
Concept explainers
(a)
Interpretation: The reaction of
Concept introduction: An atom or a group of atoms that shows characteristic physical and chemical properties are collectively known as
According to Bronsted-Lowry theory, the species that can easily accept the proton is known as base and the species that can easily donate the proton is known as acid. The reaction of an acid with a base always leads to the formation of conjugate acid and base.
Answer to Problem 3.1P
The reaction of
Explanation of Solution
The reaction of
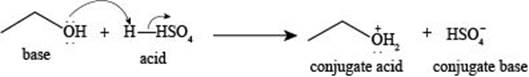
Figure 1
The reaction of ethanol
The reaction of
The reaction of ethane
The reaction of
(b)
Interpretation: The reaction of
Concept introduction: An atom or a group of atoms that shows characteristic physical and chemical properties are collectively known as functional groups. The functional group is the most reactive part present in the molecule. The main functional groups are
According to Bronsted-Lowry theory, the species that easily tends to accept the proton is known as base and the species that easily donate the proton is known as acid. The reaction of an acid with a base always leads to the formation of conjugate acid and base.
Answer to Problem 3.1P
The reaction of
Explanation of Solution
The reaction of
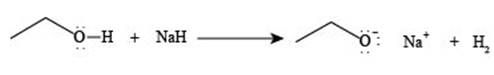
Figure 2
The reaction of ethanol
The reaction of
The reaction of ethane
The reaction of
Want to see more full solutions like this?
Chapter 3 Solutions
Organic Chemistry
- What products are formed when benzoic acid (C 6H 5COOH) is treated with each base: (a) NaOH; (b) Na 2CO 3; (c) NaHCO 3?arrow_forwardDraw the products formed when propan-2-ol [(CH3)2CHOH], the main ingredient in rubbing alcohol, is treated with each acid or base: (a) NaH (b) H2SO4 (c) Li+−N[CH(CH3)2]2 (d) CH3CO2H.arrow_forwardWhat is the product of this reaction? (A) OEt 1. LiAlH4 OEt 2.H+ (B) H ??? (C) OH (D) OHarrow_forward
- The action of heating one mole of water (containing a trace of acid catalyst) on one mole of compound Y produces one mole of CH,COOH and one mole of CH, CH,NH, The structure of Y is: Select one: O CHCNCCH, CH,CH,NHCH,CH, OH CH CHNHCH,CH, O CHCH,CNHCH; O CH;CH NHÖCH;arrow_forwardName and draw the products of each reaction. 1-propanol + hydrobromic acid → (a) (b) 1-pentanol H-SO A (c) N-propylmethanamide + water (d) 3-methyl oct-2-ene + hydrochloric acid -arrow_forwardDraw the products formed when benzoic acid (C6H5CO2H) is treated with CH3OH having its O atom labeled with 18O (CH318OH). Indicate where the labeled oxygen atom resides in the products.arrow_forward
- What is the major organic product obtained from the following reaction? OH OH H,0 CH;CH,CH,CH,C=CH OH HgSO, H,SO, ÓH O 1 O 2 O 3 O 4arrow_forwardComparing Two Different Methods of Hydration of an Alkene Draw the product formed when CH3CH2CH2CH2CH=CH2 is treated with either (a) H2O, H2SO4; or (b) BH3 followed by H2O2, HO−.arrow_forward4) Provide the starting materials for the following reactions. a) b) c) HBr 0 °C H₂O, H₂SO4 NaOH (2 equivalents) H₂O, 55 °C OH Brarrow_forward
- Name and draw a structural formula for the major product of each alkene addition reaction: CH S (a) CH₂C=CH₂ + HI →→→→ (b) CH 3 + HClarrow_forwardWhich products are formed when hydrobromic acid is added to (a) trans-2-hexene, (b) 2-methyl- 2-pentene, and (c) 4-methylcyclohexene, and how many regioisomers can be formed in each case?arrow_forwardWhat reaction occurs when CH3CH2OH is treated with (a) H2SO4? (b) NaH?arrow_forward
 Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning
Chemistry & Chemical ReactivityChemistryISBN:9781133949640Author:John C. Kotz, Paul M. Treichel, John Townsend, David TreichelPublisher:Cengage Learning

