
Interpretation:
The number of protons giving rise to each signal in the spectrum of a compound with molecular formula
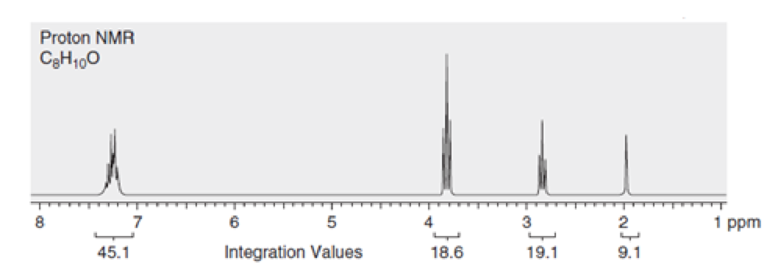
Concept Introduction:
Integration is one of the characteristics of proton NMR spectrum which is the area under each signal. After acquiring a spectrum, a computer calculates the area under each signal, and then displays this area as a numerical value placed under the signal which is relative only.
In order to convert the integration values into useful information, a smallest number should be chosen and then all integration values should be divided by this number. If the numerical values are not whole numbers, then in order to arrive at whole numbers all the numbers should be multiplied by a suitable integer.
Want to see the full answer?
Check out a sample textbook solution
Chapter 3 Solutions
Organic Chemistry As a Second Language: Second Semester Topics
- Calculate the degree of unsaturation (DU) for a molecule with the formula of C₂H, O. 10arrow_forwardA'H NMR spectrum is shown for a molecule with the molecular formula of CeH9Br. Draw the structure that best fits this data.arrow_forwardThe following molecule will give two signals in the proton (¹H) NMR. H₂C What is the ratio of the signal intensities for the two peaks? 2:2 O 3:1 3:2 CH₂ O 2:1arrow_forward
- Compound 1 has molecular formula C7H16. It shows three signals in the 1H-NMR spectrum, one at 0.85 ppm, one at 1.02 ppm, and one at 1.62 ppm. The relative integrals of these three signals are 6, 1, and 1, respectively. Compound 2 has molecular formula C7H14. It shows three signals in the 1H-NMR spectrum, one at 0.98 ppm, one at 1.36 ppm, and one at 1.55 ppm. The relative integrals of these three signals are 3, 2, and 2, respectively. Propose structures for compounds 1 and 2, explaining how you reach your conclusion.arrow_forward1Compound 1 has molecular formula C7H16. It shows three signals in the 1H-NMR spectrum, one at 0.85 ppm, one at 1.02 ppm, and one at 1.62 ppm. The relative integrals of these three signals are 6, 1, and 1, respectively. Compound 2 has molecular formula C7H14. It shows three signals in the 1H-NMR spectrum, one at 0.98 ppm, one at 1.36 ppm, and one at 1.55 ppm. The relative integrals of these three signals are 3, 2, and 2, respectively. Propose structures for compounds 1 and 2, explaining how you reach your conclusion.arrow_forwardThe organic compound 1,4-dimethylbenzene (also known as p-xylene) has the formula (CH3)2C6H4. Its structure has two CH3 (methyl) groups substituted at opposite positions on the benzene (C6H6) ring. Predict the number of peaks in the low-resolution proton NMR spectrum of this compound and the relative areas of the peaks.arrow_forward
- In a proton NMR spectrum, indicate the number of peaks and their multiplicity for the following compounds: CH3 - CH2 - CO - CH3 para-chloromethylbenzenearrow_forwardA compound with molecular formula C5H10O2has the following NMR spectrum. Determine the number of protons giving rise to the signals at 2.0 ppm and 4.0 ppm.arrow_forwardThe following 1H NMR spectra are for four compounds, each with molecular formula of C6H12O2. Identify the compounds.arrow_forward
- For the following compound how many different signals would you see in the carbon NMR? (Assume that you can see them all.) 4 O 5 O 3 09 CO |arrow_forwardShown below is the H-NMR spectra of acetaminophen. Draw the structure and correlate each hydrogen in the molecule with the various peaks in the spectra.arrow_forwardAre there signals that you can assign to specific protons in the molecule (labeled a-k)? Complete this column of the table. It may not be possible to assign each signal to a specific proton.arrow_forward
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY





