
Organic Chemistry As a Second Language: Second Semester Topics
4th Edition
ISBN: 9781119110651
Author: David R. Klein
Publisher: WILEY
expand_more
expand_more
format_list_bulleted
Concept explainers
Textbook Question
Chapter 3.2, Problem 3.13P
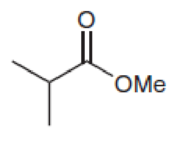
Predict the chemical shifts for the signals in the proton NMR spectrum of each of the following compounds:
Expert Solution & Answer
Want to see the full answer?
Check out a sample textbook solution
Students have asked these similar questions
A compound of molecular formula CgH,402 gives three NMR signals having the indicated
integration values: signal [A] 14 units, signal [B] 12 units, and signal [C] 44 units. How many protons
give rise to each signal?
Identify the number of signals expected in the ¹H NMR spectrum of the following compound,
&
Give three types of information you will obtain on Proton and Carbon NMR.
Chapter 3 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
Ch. 3.1 - Prob. 3.2PCh. 3.1 - Prob. 3.3PCh. 3.1 - Prob. 3.4PCh. 3.1 - Prob. 3.5PCh. 3.1 - Prob. 3.6PCh. 3.1 - Prob. 3.7PCh. 3.1 - Prob. 3.8PCh. 3.1 - Prob. 3.9PCh. 3.1 - Prob. 3.10PCh. 3.1 - If you look at your answers to the previous...
Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Predict the chemical shifts for the signals in the...Ch. 3.2 - Prob. 3.19PCh. 3.3 - Prob. 3.21PCh. 3.3 - Prob. 3.22PCh. 3.3 - Prob. 3.23PCh. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.4 - Predict the multiplicity of each signal in the...Ch. 3.5 - Below are NMR spectra of several compounds....Ch. 3.5 - Below are NMR spectra of several compounds....Ch. 3.5 - Below are NMR spectra of several compounds....Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.8 - Calculate the degree of unsaturation for each of...Ch. 3.9 - Prob. 3.43PCh. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Propose a structure for a compound with molecular...Ch. 3.9 - Prob. 3.48PCh. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...Ch. 3.10 - For each compound below, predict the number of...
Additional Science Textbook Solutions
Find more solutions based on key concepts
An experiment needs to be designed to grow a crystal in laboratory. Concept introduction: A crystal grows in tw...
Chemistry: Matter and Change
Whether the volume, height of water or both in the rain gauge changes when rainfall increases in a place needs ...
Living by Chemistry
1.1 Write a one-sentence definition for each of the following:
a. chemistry
b. chemical
Chemistry: An Introduction to General, Organic, and Biological Chemistry (12th Edition) - Standalone book
Determine the Coulombic and exchange energies for the following states, and determinewhich state is favored (ha...
Inorganic Chemistry
Determine [OH], [H+], and the pH of each of the following solutions. a. 1.0 M KCl b. 1.0 M KC2H3O2
Chemistry
For each of the following, (i) give the systematic name of the compound and specify the oxidation state of the ...
General Chemistry: Atoms First
Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Following are two constitutional isomers with the molecular formula C4H8O2. (a) Predict the number of signals in the 1H-NMR spectrum of each isomer. (b) Predict the ratio of areas of the signals in each spectrum. (c) Show how you can distinguish between these isomers on the basis of chemical shift.arrow_forwardDetermine the multiplicity and predict the chemical shifts of each signal in the expected 1H NMR spectrum of the following compound.arrow_forwardThe structure and the corresponding 1H NMR spectrum of a compound is given below. Consider the integration of the signal indicated with an arrow. Which hydrogen environment does this signal correspond to?arrow_forward
- Predict the chemical shifts for the signals in the ¹H NMR spectrum for the following compound. Your Answer: II IVarrow_forwardThe 1H-NMR spectrum of 1-chloropropane shows three signals and the 1H-NMR spectrum of 2-chloropropane shows two signals. Draw these two molecules and determine the relative integrals of each signal.arrow_forwardThe 1H NMR spectrum for the following compound was run. Determine the integration and assign peaks for the protons in the compound.arrow_forward
- Which compound gives a signal in the 1H-NMR spectrum with a larger chemical shift, furan or cyclopentadiene? Explain.arrow_forwardThe line of integration of the two signals in the 1H-NMR spectrum of a ketone with the molecular formula C7H14O rises 62 and 10 chart divisions, respectively. Calculate the number of hydrogens giving rise to each signal and propose a structural formula for this ketone.arrow_forwardWhat is the molecular formula structure and its proton environment of these two spectrum?arrow_forward
- Predict the approximate chemical shifts of the protons in the following compounds. (a) benzene (b) cyclohexanearrow_forwardSignals in a proton NMR spectrum do not provide information about Select one or more:arrow_forwardPlease look at the Proton NMR spectrum and identify the proposed molecular structure.arrow_forward
arrow_back_ios
SEE MORE QUESTIONS
arrow_forward_ios
Recommended textbooks for you
 Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
Organic Chemistry: A Guided InquiryChemistryISBN:9780618974122Author:Andrei StraumanisPublisher:Cengage Learning
 Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning
Organic ChemistryChemistryISBN:9781305580350Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. FootePublisher:Cengage Learning

Organic Chemistry: A Guided Inquiry
Chemistry
ISBN:9780618974122
Author:Andrei Straumanis
Publisher:Cengage Learning


Organic Chemistry
Chemistry
ISBN:9781305580350
Author:William H. Brown, Brent L. Iverson, Eric Anslyn, Christopher S. Foote
Publisher:Cengage Learning
Mass Spectrometry; Author: Professor Dave Explains;https://www.youtube.com/watch?v=hSirWciIvSg;License: Standard YouTube License, CC-BY