
(a)
Interpretation:
The exergonic portion present in given reaction should be identified.
Concept Introduction:
Exergonic: The reaction is considered as exergonic if energy released since the reactants loses its energy, making the free energy is more negative hence making it spontaneous reaction.
Endergonic: The reaction is considered as endergonic if it needs more energy means that activation energy is much higher making the reaction non spontaneous.
(a)
Answer to Problem 21.20UKC
The exergonic portion is conversion of succinyl phosphate into succinate.
Explanation of Solution
Analyzing the given reaction it is clear that conversion of succinyl phosphate to succinate is exergonic since the phosphate bond in succinyl phosphate gets cleaved which releases energy.
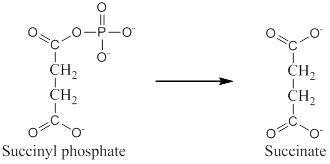
Hence, exergonic portion is conversion of succinyl phosphate into succinate.
(b)
Interpretation:
The endergonic portion present in given reaction should be identified.
Concept Introduction:
Exergonic: The reaction is considered as exergonic if energy released since the reactants loses its energy making the free energy more negative hence making it spontaneous reaction.
Endergonic: The reaction is considered as endergonic if it needs more energy means that activation energy is much higher making the reaction non spontaneous.
(b)
Answer to Problem 21.20UKC
The endergonic portion is conversion of
Explanation of Solution
Analyzing the given reaction it is clear that conversion of
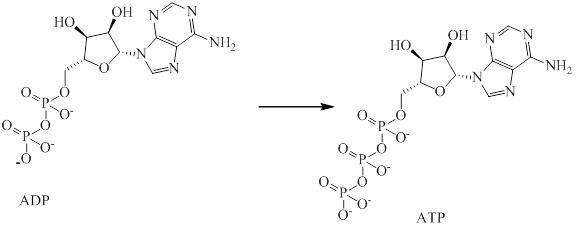
Therefore, addition of phosphate group into
Hence, endergonic portion is conversion of
Want to see more full solutions like this?
Chapter 21 Solutions
Fundamentals of General, Organic, and Biological Chemistry (8th Edition)
- which statement is true for the following reaction?arrow_forwardAn exergonic reaction has which of the following properties? A) The △G is negative and the reaction is spontaneous. B) The △G is negative and the reaction is non-spontaneous. C) The △G is positive and the reaction is spontaneous.arrow_forwardWrite a stepwise mechanism for the following reaction.arrow_forward
- Draw the product AND propose a reasonable, detailed stepwise mechanism, using curved arrow notation to show the flow of electrons, for the following reaction.arrow_forwardHow does the rate of the forward reaction compare to the rate of the reverse reaction for an endergonic reaction? For an exergonic reaction? Explain.arrow_forwardA certain first order reaction has the rate law Rate = k[A] with k=0.0068 sec-1. If the initial concentration of A is 0.75 M, what will be the concentration of A after 1 minute? What is the half-life for this reaction? How much time will it take for 75% of A to react? How much A will be left after the passage of three half-lives? What is the initial rate of the reaction?arrow_forward
- Under standard conditions, will the following reaction proceed spontaneously as written?arrow_forwardConsider the reaction below to answer the following question(s): + HBr A B Br с + D Br Enter the appropriate letter in the blank for each the following statements. The kinetically controlled product in this reaction is D B Aarrow_forwardThe equilibrium constant for the reaction Q → R is 25. Solve, (a) If 50 μM of Q is mixed with 50 μM of R, which way will the reaction proceed to generate more Q or more R? (b) Calculate the equilibrium concentrations of Q and R.arrow_forward
 BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman
BiochemistryBiochemistryISBN:9781319114671Author:Lubert Stryer, Jeremy M. Berg, John L. Tymoczko, Gregory J. Gatto Jr.Publisher:W. H. Freeman Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman
Lehninger Principles of BiochemistryBiochemistryISBN:9781464126116Author:David L. Nelson, Michael M. CoxPublisher:W. H. Freeman Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY
Fundamentals of Biochemistry: Life at the Molecul...BiochemistryISBN:9781118918401Author:Donald Voet, Judith G. Voet, Charlotte W. PrattPublisher:WILEY BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305961135Author:Mary K. Campbell, Shawn O. Farrell, Owen M. McDougalPublisher:Cengage Learning BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning
BiochemistryBiochemistryISBN:9781305577206Author:Reginald H. Garrett, Charles M. GrishamPublisher:Cengage Learning Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON
Fundamentals of General, Organic, and Biological ...BiochemistryISBN:9780134015187Author:John E. McMurry, David S. Ballantine, Carl A. Hoeger, Virginia E. PetersonPublisher:PEARSON





