
Concept explainers
(a)
Interpretation: The result of treating compound 1 with
Concept introduction:
Reduction reaction: The reaction in which addition of hydrogen takes place is known as reduction reaction.
The reduction of a
Alcohols are the hydrocarbons which have
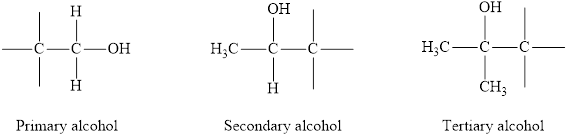
Alcohols can be of three types on the basis of degree of carbon atom to which the
Primary alcohol: The
Secondary alcohol: The
Tertiary alcohol: The
(a)
Answer to Problem 70PS
The product obtained from the reaction of given compound
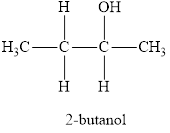
The systematic name of the product is 2-butanol and functional group is alcohol.
Explanation of Solution
Compound 1 s,
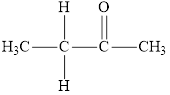
It is a ketone
The reaction between compound
The reduction of a ketone gives a secondary alcohol in presence of
The reaction is in which the compound

The product has same number of carbon atoms as in compound
The parent chain name will be butane and suffix will be “-ol” as there is one
Therefore, name of the product is 2-butanol and functional group is alcohol.
(b)
Interpretation: The structure of the reaction product from comounds 2 and 4 has to be drawn. The functional group of the product has to be identified
Concept introduction:
Ester: One
The name for an ester molecule can be written by using alkyl chain
Esterification reaction: Esters are prepared by the reaction of a
Here, the
(b)
Answer to Problem 70PS
The structure of the product is,
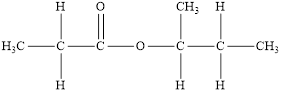
The functional group is an ester
Explanation of Solution
Compound 2 is,
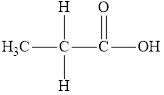
It is a carboxylic acid
Compound 4 is,
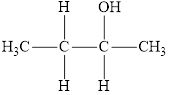
It is a secondary alcohol
Esters are prepared by the reaction of a carboxylic acid and an alcohol molecule with the elimination of water molecule
The reaction is written below in which the compound
Here, the
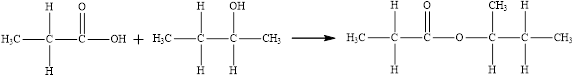
The product has
(c)
Interpretation: The compound that results from the addition of
Concept introduction:
Hydrogenation: The addition of hydrogen to unsaturated compounds to convert into saturated compounds in the presence of catalyst.
An
Alcohols are the hydrocarbons which have
Alcohols can be of three types on the basis of degree of carbon atom to which the
(c)
Answer to Problem 70PS
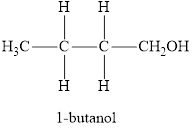
The product obtained from the reaction of given compound
Explanation of Solution
Compound 3 is,
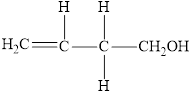
It is an alkene
The reaction between compound
In this reaction, the compound
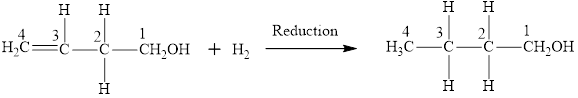
The product has same number of carbon atoms as in compound
The parent chain name will be butane
The suffix will be “-ol” as there is one
Therefore, name of the product is 1-butanol and functional group is alcohol.
(d)
Interpretation: The compound that results from the addition of
Concept introduction:
The reaction of carboxylic acid with
(d)
Answer to Problem 70PS
The product obtained from the reaction of given compound
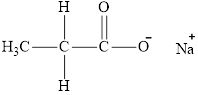
Explanation of Solution
The reaction of carboxylic acid with
The product obtained is the sodium salt of the acid and water.
The reaction of compound
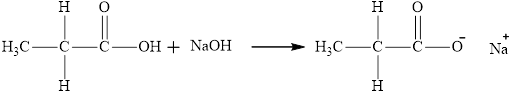
Want to see more full solutions like this?
