
Pushing Electrons
4th Edition
ISBN: 9781133951889
Author: Weeks, Daniel P.
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 2, Problem 18EQ
The cyclohexane carboxylate anion has a Lewis structure
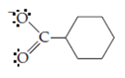
Pushing a pair of unshared electrons away from the negatively charged oxygen atom and, at the same time, pushing a pair of pi electrons toward the other oxygen will generate a second resonance structure. Thus,

Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
- Draw Lewis structures for each of the following. Give the total number of valence electrons,
select from the lists the number of Regions of Electron Density (REDs) around the central atom,
the molecular shape, and the bond angles.
- Do not put covalent bonds between metals and nonmetals.
- Put brackets around anions to show both charge and quantity.
Shapes
tetrahedral
pyramidal
bent
trigonal planar
linear
diatomic
monatomic ions
Bond Types
nonpolar covalent
polar covalent
nonpolar & polar covalent
ionic
ionic & covalent
Type of molecule
Nonpolar
Polar
Ionic
5. C2Cl2
valence e-1 =
shape=
type of bonds =
type of molecule =
6. AICI3
valence e-1 for one Cl-1 =
shape=
type of bonds =
type of molecule =
7. K3PO3
valence e-1 =
shape=
type of bonds =
type of molecule =
8. KH2PO3
valence e-1 =
shape at the P=
type of bonds =
type of molecule =
dicarbon dichloride
aluminum chloride
potassium phosphite
potassium dihydrogen phosphite
- Draw Lewis structures for each of the following. Give the total number of valence electrons,
select from the lists the number of Regions of Electron Density (REDs) around the central atom,
the molecular shape, and the bond angles.
- Do not put covalent bonds between metals and nonmetals.
- Put brackets around anions to show both charge and quantity.
Shapes
tetrahedral
pyramidal
bent
trigonal planar
linear
diatomic
monatomic ions
Bond Types
nonpolar covalent
polar covalent
nonpolar & polar covalent
ionic
ionic & covalent
Type of molecule
Nonpolar
Polar
lonic
1. HCC13
valence e-1 =
shape=
type of bonds =
type of molecule =
2. SCI2
valence e-1 =
shape=
type of bonds =
type of molecule =
3. P204
valence e-1 =
shape=
type of bonds =
type of molecule =
4. N2O4
valence e-1 =
shape=
type of bonds =
type of molecule =
hydrogen carbon trichloride
sulfur dichloride
diphosphorus tetroxide
dinitrogen tetroxide
A resonance hybrid is a structure that can be depicted by more than one valid Lewis structure.
part1: Draw the major resonance form of fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized, and it should include all nonzero formal charges and all nonbonding electrons.
part2: Draw the second most important resonance form of fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized, and it should include all nonzero formal charges and all nonbonding electrons.
part3: Draw the least important resonance contributor for fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized and should include all nonzero formal charges and all nonbonding electrons.
Chapter 2 Solutions
Pushing Electrons
Ch. 2 - One Lewis structure for the 2-butenyl cation is...Ch. 2 - Prob. 2EQCh. 2 - One structure for the conjugate acid of acetone...Ch. 2 - Similarly, a resonance structure for the conjugate...Ch. 2 - Prob. 5EQCh. 2 - Pairs of unshared electrons can be pushed. One...Ch. 2 - One structure for the acetoxonium ion is Clearly,...Ch. 2 - Prob. 8EQCh. 2 - There are no important resonance structures for...Ch. 2 - Prob. 10EQ
Ch. 2 - Prob. 11EQCh. 2 - Prob. 12EQCh. 2 - Prob. 13EQCh. 2 - Prob. 14EQCh. 2 - Prob. 15EQCh. 2 - Prob. 16EQCh. 2 - Prob. 17EQCh. 2 - The cyclohexane carboxylate anion has a Lewis...Ch. 2 - One Lewis structure for the enolate anion of...Ch. 2 - Prob. 20EQCh. 2 - Prob. 21EQCh. 2 - Prob. 22EQCh. 2 - Prob. 23EQCh. 2 - Prob. 24EQCh. 2 - Prob. 25EQCh. 2 - Prob. 26EQCh. 2 - Prob. 27EQCh. 2 - Prob. 28EQ
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- One Lewis structure for the enolate anion of acetaldehyde is Pushing the pair of unshared electrons on the carbon atom away from the center of negative charge and pushing the pi electrons of the carbon-oxygen double bond to the oxygen atom generates a second resonance structure. Thus,arrow_forwardThe Lewis structure of acetone is Circling the carbonyl carbon, i.e., the carbon atom attached to oxygen, and its octet gives Circling the oxygen atom and its octet gives Thus, atoms share electrons in making bonds, and a pair of electrons may be included in the octet of two different atoms. When computing the formal charge on an