
Concept explainers
Which of the molecules and ions given in Problem 21.15 are aromatic according to the Hückel criteria? Which, if planar, would be antiaromatic?
21.15 State the number of 2p orbital electrons in each molecule or ion.
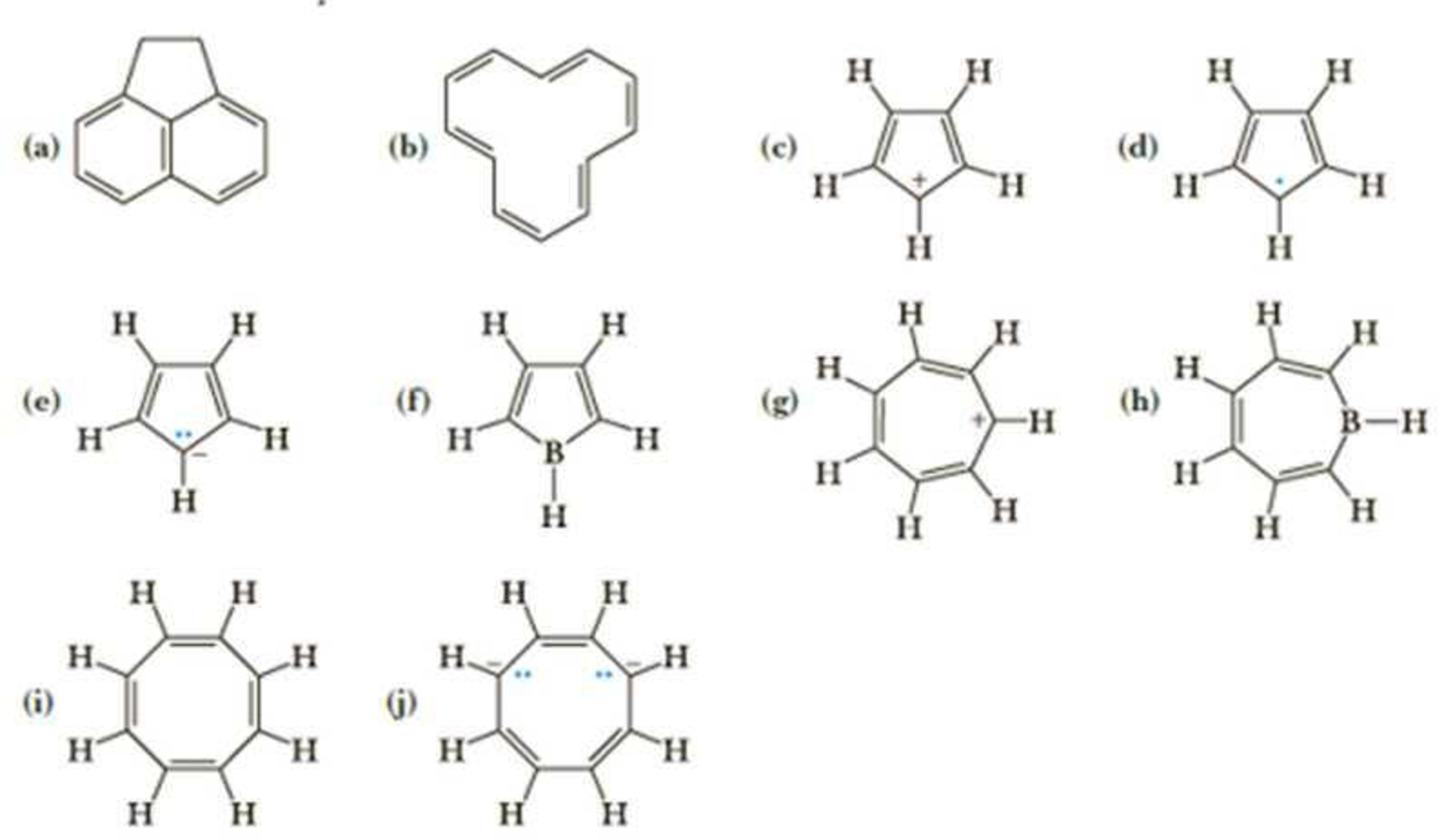
(a)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
The given compound is shown here:
There are five
Huckel’s rule of aromaticity
(b)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
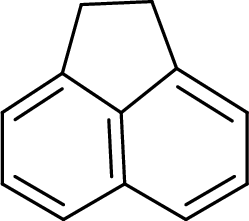
The given compound is shown here:
There are six
Huckel’s rule of aromaticity
The given compound is planar. So, it is expected to possess aromaticity due to the expected effective delocalization of
(c)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
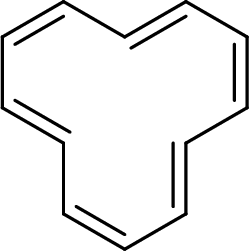
The given compound is shown here:
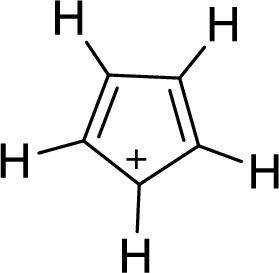
There are two
Huckel’s rule of aromaticity
The given compound is planar. So, it is expected to possess aromaticity due to the expected effective delocalization of
(d)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
The given compound is shown here:
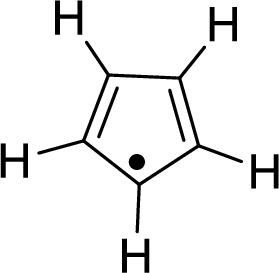
There are two
Huckel’s rule of aromaticity
(e)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
The given compound is shown here:
