
Organic Chemistry
9th Edition
ISBN: 9781305080485
Author: John E. McMurry
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 24.SE, Problem 51AP
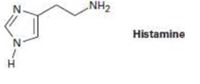
Histamine, whose release in the body triggers nasal secretions and constricted airways, has three nitrogen atoms. List them in order of increasing basicity and explain your ordering.
Expert Solution & Answer
Trending nowThis is a popular solution!

Students have asked these similar questions
List the following compounds by discussing their basicity.
Atenolol is a β (beta) blocker, a drug used to treat high blood pressure. Which of the indicated N—H bonds is more acidic? Explain your reasoning.
Using pKa Values to Determine Relative Acidity and Basicity Rank the following compounds in order of increasing acidity, and then rank their conjugate bases in order of increasing basicity.
Chapter 24 Solutions
Organic Chemistry
Ch. 24.1 - Name the following compounds:Ch. 24.1 - Prob. 2PCh. 24.1 - Prob. 3PCh. 24.3 - Prob. 4PCh. 24.3 - The benzylammonium ion (C6H5CH2NH3+) has pKa =...Ch. 24.4 - Prob. 6PCh. 24.5 - Prob. 7PCh. 24.6 - Prob. 8PCh. 24.6 - Prob. 9PCh. 24.6 - Show two methods for the synthesis of dopamine, a...
Ch. 24.6 - Prob. 11PCh. 24.6 - Prob. 12PCh. 24.6 - Prob. 13PCh. 24.7 - Prob. 14PCh. 24.7 - Prob. 15PCh. 24.8 - Prob. 16PCh. 24.8 - Propose syntheses of the following compounds from...Ch. 24.8 - How would you prepare the following compounds from...Ch. 24.8 - Prob. 19PCh. 24.9 - Draw an orbital picture of thiazole. Assume that...Ch. 24.9 - Prob. 21PCh. 24.9 - Prob. 22PCh. 24.9 - Which nitrogen atom in the hallucinogenic indole...Ch. 24.9 - Prob. 24PCh. 24.10 - Prob. 25PCh. 24.SE - Name the following amines, and identify each as...Ch. 24.SE - The following compound contains three nitrogen...Ch. 24.SE - Name the following amine, including R, S...Ch. 24.SE - Prob. 29VCCh. 24.SE - Predict the product(s) for each reaction below and...Ch. 24.SE - Predict the product(s) and provide the complete...Ch. 24.SE - Prob. 32MPCh. 24.SE - Predict the product(s) and provide the mechanism...Ch. 24.SE - The diazotization of aniline first involves the...Ch. 24.SE - Prob. 35MPCh. 24.SE - Prob. 36MPCh. 24.SE - Prob. 37MPCh. 24.SE - Prob. 38MPCh. 24.SE - Choline, a component of the phospholipids in cell...Ch. 24.SE - Prob. 40MPCh. 24.SE - -Amino acids can be prepared by the Strecker...Ch. 24.SE - Prob. 42MPCh. 24.SE - Prob. 43MPCh. 24.SE - Prob. 44MPCh. 24.SE - Propose a mechanism for the following reaction:Ch. 24.SE - Prob. 46MPCh. 24.SE - Name the following compounds:Ch. 24.SE - Prob. 48APCh. 24.SE - Prob. 49APCh. 24.SE - Prob. 50APCh. 24.SE - Histamine, whose release in the body triggers...Ch. 24.SE - Prob. 52APCh. 24.SE - Prob. 53APCh. 24.SE - Prob. 54APCh. 24.SE - Prob. 55APCh. 24.SE - Prob. 56APCh. 24.SE - Prob. 57APCh. 24.SE - Prob. 58APCh. 24.SE - Prob. 59APCh. 24.SE - Prob. 60APCh. 24.SE - Show the products from reaction of p-bromoaniline...Ch. 24.SE - Prob. 62APCh. 24.SE - How would you prepare the following compounds from...Ch. 24.SE - Prob. 64APCh. 24.SE - Phenacetin, a substance formerly used in...Ch. 24.SE - Prob. 66APCh. 24.SE - Draw the structure of the amine that produced the...Ch. 24.SE - Fill in the missing reagents a-e in the following...Ch. 24.SE - Prob. 69APCh. 24.SE - Prob. 70APCh. 24.SE - Deduce the structure of the compound with formula...Ch. 24.SE - Prob. 72APCh. 24.SE - Prob. 73APCh. 24.SE - Prob. 74APCh. 24.SE - Prob. 75APCh. 24.SE - Prob. 76APCh. 24.SE - Propose a structure for the product with formula...Ch. 24.SE - Prob. 78APCh. 24.SE - Prob. 79APCh. 24.SE - Prob. 80APCh. 24.SE - Prob. 81APCh. 24.SE - Prob. 82APCh. 24.SE - Prob. 83APCh. 24.SE - Prob. 84AP
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Draw the structures of aspirin, caffeine, and acetaminophen. Give the hybridization of each carbon atom and state which is acidic, weakly acidic, and basic. Identify the sites on each compound that make each acidic, weakly acidic, and basic.arrow_forwardCompounds like amphetamine that contain nitrogen atoms are protonated by the HCl in the gastric juices of the stomach, and the resulting salt is then deprotonated in the basic environment of the intestines to regenerate the neutral form. Write proton transfer reactions for both of these processes.arrow_forwardRank the following compounds in terms of increasing acidity (least acidic first). Explain your ranking. Making sure to say which hydrogen in each molecule is the most acidic, and discuss the relative stability of the conjugate bases.arrow_forward
- The following compound contains four hydrogen atoms labeled a-d. Rank these hydrogen atoms in increasing order of acidity (weakest to strongest). Provide a brief explanation. Make sure you draw the conjugate base corresponding to each acidic hydrogen considered. ан. LOCH3 Answer: Varrow_forwardPlace the following in order of increasing basicityarrow_forwardRank the following bases in order of increasing basicity: LDA, cyclohexane magnesium chloride, NaOme, benzylamine, water.arrow_forward
- Rank the labeled N atoms in the anticancer drug imatinib (trade nameGleevec) in order of increasing basicity. Imatinib, sold as a salt withmethanesulfonic acid (CH3SO3H), is used for the treatment of chronicmyeloid leukemia as well as certain gastrointestinal tumors.arrow_forwardAmphetamine is a powerful stimulant of the central nervous system. (a) Which proton in amphetamine is most acidic? (b) What products are formed when amphetamine is treated with NaH? (c) What products are formed when amphetamine is treated with HCl?arrow_forwardRank the labeled nitrogen atoms in each compound in order of increasing basicity. Histamine causes the runny nose and watery eyes associated with allergies, and trazodone is a drug used as a sedative and antidepressant.arrow_forward
- Draw the conjugate base and rank the compounds in order of increasing acidity. Explain the reason brieflyarrow_forwardExplain why CH3 CH2 CH2NH2 is a Brønsted base. Its water solutions are basic. All substances containing nitrogen atoms are Brønsted bases. This amine is a proton donor. This amine can ассеpt a proton from a proton donor.arrow_forwardRank the attached compounds in order of increasing acidity, and then ranktheir conjugate bases in order of increasing basicity ?arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you

 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning


Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
General Chemistry | Acids & Bases; Author: Ninja Nerd;https://www.youtube.com/watch?v=AOr_5tbgfQ0;License: Standard YouTube License, CC-BY