atom, the number of electrons that belong to that atom is compared with the number of electrons the atom would have in the unbonded and neutral state. If the two numbers are the same, the formal charge on the atom is zero. In a Lewis structure both electrons in an unshared pair belong to the atom, and one of every pair of shared (bonding) electrons belongs to the atom.arrow_forwardA resonance hybrid is a structure that can be depicted by more than one valid Lewis structure. part1: Draw the major resonance form of fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized, and it should include all nonzero formal charges and all nonbonding electrons. part2: Draw the second most important resonance form of fulminic acid, HCNO, with the atoms connected as indicated in the formula. Your structure should have nonzero formal charges minimized, and it should include all nonzero formal charges and all nonbonding electrons.arrow_forward
- Draw the Lewis dot structure of the conjugate base of cyclopentane by assuming all the atoms in the molecule obey the octet rule. Show all lone pairs and formal charges. Do no draw resonance structures.arrow_forwardPLEASE NOTE: This question has been answered, but DID NOT INCLUDE THE “DOTS” for the Lewis-dot structure. I'm needing someone to ADD THE “DOTS” and hand-draw, please. I am completely and totally lost with how to do these!!!! For the following molecules, provide ALL possible resonance structures. Calculate the formal charge for each atom in the molecule. Determine which structure is the major contributing structure for the resonance hybrid. Explain WHY it is preferred over the others. Remember your rules for setting up Lewis-dot structures. a) HCO2- b) SO3 c) Cl2CO d) OCN-arrow_forwardWhich of these atoms cannot serve as a central atom in a Lewis Structure? Sulfur, Helium, Fluorine, Hydrogen, Gallium Explain.arrow_forward
- In molecules of the type X-O-H, as the electronegativity of X increases, the acid strength increases. In addition, if the electronegativity of X is a very small value, the molecule acts as a base. Explain these observations and provide examples.arrow_forwardUnshared, or lone, electron pairs play an important role in determining the chemical and physical properties of organic compounds. Thus, it is important to know which atoms carry unshared pairs. Use the structural formulas below to determine the number of unshared pairs at each designated atom. Be sure your answers are consistent with the formal charges on the formulas. The number of unshared pairs at atom a is 4 The number of unshared pairs at atom b is 2 H The number of unshared pairs at atom c is 3 The number of unshared pairs at atom a is The number of unshared pairs at atom b is C-NEN а b H The number of unshared pairs at atom c is Submit Answer Retry Entire Group 8 more group attempts remainingarrow_forwardAnswer the questions in the table below about the shape of the phosphorus pentabromide (PBr,) molecule. How many electron groups are around the central phosphorus atom? Note: one "electron group" means one lone pair, one single bond, one double bond, or one triple bond. What phrase best describes the arrangement of these electron groups around the central phosphorus atom? (You may need to use the scrollbar to see all the choices.) (choose one) Ar Explanation Check © 2021 McGraw Hill LLC. AlI Rights Reserved. Terms of Use Privacy Center | Accessibilityarrow_forward
- Carbon, nitrogen, and oxygen form two different polyatomic ions: cyanate ion (NCO) and fulminate ion (CNO). Write Lewis structures for each anion, including near-equivalent resonance structures (do not add any arrows between structures) and indicating formal charges. The isocyanate ion also has two near-equivalent structures, but the formal charge on the nitrogen attom cannot be reduced to zero: Cyanate ion (NCO)arrow_forwardA compound composed of sulfur and fluorine has an empirical formula of SF. The structure of this compound has a formal charge of zero on all elements in the compound. Draw the Lewis structure for this substance. (Assign lone pairs and radical electrons where appropriate.)arrow_forwardProvide the Lewis structure and name of this moleculesarrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning


Chemistry: The Molecular Science
Chemistry
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:Cengage Learning
INTRODUCTION TO MOLECULAR QUANTUM MECHANICS -Valence bond theory - 1; Author: AGK Chemistry;https://www.youtube.com/watch?v=U8kPBPqDIwM;License: Standard YouTube License, CC-BY