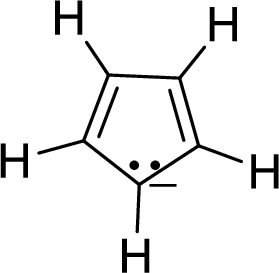
There are two
Huckel’s rule of aromaticity
(f)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
The given compound is shown here:
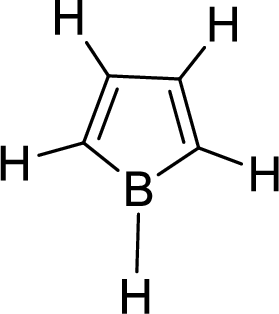
There are two
Huckel’s rule of aromaticity
The given compound is planar. So, it is expected to possess aromaticity due to the expected effective delocalization of
(g)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
The given compound is shown here:
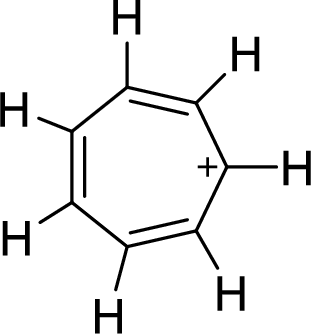
There are three
Huckel’s rule of aromaticity
(h)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
The given compound is shown here:
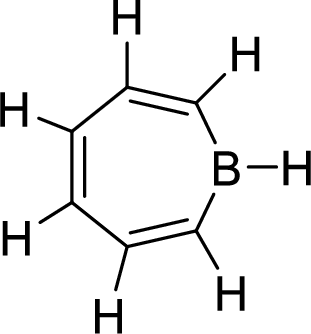
There are three
Huckel’s rule of aromaticity
(i)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
The given compound is shown here:
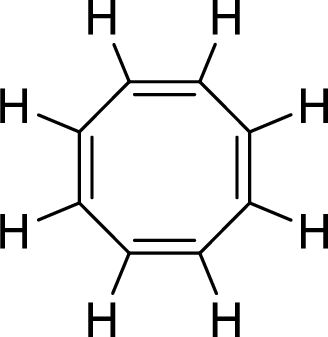
There are four
Huckel’s rule of aromaticity
(j)
Interpretation:
Which of the given molecules and ions are aromatic according to the
Concept Introduction:
The term aromaticity means “extreme stability”. So, aromatic compounds are highly stable compounds whereas anti-aromatic compounds are highly unstable compounds.
The aromatic compounds and anti-aromatic compounds can be distinguished based on Huckel’s rule of aromaticity.
