
(a)
Interpretation:
Synthesis of
Concept introduction:
Synthesis is the process of conversion of one compound into another using reagents. For the conversion of one compound into another, multiple number of steps undergoes using multiple reagents.
Answer to Problem 20.47AP
Synthesis of
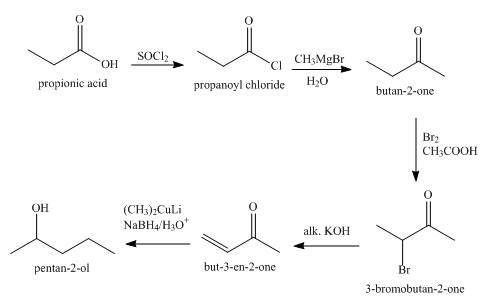
Explanation of Solution
The synthesis of

Figure 1
The total number of steps in the synthesis are five. In the first step propanoic acid is converted into acid chloride which is converted into
The ketone
Synthesis of
(b)
Interpretation:
Synthesis of the given compound from allyl alcohol using appropriate reagents is to be stated.
Concept introduction:
Synthesis is the process of conversion of one compound into another using reagents. For the conversion of one compound into another, the multiple numbers of steps undergo using multiple reagents.
Answer to Problem 20.47AP
Synthesis of the given compound from allyl alcohol using appropriate reagents is shown below.
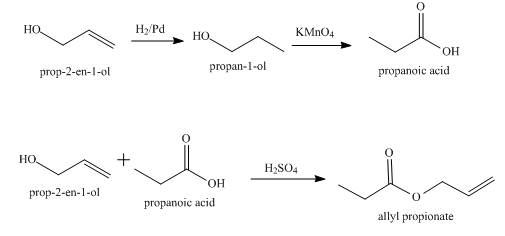
Explanation of Solution
The given compound to be synthesized is shown below.
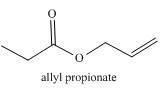
Figure 2
The synthesis of the given compound from allyl alcohol is done using multiple steps shown below.

Figure 3
The allyl alcohol,
Synthesis of the given compound from allyl alcohol using appropriate reagents is shown in Figure 3.
(c)
Interpretation:
Synthesis of
Concept introduction:
Synthesis is the process of conversion of one compound into another using reagents. For the conversion of one compound into another, the multiple numbers of steps undergo using multiple reagents.
Answer to Problem 20.47AP
Synthesis of
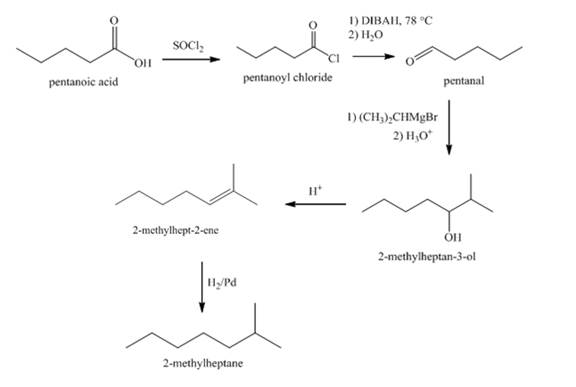
Explanation of Solution
The synthesis of

Figure 4
The conversion of pentanoic acid to acid chloride is done by using thionyl chloride. The acid chloride thus produced is reacted with DIBAH at
Synthesis of
(d)
Interpretation:
Synthesis of
Concept introduction:
Synthesis is the process of conversion of one compound into another using reagents. For the conversion of one compound into another, the multiple numbers of steps undergo using multiple reagents.
Answer to Problem 20.47AP
Synthesis of
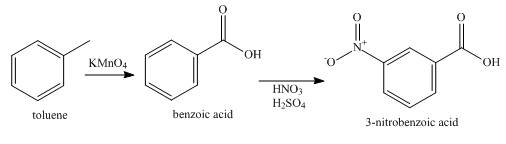
Explanation of Solution
The synthesis of

