
Concept explainers
Carvone is the major constituent of spearmint oil. What products would you expect from reaction of carvone with the following reagents?
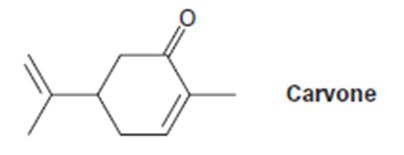
a) (CH3)2CU-Li+, then H3O+
b) LiA1H4, then H3O+
c) CH3NH2
d) C6H5MgBr, then H3O+
e) H2/Pd
f) CrO3, H3O+
g) (C6H5)3 P+C- HCH3
h) HOCH2CH2OH, HC1
a)
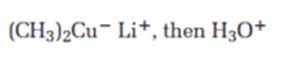
Interpretation:
The product expected when carvone reacts first with (CH3)2Cu- Li+ and then with H3O+ is to be stated.
Concept introduction:
α, β- Unsaturated ketones undergo conjugate (1,4-addition) addition reaction when treated with lithium diorganocopper reagents to yield saturated ketones as the product. Any primary, secondary, tertiary alkyl, alkenyl, and aryl halides can be used to prepare the lithium diorganocopper reagents.
To state:
The product expected when carvone reacts first with (CH3)2Cu- Li+ and then with H3O+.
Answer to Problem 64AP
The product expected when carvone reacts first with (CH3)2Cu- Li+ and then with H3O+ is
Explanation of Solution
Carvone is an α, β- unsaturated ketone. When treated with (CH3)2Cu- Li+ it undergoes a conjugate addition to yield the corresponding saturated ketone.
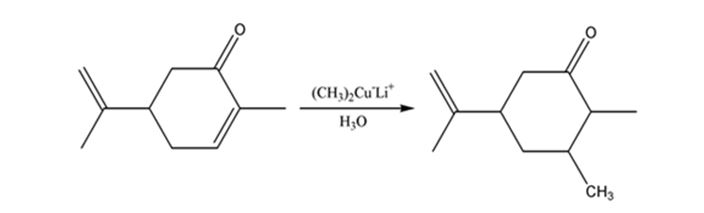
The product expected when carvone reacts first with (CH3)2Cu- Li+ and then with H3O+ is

b)
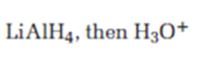
Interpretation:
The product expected when carvone reacts first with LiAlH4 and then with H3O+ is to be stated.
Concept introduction:
Unsaturated ketones can be reduced to secondary alcohols without affecting the double bond by treating with LiAlH4 and then with H3O+.
To state:
The product expected when carvone reacts first with LiAlH4 and then with H3O+.
Answer to Problem 64AP
The product expected when carvone reacts first with LiAlH4 and then with H3O+ is
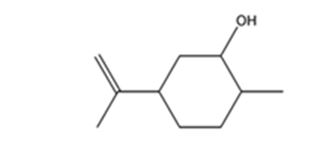
Explanation of Solution
Carvone is an α, β- unsaturated ketone. LiAlH4 reduces it to a secondary alcohol. The double bond remains unaffected during the reduction.
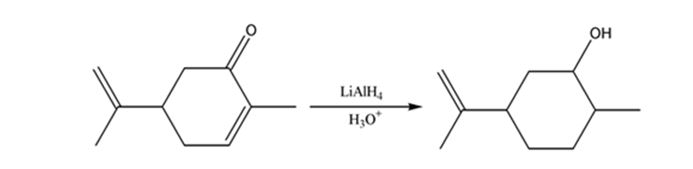
The product expected when carvone reacts first with LiAlH4 and then with H3O+ is

c)
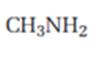
Interpretation:
The product expected when carvone reacts with CH3NH2 is to be stated.
Concept introduction:
Both primary and secondary amines react with α, β- unsaturated ketones to yield β-aminoaldehydes and ketones by conjugate addition.
To state:
The product expected when carvone reacts with CH3NH2.
Answer to Problem 64AP
The product expected when carvone reacts with CH3NH2 is
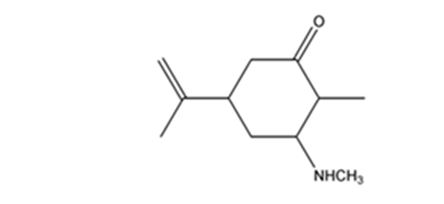
Explanation of Solution
Carvone is an α, β- unsaturated ketone. When treated with methyl amine it undergoes conjugate addition to yield the β-N-methylamino ketone as the product.
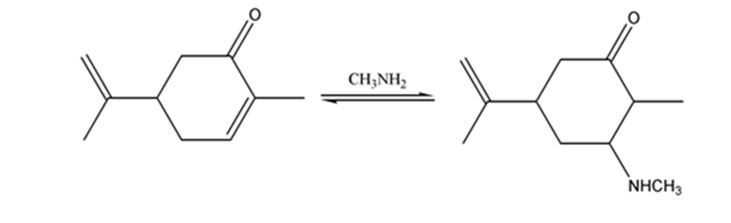
The product expected when carvone reacts with CH3NH2 is

d)

