
Interpretation:
The
Concept introduction:
Chromatography is the technique used to identify the components of a mixture. The division of the distance covered by the component of a compound to the distance covered by solvent in the chromatography technique is known as the
Answer to Problem 1ASA
The
Explanation of Solution
The chromatograph for a mixture that is prepared by a student is shown below.

Figure 1
The modified chromatogram with proper divided parts is shown below.
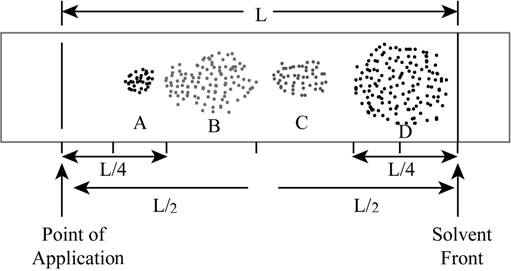
Figure 2
According to the above shown chromatogram, four compounds A, B, C and D are present in the mixture.
The total distance travelled by the solvent is
The total distance travelled by the compound A is
The total distance travelled by the compound B is
The total distance travelled by the compound C is
The total distance travelled by the compound D is
The expression to represent the
To calculate the
To calculate the
To calculate the
To calculate the
Therefore, the
The
Want to see more full solutions like this?
Chapter 2 Solutions
Chemical Principles in the Laboratory
- In molecular exclusion (size exclusion) chromatography, the molecules have the longest retention times. a) the larger molecules b) the smaller moleculesarrow_forwardWhat is the best Chromatography instrument for analysis of Soluble Protein molecules CG-FID GC-MS LC-MS IC-MS O IC-ECarrow_forwardIn all forms of chromatography, there is a mobile phase and a stationary phase. In thin layer chromatography what is the stationary phase?arrow_forward
- what is chromatography? Explain different types of chromatography and explain how to seperate compounds. indicate stationary phase mobile phase.arrow_forwardQno1: why there is a need of chromatography and chromatography thecniqs. B) when and where we should use normal and reverse phase chromatographyarrow_forwardA chemist was able to extract a biologically active compound and wants to purify the sample since he is sure that the sample still contains impurities. After running the extract in TLC, the chemist found out that the sample produced only one spot near the solvent front. Suggest how the chemist can achieve a better resolution for the chromatogram.arrow_forward
- Use a suitable model to explain how separation and identification of a mixture of organic compounds can be achieved with a thin layer chromatographic (TLC) technique.arrow_forwardPreparing a Thin Layer Chromatography experiment there are some common mistakes. Explain what happens in this situation: Sample spots too bigarrow_forwardPaper Chromatography and TLC are examples of planar chromatography. True or FalsE?arrow_forward
- What is the Rf value given the following information: Height of Chromatography Paper: 5.0 cm Distance from Bottom of Paper to Origin: 1.5 cm Distance from Origin to Spot: 1.6 cm Distance from Origin to Solvent Front: 2.9 cm Your Answer: Answerarrow_forwardthin layer chromatography: What would happen to the Rf value of a compound if you increased the polarity of the mobile phase while using a polar stationary phase? please help me this question! Thank you a lotarrow_forwardQUESTION 5 In a chromatographic separation using a polar stationary phase and a mobile phase composed of 60% hexane and 40% ethyl acetate, two compounds, benzene (peak A) and toluene (peak B), are eluted. The width of peak A at its base is 1.2 min, the width of peak B at its base is 1.3 min, and the retention time of peak A is 5.2 min and peak B is 3.5 min. What is the resolution (Rs) between these two peaks and is it acceptable? R = 1.36. It is not an acceptable resolution. R = 0.74. It is an acceptable resolution. R = 1.36. It is an acceptable resolution. R = 0.74. It is not an acceptable resolution.arrow_forward
 Chemical Principles in the LaboratoryChemistryISBN:9781305264434Author:Emil Slowinski, Wayne C. Wolsey, Robert RossiPublisher:Brooks Cole
Chemical Principles in the LaboratoryChemistryISBN:9781305264434Author:Emil Slowinski, Wayne C. Wolsey, Robert RossiPublisher:Brooks Cole Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT
EBK A SMALL SCALE APPROACH TO ORGANIC LChemistryISBN:9781305446021Author:LampmanPublisher:CENGAGE LEARNING - CONSIGNMENT