Figure 5
Toluene is first oxidized using potassium permanganate to give benzoic acid. A carboxylic acid group is a meta-directing group. The benzoic acid is then nitrated to give
Synthesis of
(e)
Interpretation:
Synthesis of
Concept introduction:
Synthesis is the process of conversion of one compound into another using reagents. For the conversion of one compound into another, the multiple numbers of steps undergo using multiple reagents.
Answer to Problem 20.47AP
Synthesis of
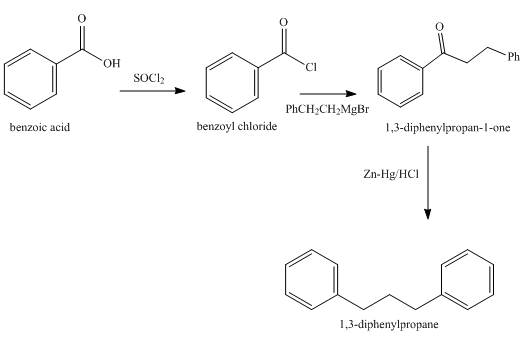
Explanation of Solution
The synthesis of

Figure 6
Benzoic acid is converted into benzoyl chloride in the first step which is then reacted with Grignard’s reagent to give an appropriate ketone which on Clemmensen reduction gives the required product
Synthesis of
(f)
Interpretation:
Synthesis of given
Concept introduction:
Synthesis is the process of conversion of one compound into another using reagents. For the conversion of one compound into another, the multiple numbers of steps undergo using multiple reagents.
Answer to Problem 20.47AP
Synthesis of given
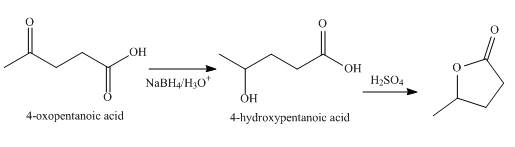
Explanation of Solution
The synthesized compound is
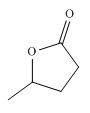
Figure 7
Synthesis of given

Figure 8
The
Synthesis of given
(g)
Interpretation:
Synthesis of given dicarboxylic acid from norbornene using appropriate reagents is to be stated.
Concept introduction:
Synthesis is the process of conversion of one compound into another using reagents. For the conversion of one compound into another, the multiple numbers of steps undergo using multiple reagents.
Answer to Problem 20.47AP
Synthesis of given dicarboxylic acid from norbornene using appropriate reagents is shown below.
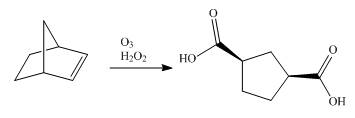
Explanation of Solution
The given dicarboxylic acid is shown below.
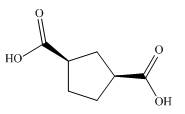
Figure 9
Synthesis of given dicarboxylic acid from norbornene using appropriate reagents is done in a single step shown below.

Figure 10
The conversion of norbornene into the required dicarboxylic acid is done in a single reaction. The ozonolysis of norbornene with oxidative work gives the required product.
Synthesis of given dicarboxylic acid from norbornene using appropriate reagents is shown in Figure 10.
(h)
Interpretation:
Synthesis of
Concept introduction:
Synthesis is the process of conversion of one compound into another using reagents. For the conversion of one compound into another, the multiple numbers of steps undergo using multiple reagents.
Answer to Problem 20.47AP
Synthesis of
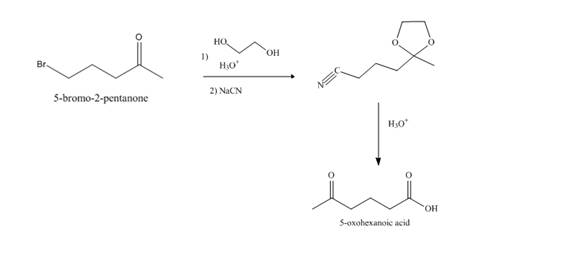
Explanation of Solution
Synthesis of

Figure 11
The compound,
Synthesis of
(i)
Interpretation:
Synthesis of
Concept introduction:
Synthesis is the process of conversion of one compound into another using reagents. For the conversion of one compound into another, the multiple numbers of steps undergo using multiple reagents.
Answer to Problem 20.47AP
Synthesis of
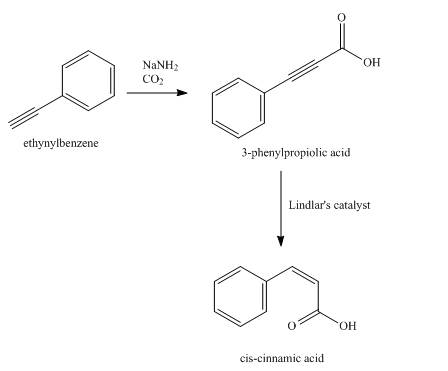
Explanation of Solution
Synthesis of