Interpretation:
The product expected when carvone reacts first with C6H5MgBr and then with H3O+ is to be stated.
Concept introduction:
Ketones undergo a 1,2-addition reaction with Grignard reagent to give an intermediate which upon acidification yields tertiary alcohols.
To show:
The product expected when carvone reacts first with C6H5MgBr and then with H3O+.
Answer to Problem 64AP
The product expected when carvone reacts first with C6H5MgBr and then with H3O+ is
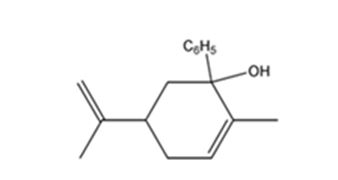
Explanation of Solution
Carvone is an α, β- unsaturated ketone. When treated with C6H5MgBr, the Grignard reagent adds to the carbonyl group to yield an intermediate which when treated with aqueous acids yield the tertiary alcohol required.
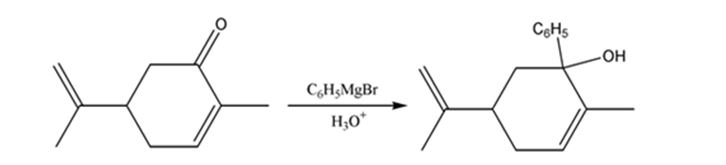
The product expected when carvone reacts first with C6H5MgBr and then with H3O+ is

e)
Interpretation:
The product expected when carvone reacts with H2/Pd is to be stated.
Concept introduction:
Hydrogen in the presence of Pd reduces the double and triple bonds in organic molecules. It does not reduce the carbonyl group in aldehydes and ketones.
To state:
The product expected when carvone reacts with H2/Pd.
Answer to Problem 64AP
The product expected when carvone reacts with H2/Pd is
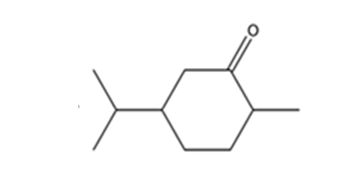
Explanation of Solution
Carvone is an α, β- unsaturated ketone. When treated with hydrogen in the presence of Pd the double bond in it is reduced and the C= O group in it remains unaffected.
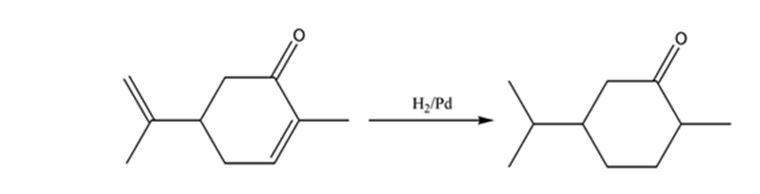
The product expected when carvone reacts with H2/Pd is

f)
Interpretation:
The product expected when carvone reacts with CrO3, H3O+ is to be stated.
Concept introduction:
Ketones are not oxidized by CrO3, H3O+.
To state:
The product expected when carvone reacts with CrO3, H3O+.
Answer to Problem 64AP
Carvone does not reacts with CrO3, H3O+.
Explanation of Solution
Carvone is an α, β- unsaturated ketone. It is not oxidized by CrO3, H3O+.
Carvone does not reacts with CrO3, H3O+.
g)
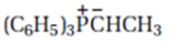
Interpretation:
The product expected when carvone reacts with (C6H5)3P+CH- CH3 is to be stated.
Concept introduction:
When ketones are treated with phosphoranes, an exchange the O in the ketone and alkyl group in phosphorane takes place to yield an alkene and phosphorus oxide as the products.
To state:
The product expected when carvone reacts with (C6H5)3P+CH- CH3.
Answer to Problem 64AP
The product expected when carvone reacts with (C6H5)3P+CH- CH3 is
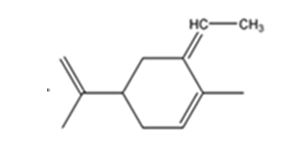
The compound exists as E and Z isomers.
Explanation of Solution
The given reaction is a Wittig reaction. When the ketone, carvone, is treated with the phosphorane, an exchange of oxygen in it and alkyl group in phosphorane takes place to yield an alkene as the product. The alkene exhibits geometrical isomerism and exists as E and Z isomers.
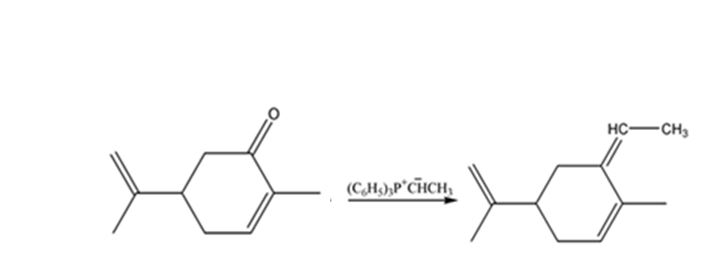
The product expected when carvone reacts with (C6H5)3P+CH- CH3 is

