
Concept explainers
(a)
Interpretation: The given reaction is to be classified as an electrocyclic reaction, cycloaddition, or a sigmatropic rearrangement. The sigma bonds that are broken or formed in the given reaction are to be labeled.
Concept introduction: A
Answer to Problem 27.1P
The reaction is classified as an electrocyclic reaction. The sigma bond broken or formed in the given reaction is,
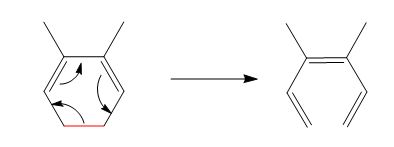
Explanation of Solution
The given reaction is shown below.

Figure 1
The sigma bond that is broken during the conversion of reactant into product is shown in red. The new bonds formed during the conversion are shown by curved arrows in the reactant. In the product, the ring is opened due to the rearrangement of
The reaction is classified as an electrocyclic reaction. The sigma bond that is broken or formed in the given reaction is labeled in Figure 1.
(b)
Interpretation: The given reaction is to be classified as an electrocyclic reaction, cycloaddition, or a sigmatropic rearrangement. The sigma bonds that are broken or formed in the given reaction are to be labeled.
Concept introduction: A chemical reaction that involves
Answer to Problem 27.1P
The reaction is classified as a cycloaddition reaction. The sigma bonds broken or formed in the given reaction are,
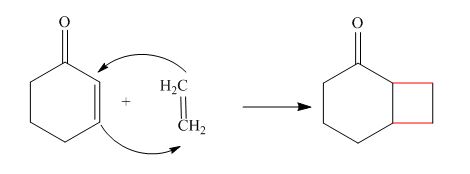
Explanation of Solution
The given reaction is shown below.

Figure 2
The sigma bonds that are formed during the conversion of reactant into a product are shown in red. In the reaction,
The reaction is classified as a cycloaddition reaction. The sigma bonds broken or formed in the given reaction are labeled in Figure 2.
(c)
Interpretation: The given reaction is to be classified as an electrocyclic reaction, cycloaddition, or a sigmatropic rearrangement. The sigma bonds that are broken or formed in the given reaction are to be labeled.
Concept introduction: A chemical reaction that involves
Answer to Problem 27.1P
The reaction is classified as an electrocyclic reaction. The sigma bond broken or formed in the given reaction is,
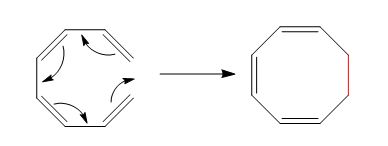
Explanation of Solution
The given reaction is shown below.

Figure 3
The sigma bond that is formed during the conversion of reactant into product is shown in red. The new bonds formed during the conversion are shown by curved arrows in the reactant. In the product, the ring gets closed due to the rearrangement of
The reaction is classified as an electrocyclic reaction. The sigma bond broken or formed in the given reaction is labeled in Figure 3.
(d)
Interpretation: The given reaction is to be classified as an electrocyclic reaction, cycloaddition, or a sigmatropic rearrangement. The sigma bonds that are broken or formed in the given reaction are to be labeled.
Concept introduction: A chemical reaction that involves
Answer to Problem 27.1P
The reaction is classified as a sigmatropic reaction. The sigma bonds broken or formed in the given reaction are,
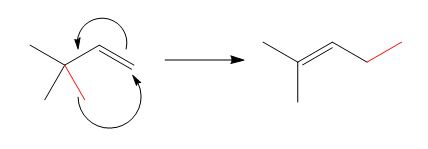
Explanation of Solution
The given reaction is shown below.

Figure 4
The sigma bond that is broken and formed during the conversion of reactant into product is shown in red. The product indicates that
The reaction is classified as a sigmatropic reaction. The sigma bonds broken or formed in the given reaction are labeled in Figure 4.
Want to see more full solutions like this?
Chapter 27 Solutions
Organic Chemistry
- Label each pair of alkenes as constitutional isomers, stereoisomers, or identical. А. and constitutional B. and isomers identical stereoisomers C. CH3 and H. constitutional isomers CH3 CH3 CH3CH2 CH2CH3 CH3CH2 CH3 D. and CH3 CH3 CH3 CH2CH3 I-arrow_forwardDraw the structures of A -D from the following electrocyclic and cycloaddition reactions. CH heat hv B D. CH3arrow_forward48. Which set of reagents would accomplish this conversion H. H. CH3OH with H* as a catalyst NaH followed by CH3OH CH3ONA followed CH3OH CH3ONA followed by CH3Br a. b. C. d.arrow_forward
- Draw the alkene that would react with the reagent given to account for the product formed. ? + H₂O H₂SO4 CH3 CH3 CHCCH3 OH CH3 • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. Sn [F ChemDoodlearrow_forward2. Explain the selectivity of the following reaction, which produces a single product despite both alkene carbons being equally substituted. H3C CF3 HBr CF3 H3C CF3 H3C Br- H3C only product -H CF3arrow_forwardDraw the alkene that would react with the reagent given to account for the product formed. ? + + H₂O **** H₂S04 • You do not have to consider stereochemistry. • You do not have to explicitly draw H atoms. • In cases where there is more than one answer, just draw one. CH3 CH3 CHCCH3 | | OH CH3 +1arrow_forward
- Carbocations are carbons bearing a formal positive charge. a. Carbocations can be stabilized by induction and hyperconjugation. Explain each of these forms of stabilization.arrow_forwardWhich alkene has (E) configuration? a. b. C. d.arrow_forward6. Classify this Payne rearrangement according to Balwin's rules and determine if it is favored or not. OH NaOH H₂O: t-BuOH но,arrow_forward
- Draw a reaction coordinate diagram for a reaction in which a. the product is thermodynamically unstable and kinetically unstable. b. the product is thermodynamically unstable and kinetically stable.arrow_forwardRank the following alkenes from most to least stable. A. В. С. D. B.arrow_forward1. Draw the products of each nucleophilic substitution reaction a. b. D b OH C. d. e. f. Br 1 NaCN + NaOCH3 H₂Oarrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