Figure 12
The compound,
Synthesis of
Want to see more full solutions like this?
Chapter 20 Solutions
Organic Chemistry
- (a) A hydrocarbon isolated from fish oil and from plankton was identified as 2,6,10,14-tetramethyl-2-pentadecene. Write its structure.(b) Alkyl isothiocyanates are compounds of the type RN C S. Write a structural formula for allyl isothiocyanate, a pungent-smelling compound isolated from mustard.(c) Grandisol is one component of the sex attractant of the boll weevil. Write a structural formula for grandisol given that R in the structure shown is an isopropenyl group.arrow_forwardDimethyl disulfide, CH,S–SCH3, found in the vaginal secretions of female hamsters, acts as a sexual attractant for the male hamster. Write an equation for its synthesis from methanethiol.arrow_forward(b) Propose a synthesis of (2,2-dimethylpropyl)benzene from benzene.arrow_forward
- Explain why each of the following carboxylic acids cannot be prepared by a malonic ester synthesis: (a) (CH3CCH;COOH; (b) C,H;CH2COOH; (c) (CHa)CCOOH.arrow_forwardElectrophilic aromatic substitution usually occurs at the 1-position of naphthalene, also called the a position. Predict the major products of the reactions of naphthalene with the following reagents.(a) isobutylene and HF (b) cyclohexanol and BF3 (c) fuming sulfuric acidarrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l). The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 8.50 gof butanoic acid and excess ethanol? Express your answer in grams to three significant figures.arrow_forward
- Ethyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) a) Given 7.70 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100% yield? b) A chemist ran the reaction and obtained 5.25 g of ethyl butyrate. What was the percent yield? c) The chemist discovers a more efficient catalyst that can produce ethyl butyrate with a 78.0% yield. How many grams would be produced from 7.70 g of butanoic acid and excess ethanol?arrow_forwardEthyl butyrate, CH3CH2CH2CO2CH2CH3, is an artificial fruit flavor commonly used in the food industry for such flavors as orange and pineapple. Its fragrance and taste are often associated with fresh orange juice, and thus it is most commonly used as orange flavoring.It can be produced by the reaction of butanoic acid with ethanol in the presence of an acid catalyst (H+): CH3CH2CH2CO2H(l)+CH2CH3OH(l)H+⟶CH3CH2CH2CO2CH2CH3(l)+H2O(l) Given 8.50 g of butanoic acid and excess ethanol, how many grams of ethyl butyrate would be synthesized, assuming a complete 100%yield? Express your answer in grams to three significant figures.arrow_forwardPredict the products of the following acid-base reactions. If the equilibrium would not result in the formation of appreciable amounts of products, you should so indicate. In each case label the stronger acid, the stronger base, the weaker acid, and the weaker base: (a) CH3CH=CH2 + NANH2 (d) CH3C=C: + CH;CH2OH → (e) CH3C=C:- + NH¾CI – | (b) CH;C=CH + NaNH2 (c) CH3CH2CH3 + NANH2 → | HASarrow_forward
- Give reasons for the following: (i) p-nitrophenol is more acidic than p-methylphenol. (ii) Bond length of C—O bond in phenol is shorter than that in methanol. (iii) (CH3)3C—Br on reaction with sodium methoxide (Na+ _OCH3) gives alkene as the main product and not an ether.arrow_forwardHow could you convert each of the following compounds into butanoic acid? Write each step showing all reagents. (a) 1-Butanol (b) 1-Bromobutane (c) 1-Butenearrow_forward(a) Tsomane and Nyiko were given a task of synthesising methylenecyclohexane 2. After a brief discussion with each other, Tsomane proposed Method A to synthesise 2 from cyclohexanone 1 while Nyiko proposed Method B that started from hydroxymethylcyclohexane 3. Each student believed that their proposed method is better than the other. (Scheme below) (1) 1 Ph THF A Ph Ph B H₂SO4 100 °C 3 OH What is the name of the reaction that is followed by reaction Method A?arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





