxidation state. The acid is added in the reaction for protonation.
The oxidizing agent is defined as the species which oxidizes others and itself gets reduced.
Answer to Problem 19.41AP
The product formed in the reaction of butyraldehyde and aqueous
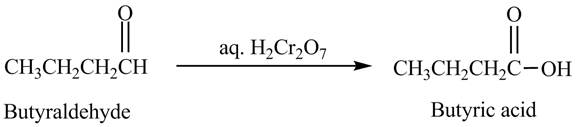
Explanation of Solution
Butyraldehyde reacts with aqueous
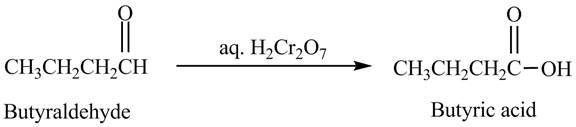
Figure 4
The product formed in the reaction of butyraldehyde and aqueous
(e)
Interpretation:
The product formed in the reaction of butyraldehyde and
Concept introduction:
Oxime belongs to the family of imines. The formula of oxime is
Answer to Problem 19.41AP
The product formed in the reaction of butyraldehyde and
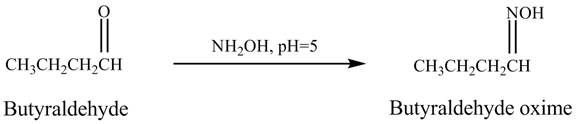
Explanation of Solution
Butyraldehyde reacts with ![]() . When the
. When the ![]() is high then the product formed is an
is high then the product formed is an ![]() is low then the product is not formed. The reaction is shown below.
is low then the product is not formed. The reaction is shown below.
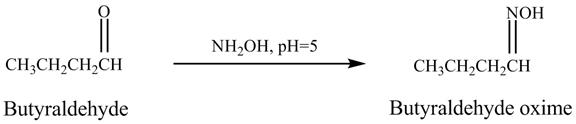
Figure 5
The product formed in the reaction of butyraldehyde and
(f)
Interpretation:
The product formed in the reaction of butyraldehyde and
Concept introduction:
Oxidation is the process of addition of oxygen. Oxidation is also defined as loss of electrons. In the oxidation process, there is increase in the oxidation state. The acid is added in the reaction for protonation.
The oxidizing agent is defined as the species which oxidizes others and itself gets reduced.
Answer to Problem 19.41AP
The product formed in the reaction of butyraldehyde and
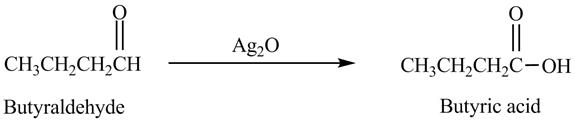
Explanation of Solution
Butyraldehyde reacts with
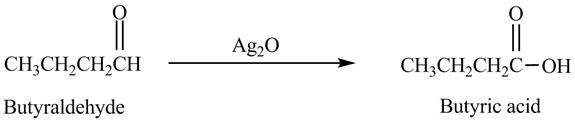
Figure 6
The product formed in the reaction of butyraldehyde and
(g)
Interpretation:
The product formed in the reaction of butyraldehyde and zinc amalgam in the presence of
Concept introduction:
Clemmensen reduction is defined as the reduction in which aldehydes or
Answer to Problem 19.41AP
The product formed in the reaction of butyraldehyde and zinc amalgam in the presence of
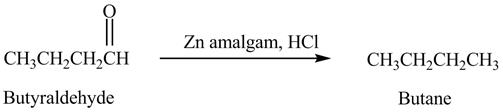
Explanation of Solution
Butyraldehyde reacts with zinc amalgam in the presence of

Figure 7
The product formed in the reaction of butyraldehyde and zinc amalgam in the presence of
(h)
Interpretation:
The product formed in the reaction of butyraldehyde and
Concept introduction:
The Wittig reaction is the
Answer to Problem 19.41AP
The geometrical isomers are formed in the reaction of butyraldehyde and
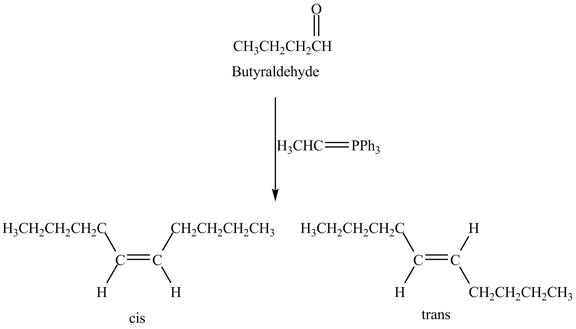
Explanation of Solution
Butyraldehyde reacts with
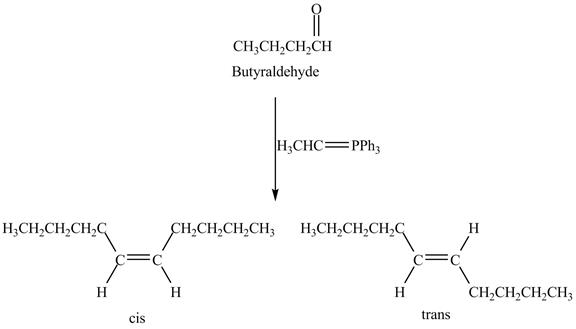
Figure 8
Butyraldehyde reacts with
Want to see more full solutions like this?
