
Concept explainers
(a)
Interpretation:
The type of isomerism that could be exhibited by the given formulae and an example that illustrates the specific type of isomerism has to be stated.
Concept introduction:
Organic compounds which have similar chemical formula but different structures, i.e., the atoms are in a different spatial arrangement, they are known as structural isomers. Cis and Trans isomers are also structural or geometrical isomers.
(a)
Answer to Problem 2Q
Explanation of Solution
The type of isomerism that could be exhibited by
The structural isomer of
The given formula,
The root word hexene indicates that six carbon atoms are present in the parent chain. Double bond is present between first and second carbon.
The structure of 1-hexene is shown
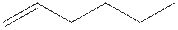
The structural isomer of
The given formula,
The root word hexene indicates that six carbon atoms are present in the parent chain. The number
The structure of 2-hexene is shown
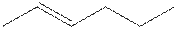
The structural isomer of
The given formula,
The root word hexene indicates that six carbon atoms are present in the parent chain. The number
The structure of 3-hexene is shown
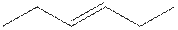
The structural isomer of
The given formula,
The root word pentene indicates that five carbon atoms are present in the parent chain. The number
The structure of

The structural isomer of
The given formula,
The root word butene indicates that six carbon atoms are present in the parent chain. The double bond is present between second and third carbon. Two methyl groups are attached at second and third carbon.
The structure of
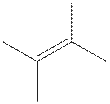
The geometric isomer of
The structure of cis-2-hexene is,
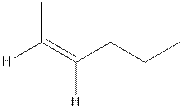
The structure of trans-2-hexene is,
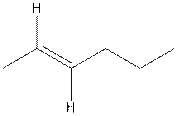
In the cis isomer, the two hydrogen of the double bonded carbons are adjacent to each other while in trans isomer, they are opposite to each other.
The geometric isomer of
The structure of cis-3-hexene is,
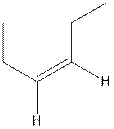
The structure of trans-3-hexene is,
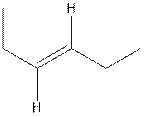
In the cis isomer, the two hydrogen of the double bonded carbons are adjacent to each other while in trans isomer, they are opposite to each other.
(b)
Interpretation:
The type of isomerism that could be exhibited by the given formulae and an example that illustrates the specific type of isomerism has to be stated.
Concept introduction:
Organic compounds which have similar chemical formula but different structures, i.e., the atoms are in a different spatial arrangement, they are known as structural isomers. Cis and Trans isomers are also structural or geometrical isomers.
(b)
Answer to Problem 2Q
Explanation of Solution
The type of isomerism that could be exhibited by
The structural isomer of
The root word pentane indicates that five carbon atoms are present in the parent chain. Hydroxyl group is attached to first carbon.
The structure of pentan-1-ol is shown,

The structural isomer of
The root word butane indicates that four carbon atoms are present in the parent chain.
The structure of
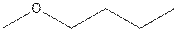
The structural isomer of
The root word propane indicates that three carbon atoms are present in the parent chain.
The structure of 2-ethoxypropane is shown

The structural isomer of
The root word butane indicates that four carbon atoms are present in the parent chain.
The structure of 2-methoxybutane is shown
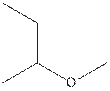
The structural isomer of
The root word pentane indicates that five carbon atoms are present in the parent chain. Hydroxyl group is attached to third carbon.
The structure of pentan-
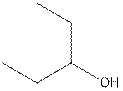
The structural isomer of
The root word pentane indicates that five carbon atoms are present in the parent chain. Hydroxyl group is attached to second carbon.
The structure of pentan-

(c)
Interpretation:
The type of isomerism that could be exhibited by the given formulae and an example that illustrates the specific type of isomerism has to be stated.
Concept introduction:
Organic compounds which have similar chemical formula but different structures, i.e., the atoms are in a different spatial arrangement, they are known as structural isomers. Cis and Trans isomers are also structural or geometrical isomers.
(c)
Answer to Problem 2Q
Explanation of Solution
The type of isomerism that could be exhibited by
The structural isomer of
In
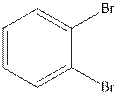
The structural isomer of
In
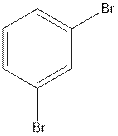
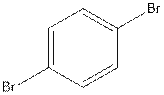
The structural isomer of
In
Want to see more full solutions like this?
Chapter 22 Solutions
OWLv2 with MindTap Reader, 4 terms (24 months) Printed Access Card for Zumdahl/Zumdahl's Chemistry, 9th
- A student adds bromine solution to a test tube containing an isomer of C3H6. After shaking the sample and exposing it to UV light, the student observes that the colour of the bromine solution changes from orange to clear. Provide a hypothesis for what happened in the test tube. What do you believe to be the structure of the original isomer, and what was the final product, if a reaction occurred?arrow_forwardWhich of the following organic compounds is the least soluble in H2O? A. carboxylic acids B. alcohols C. primary amines D. ethersarrow_forwarda. Give all the structural isomers of the hydrocarbons C5H12.(Hint: 3 isomers) b. Give all the structural isomers of the compound C3H5Cl which contains one C=C bond. (Hint: 3 isomers) c. One of the structural isomers in part b shows cis-trans isomerism. Draw and name this pair of isomers.arrow_forward
- 4. Which of the following represents the correct ranking of biologičal molecules from smallest to largest? A. Macromolecule > polymer monomer carbon atom B. Polymer-> monomer -→ macromolecule > carbon atom C. Carbon atom macromolecule polymer > monomer D. Carbon atom monomer polymer → macromoleculearrow_forwardWhich of the following is not considered a true organometallic compound? a. Ziese's salt b.cis-platin c. Ferrocene d. Vitamine B12 co-enzyme, "cobalamine"arrow_forwarda chemical equation is given below. how would you classify this reaction 2NH3 h2so4->(nh4)2SO4 A. double replacement B. decomposition C. synthesis D. single replacementarrow_forward
- 1. What are elements and compounds? Give an example of each. 2. What determines the primary structure of a protein? 3. State two functions of proteins. 4. Identify the three parts of a nucleotide. 5. Identify the roles of reactants and products in chemical reactions. 6. What are biochemical reactions? What is an example? 7. Where is most of Earth's water found? 8. What is polarity? Describe the polarity of water.arrow_forwardComplete and balance the following combustion reactions. Assume that each hydrocarbon is converted completely to carbon dioxide and water. (a) Propane + O2 (b) Octane + O2 (c) Cyclohexane + O2 (d) 2-Methylpentane + O2arrow_forwardgive the nomenclature or the structure of the following moleculesarrow_forward
 Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
Chemistry: An Atoms First ApproachChemistryISBN:9781305079243Author:Steven S. Zumdahl, Susan A. ZumdahlPublisher:Cengage Learning
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781337399074Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning





