
Concept explainers
(a)
Interpretation: The reason behind equal potential energies for all four
Concept introduction: Two or more of orbitals undergo redistributions of their different energies so as to form mathematically averaged orbitals in terms of energy although they may differ in shape and orientation. This phenomenon is referred as hybridization.
(b)
Interpretation: The reason behind energy level line for
Concept introduction: Two or more of orbitals undergo redistributions of their different energies so as to form mathematically averaged orbitals in terms of energy although they may differ in shape and orientation. This phenomenon is referred as hybridization. The new hybrid orbitals are always equal in number to number of atomic orbitals that combine.
(c)
Interpretation: The diagram given below for
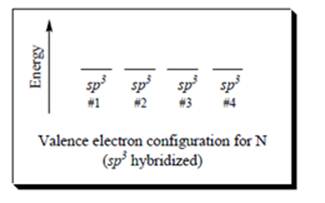
Concept introduction: Two or more of orbitals undergo redistributions of their different energies so as to form mathematically averaged orbitals in terms of energy although they may differ in shape and orientation.
The energy and orientation of the new hybrid orbital depends upon by the kind and number of orbitals used in the hybridization. For instance, when one
(d)
Interpretation: The second-row element shown in left diagram of below image and hybridization state in right diagram of below image should be identified.
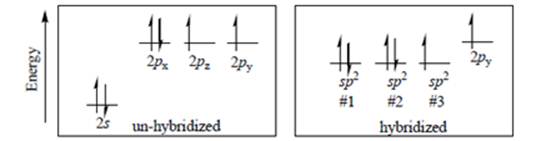
Concept introduction: Hund’s rule of maximum multiplicity states that electrons are not allowed to be paired up until each degenerate set of orbital has got at least one electron.
(d)
Interpretation: Whether the unpaired electrons in below figure are in accordance with Hund’s rule or not should be identified.
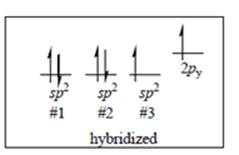
Concept introduction: Hund’s rule of maximum multiplicity states that electrons are not allowed to be paired up until each degenerate set of orbital has got at least one electron.
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Organic Chemistry: A Guided Inquiry
- 2. The following questions apply to molecules X and Y. a. Determine the hybridisation of all atoms in the two molecules. b. Label each o bond, give the type of both orbitals (sp³, sp², sp or s) that make up the bond. Which orbitals are used to make up the bonds? c. Using your molecular model kit, choose atoms of the correct colour (white = H, black = C, blue = N and red = O) and correct shape for their hybridization (sp³ = 4 lobes, sp² = 5 lobes and sp= 6 lobes). Build the two models showing sigma bonds in grey and pi bonds in white. What are the angles around the central atom for sets of three atoms? O || H3C-C-CH3 and H₂C=C=CH₂ X Yarrow_forwardWhy is L46. Sketch the molecular orbital and label its type (o or T; bond- r antibonding) that would be formed when the following atomic orbitals overlap. Explain your labels. a. +. b. C. d. the ie bond.To Whioh +. 1.arrow_forwardST 9C.2- Calculate the energy (in eV) of an antibonding molecular orbital for the molecule XY (with X being more electronegative) given the information below. Note how much closer (a little bit higher) it is to the energy of Y's atomic orbital than X's atomic orbital. a (X) = -11.2 eV a (Y) = -8.8 eV B = -1.0 eV Type your answer...arrow_forward
- a. Using the molecular orbital diagram, calculate the bond order of F2+. Show show your work or give a brief explanation of the process. b. Do you expect this to have a shorter or longer bond length than F2? Explain your answer. c. Do you expect F2+ to be paramagnetic or diamagnetic? Explain your answer. Please label your answers a-c and place them on separate lines. Be as specific in your explanation as you can.arrow_forward4. Draw the molecular orbital diagram for acetic acid. Label the hybridized orbitals sp, sp2, sp3, s, p. Draw in electrons pairs when needed. Indicate sigma and pi bonds. Hi sps spe Sp 3 o lbonds =6+1=7 %3D TY bnds = 1arrow_forwardB. Answer the next two questions about the Lewis structure of XYZ shown below. メー三 -YEz: I. What is the hybridization of the central atom? II. For all covalent bonds, specify the type of bond (o vs. T) and the atomic orbitals that overlap to form the bond. For example, in a molecule of H2, the only bond is o: H(s)-H(s).arrow_forward
- 10 2. Draw the four n-molecular orbitals for the molecule butadiene, drawn below. Label the four orbitals as bonding, non-bonding or antibonding. Which of the four orbitals are occupied? Which is the LUMO and which is the HOMO? sib laidio mlunal 4 Pi electrons by 88- D gilnoo slengoggu or o ed bos 2olunolom od om baod 1915b owi ni 29luoslom pa sbuloni ot olduob orb juods ohib owi ow nisiqxo HSG CH3 88 88 OCH-CH? CCICHCCHCH?arrow_forward1. Do all of the following for each molecule or ion. a. Draw the Lewis Dot structure that minimizes formal charges. b. Draw the 3-D structure. c. Determine the Hybridization designation of the central atom. d. Determine the VSEPR designation. e. State what is its Molecular shape. f. Determine whether it is Polar (P) or Nonpolar (N). A. SO3²- B. CN- C. BrF4+ D. IF5 E. SeO2arrow_forwardesc ck For SiSe2 The sigma bond between the Silicon and one of the seleniums is best described as a result of O The overlap of an sp3 hybrid orbital on silicon with an sp3 hybrid orbital on selenium. O The overlap of an sp hybrid orbital on silicon with an sp2 hybrid orbital on selenium O the overlap of an sp hybrid orbital on selenium with an sp2 hybrid orbital on silicon O The overlap of an sp hybrid orbital on icon with an sp hybrid orbital on selenium A O the overlap of unhybridized p orbitals on selenium and silicon ! 1 Q A 1 control option 2 N W S #3 X command D $ 4 C R F % 5 T V MacBook Pro A 6 G Y B & 7 H U * N 8 Jarrow_forward
- 31. TRUE or FALSE: The orbital energy diagram represents a molecule in its ground state electron configuration? o u2s Energy g2p A. TRUE B. FALSE 11 2px 2py 2pz 2px 2py 2pz 1 Og2p Tu2p 32. TRUE or FALSE: The molecular orbital energy diagram describes the bonding in the diatomic molecule B21-? u2s A. TRUE B. FALSE 2s 2s Og2s 33. TRUE or FALSE: The molecular orbital energy diagram is representative of a paramagnetic molecule? A. TRUE B. FALSE 34. According to the molecular orbital energy diagram, the bond order of the molecule described is...? А. 1.0 В. 1.5 С. 2.0 D. 2.5 E. None of the above 35. Of the following molecular orbitals of this molecule that do NOT have cylindrical symmetry, which is the highest in energy...? A. oʻu2p В. п'92p C. Og2p D. πυ2p E. None of the abovearrow_forward1. Predict the relative 0-0 bond distances for 02, [02]1+ and [02]2+ from shortest to longest. 2. Predict the relative 0-0 bond distances for 02, [02]1- and [02]2- from shortest to longest. 3. Show a o bond, o* anti-bond and a t bond and a non-bonding interaction between two p orbitals on adjacent atoms in a diatomic molecule. (Hint: Make 4 separate drawings.)arrow_forward15 The semi-structural formulas of two hydrocarbons are shown below: A. CH, = CH- CH, – CH, B. HC =C- CH, Complete the table based on your analysis of the group formulas of the hydrocarbons given. Number of carbon atoms in hybridization state Number of sp? Link Number of chemical sp sp bonds of type bonds of type TT A В.arrow_forward
 Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning
Chemistry: The Molecular ScienceChemistryISBN:9781285199047Author:John W. Moore, Conrad L. StanitskiPublisher:Cengage Learning Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning
Chemistry: Principles and PracticeChemistryISBN:9780534420123Author:Daniel L. Reger, Scott R. Goode, David W. Ball, Edward MercerPublisher:Cengage Learning


