
Concept explainers
Fill the blanks in the following table.
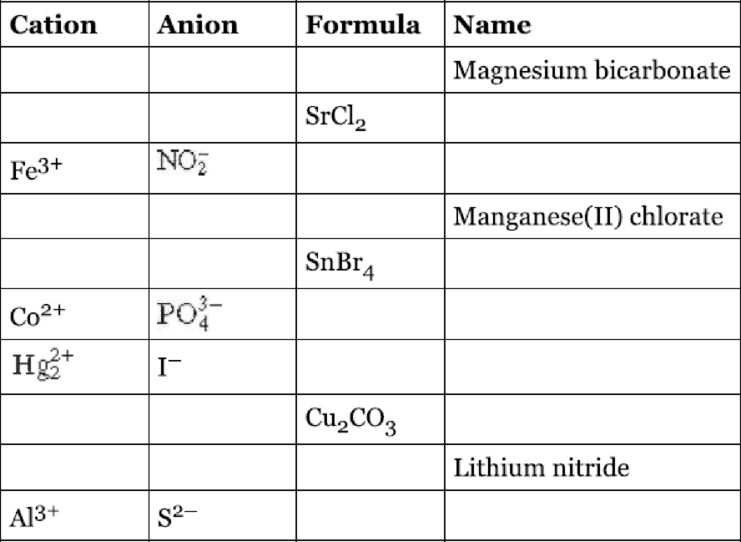
Interpretation:
The given table has to be filled.
Explanation of Solution
The given name is Magnesium bicarbonate. The cation and anion present in the compound are
The given formula is
The cation and anion present in the compound is
The given name is Manganese
The given formula is
The cation and anion present in the compound are
The cation and anion present in the compound are
The given formula is
The given name is Lithium nitride. The cation and anion present in the compound are
The cation and anion present in the compound are
The completed table is shown below,
| Cation | Anion | Formula | Name |
| Magnesium bicarbonate | |||
| Strontium chloride | |||
| Iron | |||
| Manganese | |||
| Tin | |||
| Cobalt | |||
| Mercury | |||
| Copper | |||
| Lithium nitride | |||
| Aluminum sulphide |
Want to see more full solutions like this?
Chapter 2 Solutions
Chemistry
- Imagine that you have discovered several stable isotopes of a new element, which you call element X. The measured isotope masses and abundances are given in the table below. Percent Abundance Mass (amu) 353.1165 354.1330 356.3666 Isotope 353X 354X 356X 10.909% 41.304% 47.787% What is the average atomic mass of this new element X? Provide at least 5 significant figures (2 decimal places) in your answer. Show all of your work and calculations in the answer box below. You will not get full credit for this question if you do not show your work. For the toolbar, press ALT+F10 (PC) or ALT+FN+F10 (Mac). R T U f Paragraph ✓ Arial 10pt Y A Oarrow_forwardComplete the following table by filling in the blanks in each row. The first row has been completed as an example.arrow_forwardFill in the blanks in each line in the following table. The first line is already completed as an example.arrow_forward
- Which of the following is true about an individual atom? Explain. a. An individual atom should be considered to be a solid. b.An individual atom should be considered to be a liquid. c. An individual atom should be considered to be a gas. d. The state of the atom depends on which element it is. e. An individual atom cannot be considered to be a solid, liquid, or gas. Justify your choice, and for choices you did not pick, explain what is wrong with them.arrow_forward7.101 Lead selenide nanocrystals may provide a breakthrough in the engineering of solar panels to be efficient enough to be an economical source of electricity. Selenium is generally considered a nonmetal while lead is considered a metal. Is this distinction enough to suggest that this compound should be ionic? Explain your answer.arrow_forwardTwo samples of different compounds of nitrogen and oxygen have the following compositions. Show that the compounds follow the law of multiple proportions. What is the ratio of oxygen in the two compounds for a fixed amount of nitrogen? Amount N Amount O Compound A 1.206 g 2.755 g Compound B 1.651 g 4.714 garrow_forward
- The isotope of an unknown element, X, has a mass number of 79. The most stable ion of the isotope has 36 electrons and forms a binary compound with sodium, having a formula of Na2X. Which of the following statements is(are) true? For the false statements, correct them. a. The binary compound formed between X and fluorine will be a covalent compound. b. The isotope of X contains 38 protons. c. The isotope of X contains 41 neutrons. d. The identity of X is strontium, Sr.arrow_forwardTwo samples of different compounds of sulfur and oxygen have the following compositions. Show that the compounds follow the law of multiple proportions. What is the ratio of oxygen in the two compounds for a fixed amount of sulfur? Amount S Amount O Compound A l.210g 1.811 g Compound B 1.783 g 1.779 garrow_forwardThe element europium exists in nature as two isotopes: 151Eu has a mass of 150.9196 amu, and 153Eu has a mass of 152.9209 amu. The average atomic mass of europium is 151.96 amu. a. Calculate the relative abundance of the two europium isotopes. b. Graph each fractional abundance value as a y-axis value in association with its corresponding mass value on the x-axis. Starting from each x-axis value, where y = 0, draw a vertical line up to the fractional abundance value. The result will approximate the type of visual graph a mass spectrometer would yield for europium in the 150155 amu range.arrow_forward
 General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning
General, Organic, and Biological ChemistryChemistryISBN:9781285853918Author:H. Stephen StokerPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning
Introduction to General, Organic and BiochemistryChemistryISBN:9781285869759Author:Frederick A. Bettelheim, William H. Brown, Mary K. Campbell, Shawn O. Farrell, Omar TorresPublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning
General Chemistry - Standalone book (MindTap Cour...ChemistryISBN:9781305580343Author:Steven D. Gammon, Ebbing, Darrell Ebbing, Steven D., Darrell; Gammon, Darrell Ebbing; Steven D. Gammon, Darrell D.; Gammon, Ebbing; Steven D. Gammon; DarrellPublisher:Cengage Learning