Chapter 23 Solutions
Chemistry & Chemical Reactivity
- 2.) Draw 1 isomer of the compounds in item no. 3, and state the type of isomerism. Example: 2-hydroxy-pentanal OH Structure: Isomer: OH Type of isomerism: Functional Isomerism 3.) Predict the product of the following chemical reactions and state where the product (waste) should the waste should be disposed: Example: OH 1. NaBH, ethanol 2. HO CH,CH,CH,CH CH;CH,CH,CH H Butanal 1-Butanol Waste: G704 (Non-halogenated organic solvents) 1. NaBH,, ethanol a.) Cyclohexanecarbaldehyde 2. H,O" b.) Sodium Flouride + 3-iodo-3-methylpentane (hint: substitution or elimination?) c.) Benzoic acid + Phenol РСС d.) 3-pentanol H>CrO4 e.) Cyclohexylmethanolarrow_forward(a) When a compound containing C, H, and O is completelycombusted in air, what reactant besides the hydrocarbonis involved in the reaction? (b) What products form in thisreaction? (c) What is the sum of the coefficients in the balancedchemical equation for the combustion of one mole ofacetone, C3H6O1l2, in air?arrow_forward(i) Draw the structural formula of compounds L, M, N and P (ii) Name the type of chemical reaction for the formation of compound N.arrow_forward
- Write the reagent or draw structures of the starting material or organic product(s) in the following reactions. If more than one product is formed, identify the major product where possible. (a) (b) HO OH OH H2SO4 ? Cl₂ ? FeCl3arrow_forward5. Give the structural formulae and name the functional groups of the following compounds. (a) 3-chlorobut-1-ene (b) butanedioic acid Name the functional group: (c) propanamide Name the functional group: (d) 3-methylbutanal Name the functional group: Name the functional group:arrow_forward(1) Write a complete chemical equation showing reactants, products, and catalysts needed (if any) for the following reaction and (2) Draw and name the organic compound found in every reaction. (a) Complete hydrogenation of 2-Methylhexa-1,5-diene (b) Complete halogenation (Br2) of 3-Ethyl-2,2-dimethylhept-3-ene (c) Reaction of (4E)-2.4-Dimethylhexa-1,4-diene with a mole of water (d) Reaction of cis-3,3-Dimethyl-4-propylocta-1,5-diene with two mole of HBr (e) Reaction of trans-1-Bromo-3-chlorocyclopentane with potassium hydroxide (f) Formation of Gilman reagent using isopropyl bromide (g) Ozonolysis of 3,3-Dimethyloct-4-yne (h) Complete halogenation (Cl2) of 3-Ethyl-5-methyl-1,6,8-decatriyne (i) Partial hydrogenation using Lindlar's Catalyst 2,2,5,5-Tetramethylhex-3-yne (i) Reaction of 3.4-Dimethylcyclodecyne with sodium amidearrow_forward
- Draw the organic product formed when the following compounds undergo a substitution reaction: (a) acetic acid and 1-hexanol; (b) propanoic acid and dimethyl-amine; (c) ethanoic acid and diethylamine.arrow_forward2. Write the structure for the most oxidized organic product when each of the following is reacted with potassium dichro- mate. Write NR if no reaction occurs.(a) methanol (b) ethyl alcohol (c) 2,3,4-trimethyl-2-pentanol (d) 3-hexanol 3. Alcohols can be made by reacting alkyl halides with sodium hydroxide as follows: RX +. NaOH ROH + NaX Give the names and formulas of the alcohols produced from the following alkyl bromides by this method:(a) 2-bromobutane(b) 2-bromo-3-ethylpentane (c) bromocyclopentanearrow_forwardGive one example for the hydration of Alkenes: that is: the addition of H2O by Hydroboration.arrow_forward
- Sodium hydride, NaH, is available commercially as a gray-white powder. It melts at 800°C with decomposition. It reacts explosively with water and ignites spontaneously upon standing in moist air. Q) Write an equation for the reaction between sodium hydride and 1-butyne, CH3CH2C = CH. Use curved arrows to show the flow of electrons in this reactionarrow_forward2. An unknown hydrocarbon A with the formula C 6H 12 reacts with 1 molar equivalent of H 2 over palladium catalyst. Hydrocarbon A also reacts with OsO 4 to give diol B. When oxidized with KMnO 4 in acidic solution, A gives two fragments. One fragment is propanoic acid, CH3CH 2CO 2H and the other fragment is a ketone. Which of the following are correctarrow_forwarda) b) O 4. Draw the correct structure of the alcohol starting material that would give the following oxidized products: 0. RE 0= forarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