The compound exists as E and Z isomers.
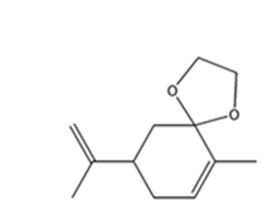
h)
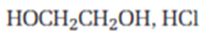
Interpretation:
The product expected when carvone reacts with HOCH2CH2OH is to be stated.
Concept introduction:
Aldehydes and ketones, when treated with dihydric alcohols in the presence of acid catalyst yield cyclic acetals as products.
To state:
The product expected when carvone reacts with HOCH2CH2OH.
Answer to Problem 64AP
The product expected when carvone reacts with HOCH2CH2OH is
Explanation of Solution
Carvone is an α, β- unsaturated ketone. Hence when treated with glycol, a dihydric alcohol, in the presence of HCl it yields a cyclic acetal as product.
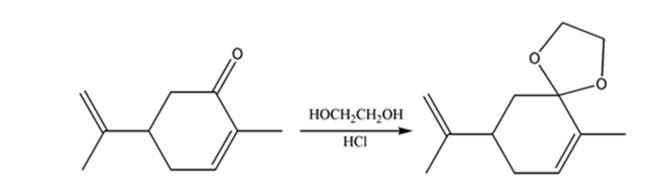
The product expected when carvone reacts with HOCH2CH2OH is

Want to see more full solutions like this?
Chapter 19 Solutions
Organic Chemistry
- Nitriles, R–=C≡N, undergo a hydrolysis reaction when heated with aqueous acid. What is the structure of the compound produced by hydrolysis of propanenitrile, CH3CH2C≡N, if it has IR absorptions from 2500–3100 cm-1 and at 1710 cm-1, and has M+=74?arrow_forwardOne frequently used method for preparing methyl esters is by reaction of carboxylic acids with diazomethane, CH2N2. The reaction occurs in two steps: (l) protonation of diazomethane by the carboxylic acid to yield methyldiazonium ion, CH3N2+, plus a carboxylate ion; and (2) reaction of the carboxylate ion with CH3N2+. (a) Draw two resonance structures of diazomethane, and account for step 1. (b) What kind of reaction occurs in step 2?arrow_forwardGive the expected major product of the following reaction: OH 1) LIAIH,arrow_forward
- Synthesize the following compound from benzonitrile (C6H5CN):arrow_forwardProvide synthetic routes for the preparation of the starting materials of azo dye Alizarine Yellow R starting from benzene O₂N -NEN OH CO₂Harrow_forwardOne compound that contributes to the “seashore smell” at beaches in Hawai‘i is dictyopterene D', a component of a brown edible seaweed called limu lipoa. Hydrogenation of dictyopterene D' with excess H2 in the presence of a Pd catalyst forms butylcycloheptane. Ozonolysis with O3 followed by (CH3)2S forms CH2(CHO)2, HCOCH2CH(CHO)2, and CH3CH2CHO. What are possible structures of dictyopterene D'?arrow_forward
- Propose a synthesis for the systemic agricultural fungicide tridemorph from dodecanoic acid (lauric acid), propene, and a one-carbon building block. How many stereoisomers are possible for tridemorph? CH3 →CH3(CH2)10COOH + CH3CH=CH, H3C (CH2) 12CH3 Tridemorph Dodecanoic acid Propene (Lauric acid)arrow_forward5. Compound A, C 10H 18O, undergoes reaction with dilute H 2SO 4 at 50 °C to yield a mixture of two alkenes, C 10H 16. The major alkene B, gives only cyclopentanone after ozone treatment followed by reduction with zinc in acetic acid. Which of the following reactions are correct.arrow_forwardDraw the structure of the organic product formed when 2,3-butanedione reacts with the following reagents. CH3 -C-C-CH3 | (a)H2/Ni (b)l2/NAOH 3Darrow_forward
- 4. Compound A has the formula C 8H 8. It reacts rapidly with KMnO 4 to give CO 2 and a carboxylic acid, B (C 7H 6O 2), but reacts with only 1 molar equivalent of H 2 on catalytic hydrogenation over a palladium catalyst. On hydrogenation under conditions that reduce aromatic rings, 4, equivalents of H 2 are taken up and hydrocarbon C (C 8H 16) is produced. What are the structures of A, B, and C.arrow_forward1. How would you prepare the following alkyl halides from the appropriate alcohols? (a) ÇI (b) Br CH3 CH3CH3 CH;CHCH,CHCH3 ČH3 2. Predict the products of the following reaction: (a) он CH3 (b) H2C CH;CH,CHCH,CHCH, Per, socl2. PBr3 ? H3Carrow_forwardProvide the major product for the following reaction? (1) BH3, ether (2) H2O2, OHarrow_forward