Chapter 19 Solutions
Organic Chemistry
- Write the products of the following acid-base reactions: (a) CH3OH + H2SO4 ² ? (b) CH3OH + NANH2 2 ? (c) CH3NH3+ Cl- + NaOH ?arrow_forward(a) Draw the three isomers of benzenedicarboxylic acid.(b) The isomers have melting points of 210 °C, 343 °C, and 427 °C. Nitration of the isomers at all possible positions was once used to determine their structures. The isomer that melts at 210 °C gives two mononitro isomers. The isomer that melts at 343 °C gives three mononitro isomers. The isomer that melts at 427 °C gives only one mononitro isomer. Show which isomer has which melting point.arrow_forward(a) Explain how NaBH, in CH;OH can reduce hemiacetal A to 1,4-butanediol (HOCH,CH,CH,CH,OH). (b) What product is formed when A is treated with Ph;P=CHCH,CH(CH),? (c) The drug isotretinoin is formed by reaction of X and Y. What is the structure of isotretinoin? Although isotretinoin (trade name Accutane or Roaccutane) is used for the treatment of severe acne, it is dispensed under strict controls because it also causes birth defects. PPha NaOCH,CH3 HO- isotretinoin HO A Br X Yarrow_forward
- Write the equilibrium-constant expressions and obtain numerical values for each constant in (a) the basic dissociation of aniline, C6H5NH2. (b) the acidic dissociation of hypochlorous acid, HClO. (c) the acidic dissociation of methyl ammonium hydrochloride, CH3NH3Cl. (d) the basic dissociation of NaNO2. (e) the dissociation of H3AsO3 to H3O+ and AsO33- just answer the letters C, D and E.arrow_forwardChemical Equilibrium Write the equilibrium-constant expressions and obtain numerical values for each constant in (a) the basic dissociation of aniline, C6H5NH2. (b) the acidic dissociation of hypochlorous acid, HClO. (c) the acidic dissociation of methyl ammonium hydrochloride, CH3NH3Cl. (d) the basic dissociation of NaNO2. (e) the dissociation of H3AsO3 to H3O+ and AsO33- Using step-by-step processarrow_forwardChlorination of 2-butanone yields two isomeric products, each having the molecular formula C4H7ClO. (a) What are these two compounds? (b) Write structural formulas for the enol intermediates that lead to each of these compounds. (c) Using curved arrows, show the flow of electrons in the reaction of each of the enols with Cl2.arrow_forward
- (b) 3-methyl-2-butanol reacts with concentrated sulphuric acid to form 2-methyl-2- butene. Write the mechanism for the reaction.arrow_forwardConsider the following structural formulas. OH -осн A в D E (a) Which of the compounds A-E can easily be oxidized? (b) Provide the typical oxidation products for all compounds selected in (a). (c) Explain why the compounds not selected in (a) cannot be easily oxidized.arrow_forwardIdentify (A) in the following reaction. 2H2 Pt (A) KMNO4 Warm conc. || С — С — о—н |CO,H + HO CO2H cis-cyclo hexane 1,2-dicarboxylic acid (a) (b) (c) (d)arrow_forward
- (b) A compound with the molecular formula C4H9CI has four structural isomers. Write the structural formulas for all the four isomers. (c) Arrange the following compounds in order of increasing acidity, and explain the reasons forarrow_forwardUsing the data in Appendix C, determine which of the following bases is strong enough to deprotonate acetonitrile (CH3CN), so that equilibrium favors the products: (a) NaH; (b) Na2CO3; (c) NaOH; (d) NaNH2; (e) NaHCO3.arrow_forwardBiphenyl has the following structure.(a) Is biphenyl a (fused) polynuclear aromatic hydrocarbon?(b) How many pi electrons are there in the two aromatic rings of biphenyl? How does this number compare with that for naphthalene?(c) The heat of hydrogenation for biphenyl is about 418 kJ>mol (100 kcal>mol). Calculate theresonance energy of biphenyl.(d) Compare the resonance energy of biphenyl with that of naphthalene and with that of two benzene rings. Explain thedifference in the resonance energies of naphthalene and biphenyl.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