Huckel’s rule of aromaticity is
If
If
If
Explanation of Solution
The given compound is shown here:
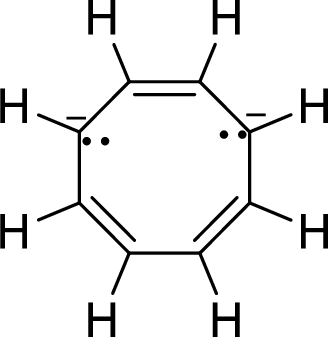
There are three
Huckel’s rule of aromaticity
Want to see more full solutions like this?
Chapter 21 Solutions
Organic Chemistry
- Of Hückel's original rules for aromaticity, one needed to be revised. Which one is it? The ring system must be planar. There must be (4n + 2) pi electrons. The molecule must be monocyclic. The pi electrons must be resonance delocalized over the entire ring. QUESTION 17arrow_forward3) In the mid-1930s, the German theoretical chemist Erich Hückel developed a rule that dealt with the aromaticity of various compounds, which became known as the Hückel Rule. Which (parts) of the compounds listed below are aromatic? Justify your answer based on Hückel's rule. You can treat the rings separately or together as you wish. IMAGE IV: colchicine: a highly poisonous alkaloid, obtained from autumn turmeric and used to treat gout.arrow_forward3) In the mid-1930s, the German theoretical chemist Erich Hückel developed a rule that dealt with the aromaticity of various compounds, which became known as the Hückel Rule. Which (parts) of the compounds listed below are aromatic? Justify your answer based on Hückel's rule. You can treat the rings separately or together as you wish.image iv: colchicine: a highly poisonous alkaloid,obtained from autumn turmeric and used to treat gout.arrow_forward
- Why is benzene less reactive than hexane in terms of stability of free radicals?arrow_forwardPyrene has been determined experimentally to be aromatic. At first glance, however, its structure appears to break Hückel’s rule. How so? Can you explain why pyrene exhibits aromaticity?Hint: What are the characteristics of the π system on the periphery of the molecule?arrow_forward5. Which of these structures can be classified as aromatic and why? III IIarrow_forward
- Determine if the following compounds are aromatic, non-aromatic, or anti-aromatic. Show a structure that helps to support your classification. This structure should not just redraw the compound; show lone pairs, resonance, and/or contributing π bonds as part of your explanation.arrow_forward1. Which of the following structures is considered aromatic?arrow_forwardWhich among the five aromatic molecules will have the least ability to undergo molecularstacking?A. Structure 1B. Structure 2C. Structure 3D. Structure Which aromatic structures contains functional groups which are electron-withdrawing?A. Structures 1, 2 and 4B. Structures 2, 3 and 5C. Structures 2, 4, and 5D. Structures 1, 3, and 4 Which aromatic structures contain functional groups which are electron-donating?A. Structures 1 and 3B. Structures 1 and 4C. Structures 3 and 5D. Structures 3 and 4 In which structure do you expect two products after alkylation?A. Structure 1B. Structure 2C. Structure 3D. Structure 4 In which carbon position will an incoming halogen be located in Structure 3, relative tothe OH group?A. Ortho positionB. Meta positionC. Para positionD. Ortho and para position In which aromatic molecule do you expect to have a carbocation form during resonancestabilization?A. Structures 2, 3 and 4B. Structures 2, 4 and 5C. Structures 2, 3 and 5D. Structures 1, 3 and 4arrow_forward
- if boron replaces acarbon in a benzene ring, is the ring still aromatic? or is it antiaromaticarrow_forwardWhat is Resonance Theory? Sate five conclusions that can be drawn from the theory. State the two main experiments that were used to establish the extra stability of the benzene molecule. What are the factors that confer Aromaticity to an organic molecule? i. State the effects of substituents on a benzene derivative towards further aromatic substitution. Based on the above suggest the various types of substituents that can be attached to Benzene. What are the various ways by which alkenes may be synthesized? i. Give two examples each of Unsymmetrical alkenes and reagents. Give two examples of reactions of alkenes that result in Anti-Markonikov’s addition productsarrow_forwardIs the molecule aromatic, anti-aromatic, or non aromatic?arrow_forward
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning


